Abstract
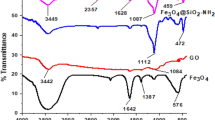
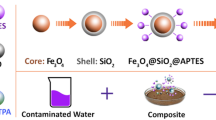
This study describes the use of a ternary composite of Fe3O4, chitosan and graphene quantum dots (Fe3O4/GQDs-Chit) as an efficient magnetic adsorbent for the removal of Pb2+ from aqueous solutions. A number of methods were employed to describe the prepared adsorbent including scanning and transmission electron microscopy, vibrating sample magnetometer, Fourier-transform infrared spectroscopy and X-ray diffraction. Removal experiments were conducted in a batch system to optimize adsorbent amount, initial Pb2+ concentration, solution pH, and contact time. To explore the equilibrium isotherm and calculate the isotherm constants, Langmuir and Freundlich adsorption models were used. Accordingly, the Fe3O4/GQDs-Chit were successful in removing Pb2+ up to 117.65 mg g−1 from water, with good agreement to Langmuir isotherm model (R2 = 0.9957). The adsorption kinetic was well fitted (R2 > 0.9940) with pseudo-second-order model indicating chemical adsorption as the rate-limiting step in the adsorption process. Furthermore, thermodynamic parameters revealed that the adsorption of Pb2+ on the Fe3O4/GQDs-Chit was exothermic and spontaneous. As evidenced by regeneration experiments, Fe3O4/GQDs-Chit were a highly stable and reproducible adsorbent, which can function as an efficient adsorbent in water treatment.











Similar content being viewed by others
References
Ahadi N, Askari S, Fouladitajar A, Akbari (2022) Facile synthesis of hierarchically structured mil-53(al) with superior properties using an environmentally-friendly ultrasonic method for separating lead ions from aqueous solutions. Sci Rep 12(1):2649. https://doi.org/10.1038/s41598-022-06518-8
Alkan M, Demirbas O, Dogan M (2007) Adsorption kinetics and thermodynamics of an anionic dye onto sepiolite. Microporous Mesoporous Mater 101(3):388–396. https://doi.org/10.1016/j.micromeso.2006.12.007
Chin JF, Heng ZW, Teoh HC et al (2022) Recent development of magnetic biochar crosslinked chitosan on heavy metal removal from wastewater-modification, application and mechanism. Chemosphere 291:133035. https://doi.org/10.1016/j.chemosphere.2021.133035
Dong Y, Shao J, Chen C et al (2012) Blue luminescent graphene quantum dots and graphene oxide prepared by tuning the carbonization degree of citric acid. Carbon 50(12):4738–4743. https://doi.org/10.1016/j.carbon.2012.06.002
El-Wakeel ST, El-Tawil RS, HaM A et al (2017) Synthesis and structural properties of MnO2 as adsorbent for the removal of lead (Pb2+) from aqueous solution. J Taiwan Inst Chem Eng 72:95–103. https://doi.org/10.1016/j.jtice.2017.01.008
Ghaffarkhah A, Hosseini E, Kamkar M et al (2022) Synthesis, applications, and prospects of graphene quantum dots: a comprehensive review. Small 18(2):2102683. https://doi.org/10.1002/smll.202102683
Grant LD (2020) Lead and compounds. Environmental toxicants: human exposures and their health effects. Wiley, New York, pp 627–675. https://doi.org/10.1002/9781119438922.ch17
Hadi Najafabadi H, Irani M, Roshanfekr Rad L et al (2015) Removal of Cu2+, Pb2+ and Cr6+ from aqueous solutions using a chitosan/graphene oxide composite nanofibrous adsorbent. RSC Adv 5(21):16532–16539. https://doi.org/10.1039/C5RA01500F
Hou L, Yang C, Rao X et al (2021) Fabrication of recoverable magnetic surface ion-imprinted polymer based on graphene oxide for fast and selective removal of lead ions from aqueous solution. Colloids Surf A Physicochem Eng Asp 625:126949. https://doi.org/10.1016/j.colsurfa.2021.126949
