Abstract
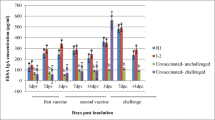
Layer can be used as a biological source to produce specific antibodies called immunoglobulin Y (IgY) for various infectious diseases. This research aims to produce influenza-specific IgY in layers vaccinated with quadrivalent influenza vaccine and determine its cross-reaction ability with other influenza A subtypes. Three Hy-Line Brown layers at 24 weeks of age were used in this study, and influenza-specific antibodies in serum and egg yolk were evaluated from the 1 to 20 weeks after initial vaccination. The presence of influenza-specific antibodies in serum and egg yolk was identified using agar gel precipitation (AGP) test and hemagglutination inhibition (HI). Serum neutralization (SN) test was carried out to determine influenza antibodies ability to cross-neutralize other influenza virus subtypes. At 10 weeks post-first vaccination (p.f.v) was shown to have the highest mean agar gel precipitation (AGP) titer in serum (24.7), and the highest AGP titer in egg yolk was shown at 5 weeks p.f.v. The highest HI geometric mean titer (GMT) of A/H1N1 antibody reached 26.67 HIU at 9 weeks p.f.v., while A/H3N2 antibody reached 26 HIU at 6 weeks p.f.v. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) result of purified antibodies from egg yolk indicated the presence of whole IgY structure showing a protein band at ± 188 kDa. Cross-reactivity with influenza A/H5N1 and A/H9N2 was seen in AGP test results, while cross-neutralization with influenza A/H5N1 was observed in SN results. Cross-reactivity and cross-neutralization shown by our resulting antibodies indicating broad-spectrum protection against various influenza virus subtypes.




Similar content being viewed by others
References
Moghadami M (2017) A Narrative review of influenza: a seasonal and pandemic disease. Iran J Med Sci 42(1):2–13
Chow EJ, Doyle JD, Uyeki TM (2019) Influenza virus-related critical illness: prevention, diagnosis, treatment. Crit Care 23(214):1–11. https://doi.org/10.1186/s13054-019-2491-9
Barry MA, Aljammaz KI, Alrashed AA (2020) Knowledge, attitude, and barriers influencing seasonal influenza vaccination uptake. Can J Infect Dis Med Microbiol 2020:1–6. https://doi.org/10.1155/2020/7653745
Marshall JS, Warrington R, Watson W, Kim HL (2018) An introduction to immunology and immunopathology. Allergy Asthma Clin Immunol 14(2):5–14. https://doi.org/10.1186/s13223-018-0278-1
Bentes GA, Lanzarini NM, Lima LRP, Manso PPA, da Silva ADS, Junior SDSM, Guimarães JR, de Moraes MTB, Pelajo-Machado M, Pinto MA (2015) Using immunoglobulin Y as an alternative antibody for the detection of hepatitis A virus in frozen liver sections. Mem Inst Oswaldo Cruz 110(4):577–579. https://doi.org/10.1590/0074-02760140457
Beltrão MROC, Alencar CAS, Leite AS, Freitas LT, Gonzalez JC, Santana VLA, Manso—Filho HS, (2015) Comparison of two protocols of agar gel immunodiffusion (AGID) used to diagnose of equine infectious anemia (EIA). Open J Vet Med 5:169–174. https://doi.org/10.4236/ojvm.2015.57023
OIE Terrestrial Manual (2021) Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2021, chapter 3.3.4 https://www.oie.int/fileadmin/Home/fr/Health_standards/tahm/3.03.04_AI.pdf.
