Abstract
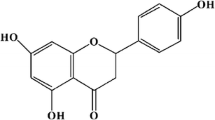
This study was aimed at developing a naringenin formulation to enhance its solubility and intestinal permeability and, thereby, overcome its low bioavailability. The naringenin-loaded mixed micelle formulation with a naringenin:Pluronic F127:Tween 80 ratio of 1:10:0.2 (w/w/w) was prepared using a thin-film hydration method. The solubility, size distribution, and cell viability were characterized. Subsequently, in vivo pharmacokinetic parameters of the naringenin-loaded mixed-micelle formulation were investigated in rats. The formulation increased the solubility of naringenin by 27-fold without a significant decrease in the viability of treated cells. The absolute bioavailability of naringenin dramatically increased from 4.1 to 26.9 % following per oral administration of naringenin-loaded mixed-micelle form. Absorption permeability of naringenin from the developed formulation increased by 1.7-fold compared to that of naringenin administered alone. In conclusion, the enhanced oral bioavailability of naringenin from the loaded mixed micelle formulation can be attributed to its increased solubility and intestinal permeation.




Similar content being viewed by others
References
Batrakova EV, Kabanov AV (2008) Pluronic block copolymers: evolution of drug delivery concept from inert nanocarriers to biological response modifiers. J Control Release 130:98–106
Bodet C, La VD, Epifano F, Grenier D (2008) Naringenin has anti-inflammatory properties in macrophage and ex vivo human whole-blood models. J Periodontal Res 43:400–407
Bromberg L, Alakhov V (2003) Effects of polyether-modified poly(acrylic acid) microgels on doxorubicin transport in human intestinal epithelial Caco-2 cell layers. J Control Release 88:11–22
Choi MK, Song IS (2012) Characterization of efflux transport of the PDE5 inhibitors, vardenafil and sildenafil. J Pharm Pharmacol 64:1074–1083
Choi YA, Yoon YH, Choi K, Kwon M, Goo SH, Cha JS, Choi MK, Lee HS, Song IS (2015) Enhanced oral bioavailability of morin administered in mixed micelle formulation with PluronicF127 and Tween80 in rats. Biol Pharm Bull 38:208–217
Deferme S, Augustijns P (2003) The effect of food components on the absorption of P-gp substrates: a review. J Pharm Pharmacol 55:153–162
Deferme S, Van Gelder J, Augustijns P (2002) Inhibitory effect of fruit extracts on P-glycoprotein-related efflux carriers: an in-vitro screening. J Pharm Pharmacol 54:1213–1219
Harmon AW, Patel YM (2004) Naringenin inhibits glucose uptake in MCF-7 breast cancer cells: a mechanism for impaired cellular proliferation. Breast Cancer Res Treat 85:103–110
Kabanov AV, Batrakova EV, Miller DW (2003) Pluronic block copolymers as modulators of drug efflux transporter activity in the blood-brain barrier. Adv Drug Deliv Rev 55:151–164
Kanaze FI, Bounartzi MI, Georgarakis M, Niopas I (2007) Pharmacokinetics of the citrus flavanone aglycones hesperetin and naringenin after single oral administration in human subjects. Eur J Clin Nutr 61:472–477
Kim AR, Lim SJ, Lee BJ (2009) Metabolic inhibition and kinetics of raloxifene by pharmaceutical excipients in human liver microsomes. Int J Pharm 368:37–44
Kim DH, Jung EA, Sohng IS, Han JA, Kim TH, Han MJ (1998) Intestinal bacterial metabolism of flavonoids and its relation to some biological activities. Arch Pharm Res 21:17–23
Kim HP, Son KH, Chang HW, Kang SS (2004) Anti-inflammatory plant flavonoids and cellular action mechanisms. J Pharmacol Sci 96:229–245
Lee SH, Park YB, Bae KH, Bok SH, Kwon YK, Lee ES, Choi MS (1999) Cholesterol-lowering activity of naringenin via inhibition of 3-hydroxy-3-methylglutaryl coenzyme A reductase and acyl coenzyme A:cholesterol acyltransferase in rats. Ann Nutr Metab 43:173–180
Lin H, Gebhardt M, Bian S, Kwon KA, Shim CK, Chung SJ, Kim DD (2007) Enhancing effect of surfactants on fexofenadine.HCl transport across the human nasal epithelial cell monolayer. Int J Pharm 330:23–31
Nait Chabane M, Al Ahmad A, Peluso J, Muller CD, Ubeaud G (2009) Quercetin and naringenin transport across human intestinal Caco-2 cells. J Pharm Pharmacol 61:1473–1483
Peng HW, Cheng FC, Huang YT, Chen CF, Tsai TH (1998) Determination of naringenin and its glucuronide conjugate in rat plasma and brain tissue by high-performance liquid chromatography. J Chromatogr B 714:369–374
Takanaga H, Ohnishi A, Matsuo H, Sawada Y (1998) Inhibition of vinblastine efflux mediated by P-glycoprotein by grapefruit juice components in caco-2 cells. Biol Pharm Bull 21:1062–1066
Yang S, Liu J, Chen Y, Jiang J (2012) Reversal effect of Tween-20 on multidrug resistance in tumor cells in vitro. Biomed Pharmacother 66:187–194
Zhang W, Parniak MA, Sarafianos SG, Empey PE, Rohan LC (2014) In vitro transport characteristics of EFdA, a novel nucleoside reverse transcriptase inhibitor using Caco-2 and MDCKII cell monolayers. Eur J Pharmacol 732:86–95
Zhou J, Zhou M, Yang FF, Liu CY, Pan RL, Chang Q, Liu XM, Liao YH (2015) Involvement of the inhibition of intestinal glucuronidation in enhancing the oral bioavailability of resveratrol by labrasol containing nanoemulsions. Mol Pharm 12:1084–1095
Zhou S, Lim LY, Chowbay B (2004) Herbal modulation of P-glycoprotein. Drug Metab Rev 36:57–104
Acknowledgments
All authors (I.-S. Song, J.-S. Cha, M.-K. Choi) declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, IS., Cha, JS. & Choi, MK. Enhanced oral bioavailability of naringenin administered in a mixed micelle formulation with Pluronic F127 and Tween 80 in rats. Journal of Pharmaceutical Investigation 45, 633–640 (2015). https://doi.org/10.1007/s40005-015-0216-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-015-0216-x