Januario EFD, Vidovix TB, Beluci NDCL et al (2021) Advanced graphene oxide-based membranes as a potential alternative for dyes removal: a review. Sci Total Environ 789:147957. https://doi.org/10.1016/j.scitotenv.2021.147957
Jiang H, Cao Y, Zeng F et al (2021) A novel Fe3O4/graphene oxide composite prepared by click chemistry for high-efficiency removal of Congo red from water. J Nanomater 2021:9716897. https://doi.org/10.1155/2021/9716897
Jiang S, Yan L, Wang R et al (2022) Recyclable nitrogen-doped biochar via low-temperature pyrolysis for enhanced lead(II) removal. Chemosphere 286:131666. https://doi.org/10.1016/j.chemosphere.2021.131666
Kucherova AE, Romantsova IV, Burakov AE et al (2017) Graphene-based nanocomposites for enhanced Pb2+ adsorption. Nano Hybrids Compos 13:323–329. https://doi.org/10.4028/www.scientific.net/NHC.13.323
Li LS, Yan X (2010) Colloidal graphene quantum dots. J Phys Chem Lett 1(17):2572–2576. https://doi.org/10.1021/jz100862f
Liu X, Ma R, Wang X et al (2019) Graphene oxide-based materials for efficient removal of heavy metal ions from aqueous solution: a review. Environ Pollut 252:62–73. https://doi.org/10.1016/j.envpol.2019.05.050
Mahvi AH, Balarak D, Bazrafshan E (2021) Remarkable reusability of magnetic Fe3O4-graphene oxide composite: a highly effective adsorbent for Cr(VI) ions. Int J Environ Anal Chem. https://doi.org/10.1080/03067319.2021.1910250
Mal J, Sinharoy A, Lens PNL (2021) Simultaneous removal of lead and selenium through biomineralization as lead selenide by anaerobic granular sludge. J Hazard Mater 420:126663. https://doi.org/10.1016/j.jhazmat.2021.126663
Mao J, Liu J, Zhang Z et al (2021) High adsorption capacity and selectivity of layered metal sulfide (kzts) for effective removal of lead ions from wastewater. J Mater Sci 56(32):18233–18247. https://doi.org/10.1007/s10853-021-06486-w
Mohammad-Rezaei R, Jaymand M (2019) Graphene quantum dots coated on quartz sand as efficient and low-cost adsorbent for removal of Hg2+ and Pb2+ from aqueous solutions. Environ Prog Sustain Energy 38(s1):S24–S31. https://doi.org/10.1002/ep.12911
Mustapha S, Shuaib DT, Ndamitso MM et al (2019) Adsorption isotherm, kinetic and thermodynamic studies for the removal of Pb(II), Cd(II), Zn(II) and Cu(II) ions from aqueous solutions using albizia lebbeck pods. Appl Water Sci 9(6):142. https://doi.org/10.1007/s13201-019-1021-x
Nuengmatcha P (2021) Mercapto-functionalized magnetic graphene quantum dots as adsorbent for Cd2+ removal from wastewater. Environ Process 8(3):1289–1306. https://doi.org/10.1007/s40710-021-00523-1
Omer AM, Dey R, Eltaweil AS, Abd El-Monaem EM, Ziora ZM (2022) Insights into recent advances of chitosan-based adsorbents for sustainable removal of heavy metals and anions. Arab J Chem 15(2):103543. https://doi.org/10.1016/j.arabjc.2021.103543
Revellame ED, Fortela DL, Sharp W et al (2020) Adsorption kinetic modeling using pseudo-first order and pseudo-second order rate laws: a review. Clean Eng Technol 1:100032. https://doi.org/10.1016/j.clet.2020.100032
Rezania S, Mojiri A, Park J et al (2022) Removal of lead ions from wastewater using lanthanum sulfide nanoparticle decorated over magnetic graphene oxide. Environ Res 204:111959. https://doi.org/10.1016/j.envres.2021.111959
