Abstract
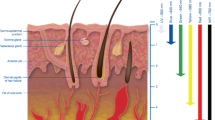
Understanding the thermal response of laser-irradiated biological samples is important to optimize the efficiency of photo-thermal therapy for selective destruction of cancerous cells. The present review article discusses the attempts that have been made to numerically model the process of light propagation through biological tissue samples and to understand the thermal response of such laser-irradiated samples. Numerical approaches for solving the classical form of radiative transfer equation (RTE) and the subsequent coupling of the solution of RTE with various bio-heat transfer models (Fourier as well as non-Fourier) for finding the temperature distribution within the volume of the tissue phantom that is subjected to laser-irradiation are discussed. Applications of contrast enhancing reagents (for instance, gold nanorods and/or nanoshells) for improving the therapeutic performance of laser-based photo-thermal therapy is also highlighted.










Similar content being viewed by others
Notes
One of the major implications of using short pulse lasers as the light source during numerical modeling of thermal response of laser-irradiated tissue samples comes in the form that due to the inherent time lag between the driving potential (incident laser pulses) and the response of the irradiated sample (temperature changes), the assumption of infinite speed of propagation of thermal front in the medium cannot be strictly followed and hence one has to use non-Fourier heat conduction models for accurately modeling the thermal response of biological tissue samples. Such developments are discussed in the latter half of this article.
References
Chandrasekhar S (1960) Radiative transfer. Dover Publications Inc., New York
Mitra K, Kumar S (1999) Development and comparison of models for light-pulse transport through scattering-absorbing media. Appl Opt 38:188–196
Mishra SC, Chugh P, Kumar P, Mitra K (2006) Development and comparison of the DTM, the DOM and the FVM formulations for the short-pulse laser transport through a participating medium. Int J Heat Mass Transf 49:1820–1832
Hunter B, Guo Z (2011) Comparison of the discrete-ordinates method and the finite-volume method for steady-state and ultrafast radiative transfer analysis in cylindrical coordinates. Numer Heat Transf B 59:339–359
Dombrovsky LA, Timchenko V, Jackson M (2012) Indirect heating strategy of laser induced hyperthermia: an advanced thermal model. Int J Heat Mass Transf 55:4688–4700
Crezee J, Lagendijk JJW (1992) Temperature uniformity during hyperthermia: the impact of large vessels. Phys Med Biol 37:1321–1337
Kolios MC, Sherar MD, Hunt JW (1996) Blood flow cooling and ultrasonic lesion formation. Med Phys 23:1287–1298
Dua R, Chakraborty S (2005) A novel modeling and simulation technique of photo–thermal interactions between lasers and living biological tissues undergoing multiple changes in phase. Comput Biol Med 35:447–462
Kumar S, Srivastava A (2014) Numerical investigation of thermal response of laser irradiated tissue phantoms embedded with optical inhomogeneities. Int J Heat Mass Transf 77:262–277
Lagendijk JJW (1982) The influence of blood flow in large vessels on the temperature distribution in hyperthermia. Phys Med Biol 27:17–23
Shih TC, Liu HL, Horng ATL (2006) Cooling effect of thermally significant blood vessels in perfused tumor tissue during thermal therapy. Int Commun Heat Mass Transf 33:135–141
Craciunescu OI, Clegg ST (2001) Pulsatile blood flow effects on temperature distribution and heat transfer in rigid vessels. ASME J Biomech Eng 123:500–505
Horng TL, Lin WL, Liauh CT, Shih TC (2007) Effects of pulsatile blood flow in large vessels on thermal dose distribution during thermal therapy. Med Phys 34:1312–1320