Redwan EM, Aljadawi AA, Uversky VN (2020) Simple and efficient protocol for immunoglobulin Y purification from chicken egg yolk. Poult Sci 100(3):1–8. https://doi.org/10.1016/j.psj.2020.12.053
El-Husseiny MH, Hagag NM, Pushko P, Tretyakova I, Naguib MM, Arafa AS (2021) Evaluation of protective efficacy of influenza virus like particles prepared from H5N1 virus of clade 2212 in Chickens. Vaccines 9(715):1–7. https://doi.org/10.3390/vaccines9070715
Frasca D, Diaz A, Romero M, Blomberg BB (2016) The generation of memory B cells is maintained, but the antibody response is not, in the elderly after repeated influenza immunizations. Vaccine 34(25):2834–2840. https://doi.org/10.1016/j.vaccine.2016.04.023
Slifka MK, Amanna IJ (2019) Role of multivalency and antigenic threshold in generating protective antibody responses. Front Immunol 10(956):1–16. https://doi.org/10.3389/fimmu.2019.00956
Pauly D, Chacana PA, Calzado EG, Brembs B, Schade R (2011) IgY technology: extraction of chicken antibodies from egg yolk by polyethylene glycol (PEG) precipitation. J Vis Exp 51:1–6. https://doi.org/10.3791/3084
Ulmer-Franco AM, Cherian G, Quezada N, Fasenko GM, McMullen LM (2012) Hatching egg and newly hatched chick yolk sac total IgY content at 3 broiler breeder flock ages. Poult Sci J 91:758–764. https://doi.org/10.3382/ps.2011-01757
Kowalczyk J, Smialek M, Tykalowski B, Dziewulska D, Stenzel T, Koncicki A (2019) Field evaluation of maternal antibody transfer from breeder turkey hens to egg yolks, egg whites, and poults. Poult Sci 98(8):3150–3157. https://doi.org/10.3382/ps/pez126
Agrawal R, Hirpurkar SD, Sannt C, Gupta AK (2016) Comparative study on immunoglobulin Y transfer from breeding hens to egg yolk and progeny chicks in different breeds of poultry. Vet World 9(4):425–431. https://doi.org/10.14202/vetworld.2016.425-431
Amro WA, Al-Qaisi W, Al-Razem F (2018) Production and purification of IgY antibodies from chicken egg yolk. J Genet Eng Biotechnol 16(1):99–103. https://doi.org/10.1016/j.jgeb.2017.10.003
Lee L, Samardzic K, Wallach M, Frumkin LR, Mochly-Rosen D (2021) Immunoglobulin Y for potential diagnostic and therapeutic applications in infectious diseases. Front Immunol 12:1–23. https://doi.org/10.3389/fimmu.2021.696003
Chen YQ, Wohlbold TJ, Zheng NY, Huang M, Huang Y, Neu KE, Lee J, Wan H, Rojas KT, Kirkpatrick E, Henry C et al (2018) Influenza infection in humans induces broadly cross-reactive and protective neuraminidase-reactive antibodies. Cell 173(2):417–429. https://doi.org/10.1016/j.cell.2018.03.030
Wallach MG, Webby RJ, Islam F, Walkden-Brown S, Emmoth E, Feinstein R, Gronvik KO (2011) Cross-protection of chicken immunoglobulin Y antibodies against H5N1 and H1N1 viruses passively administered in mice. Clin Vaccine Immunol 18(7):1083–1090. https://doi.org/10.1128/CVI.05075-11
Adachi K, Takama K, Tsukamoto M, Inai M, Handharyani E, Hiroi S, Tsukamoto Y (2010) Ostrich produce cross-reactive neutralization antibodies against pandemic influenza virus A/H1N1 following immunization with a seasonal influenza vaccine. Exp Ther Med 2(1):41–45. https://doi.org/10.3892/etm.2010.180
Acknowledgements
We want to thank Ministry of Research and Innovation Agency of Republic Indonesia for supporting our research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Significance statements: Our result study is significant to show that layers are able to produce equivalent antibody titer for each vaccine virus strain, which indicate that one layer is able to produce several specific antibodies.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saputri, M.E., Esfandiari, A., Rachmawan, W.P. et al. Influenza Immunoglobulin (Ig) Y Derived from Chicken Egg Yolk: Production, Characterization, and Its Cross-Reactivity. Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 94, 23–30 (2024). https://doi.org/10.1007/s40011-023-01510-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40011-023-01510-2