Kumar S, Srivastava Atul (2016) Numerical investigation of the influence of pulsatile blood flow on temperature distribution within the body of laser-irradiated biological tissue phantoms. Int J Heat Mass Transf 95:662–677
Bayazitoglu Y, Kheradmand S, Tullius TK (2013) An overview of nanoparticle assisted laser therapy. Int J Heat Mass Transf 67:469–486
Hirsch LR, Stafford RJ, Bankson JA, Sershen SR, Rivera B, Price RE, Hazle JD, HalasN J, West JL (2003) Nanoshell-mediatednear-infraredthermal therapyoftumorsundermagneticresonanceguidance. Proc Natl Acad Sci USA 100:13549–13554
Soni S, Tyagi H, Taylor RA, Kumar A (2013) Role of optical coefficients and healthy tissue-sparing characteristics in gold nanorod-assisted thermal therapy. Int J Hyperth 29:87–97
Soni S, Tyagi H, Taylor RA, Kumar A (2014) Investigation on nanoparticle distribution for thermal ablation of a tumour subjected to nanoparticle assisted thermal therapy. J Therm Biol 43:70–80
Soni S, Tyagi H, Taylor RA, Kumar A (2015) The influence of tumour blood perfusion variability on thermal damage during nanoparticle-assisted thermal therapy. Int J Hyperth 31:615–625
Welch AJ, Gemert MJV (2010) Optical-thermal response of laser-irradiated tissue, 2nd edn. Springer, New York
Minkowycz WJ, Sparrow EM, Abraham JP (2009) Advances in numerical heat transfer. CRC Press, Boca Raton
Das C, Trivedi A, Mitra K, Vo-Dinh T (2003) Experimental and numerical analysis of short-pulse laser interaction with tissue phantoms containing inhomogeneities. Appl Opt 42:5173–5180
Fanjul-Vélez F, Romanov OG, Arce-Diego JL (2009) Efficient 3D numerical approach for temperature prediction in laser irradiated biological tissues. Comput Biol Med 39:810–817
Singh R, Das K, Mishra SC (2014) Laser-induced hyperthermia of nanoshell mediated vascularized tissue: a numerical study. J Thermal Biol 44:55–62
Singh R, Das K, Okajima J, Maruyama S, Mishra SC (2015) Modelling skin cooling using optical windows and cryogens during laser induced hyperthermia in a multilayer vascularized tissue. Appl Therm Eng 89:28–35
Cattaneo C (1958) A form of heat conduction equation which eliminates the paradox of instantaneous propagation. Compt Rend 247:431–433
Vernotte P (1961) Some possible complications in the phenomena of thermal conduction. Compt Rend. 252:2190–2191
Tzou DY (1995) A unified field approach for heat conduction from macro- to micro-scales. ASME J Heat Transf 117:8–16
Liu K-C (2011) Nonlinear behavior of thermal lagging in concentric living tissues with Gaussian distribution source. Int J Heat Mass Transf 54:2829–2836
Kumar S, Srivastava Atul (2015) Thermal analysis of laser-irradiated tissue phantoms using dual phase lag model coupled with transient radiative transfer equation. Int J Heat Mass Transf 90:466–479
Narasimhan A, Sadasivam S (2013) Non-Fourier bio heat transfer modelling of thermal damage during retinal laser irradiation. Int J Heat Mass Transf 60:591–597
Sahoo N, Ghosh S, Narasimhan A, Das SK (2014) Investigation of non-Fourier effects in bio-tissues during laser assisted photothermal therapy. Int J Therm Sci 76:208–220
Acknowledgements
I express my sincere gratitude to Prof. P.K. Gupta for inviting me to write this review article and for providing useful comments/suggestions for improving the technical contents and the readability of the article. Some of the material covered in the review article is based on the Ph.D. research work of Dr. Sumit Kumar. The author acknowledges the valuable inputs of Dr. Sumit Kumar in this context.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Srivastava, A. Modelling of Laser-Induced Heating of Tissues for Therapeutic Applications. Proc. Natl. Acad. Sci., India, Sect. A Phys. Sci. 88, 461–472 (2018). https://doi.org/10.1007/s40010-018-0520-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40010-018-0520-8