Rusmin R, Sarkar B, Mukhopadhyay R et al (2022) Facile one pot preparation of magnetic chitosan–palygorskite nanocomposite for efficient removal of lead from water. J Colloid Interface Sci 608:575–587. https://doi.org/10.1016/j.jcis.2021.09.109
Shahzad A, Miran W, Rasool K et al (2017) Heavy metals removal by edta-functionalized chitosan graphene oxide nanocomposites. RSC Adv 7(16):9764–9771. https://doi.org/10.1039/C6RA28406J
Sharma P, Singh AK, Shahi VK (2019) Selective adsorption of Pb(II) from aqueous medium by cross-linked chitosan-functionalized graphene oxide adsorbent. ACS Sustain Chem Eng 7(1):1427–1436. https://doi.org/10.1021/acssuschemeng.8b05138
Shen J, Zhu Y, Yang X, Li C (2012) Graphene quantum dots: emergent nanolights for bioimaging, sensors, catalysis and photovoltaic devices. Chem Commun 48(31):3686–3699. https://doi.org/10.1039/C2CC00110A
Shen Y, Jiang B, Xing Y (2021) Recent advances in the application of magnetic Fe3O4 nanomaterials for the removal of emerging contaminants. Environ Sci Pollut Res 28(7):7599–7620. https://doi.org/10.1007/s11356-020-11877-8
Tshangana CS, Muleja AA, Kuvarega AT et al (2021) The applications of graphene oxide quantum dots in the removal of emerging pollutants in water: an overview. J Water Process Eng 43:102249. https://doi.org/10.1016/j.jwpe.2021.102249
Van Lam P, Duong NB, Trang QTT, Tuan VA (2018) Removal of Pb2+ and Cd2+ ions from aqueous solutions using Fe3O4/bentonite nanocomposite. Vietnam J Chem 56(5):617–622. https://doi.org/10.1002/vjch.201800058
Wang J, Zhang J, Han L et al (2021) Graphene-based materials for adsorptive removal of pollutants from water and underlying interaction mechanism. Adv Colloid Interface Sci 289:102360. https://doi.org/10.1016/j.cis.2021.102360
Wu X, Zhang Y, Han T et al (2014) Composite of graphene quantum dots and Fe3O4 nanoparticles: Peroxidase activity and application in phenolic compound removal. RSC Adv 4(7):3299–3305. https://doi.org/10.1039/C3RA44709J
Yap PL, Nine MJ, Hassan K et al (2021) Graphene-based sorbents for multipollutants removal in water: a review of recent progress. Adv Funct Mater 31(9):2007356. https://doi.org/10.1002/adfm.202007356
Zarenezhad M, Zarei M, Ebratkhahan M, Hosseinzadeh M (2021) Synthesis and study of functionalized magnetic graphene oxide for Pb2+ removal from wastewater. Environ Technol Inno 22:101384. https://doi.org/10.1016/j.eti.2021.101384
Zaynab M, Al-Yahyai R, Ameen A et al (2022) Health and environmental effects of heavy metals. J King Saud Univ Sci 34(1):101653. https://doi.org/10.1016/j.jksus.2021.101653
Zhang Q, Wu R, Zhou Y et al (2022) A novel surface-oxidized rigid carbon foam with hierarchical macro-nanoporous structure for efficient removal of malachite green and lead ion. J Mater Sci Technol 103:15–28. https://doi.org/10.1016/j.jmst.2021.07.012
Zhang W, An Y, Li S et al (2020) Enhanced heavy metal removal from an aqueous environment using an eco-friendly and sustainable adsorbent. Sci Rep 10(1):16453. https://doi.org/10.1038/s41598-020-73570-7
Zhong L, Zhang Q, Sun M et al (2016) Fabrication and characterization of polyethyleneimine immobilized on chloropropyl- and silica-coated magnetic nanoparticles for Pb2+ removal from aqueous solution. Desalin Water Treat 57(29):13701–13710. https://doi.org/10.1080/19443994.2015.1060535
Zhou Y, Fan Y, Zhang J et al (2021) Topological tuning of two-dimensional polytriazine imides by halide anions for selective lead removal from wastewater. Sep Purif Technol 278:119595. https://doi.org/10.1016/j.seppur.2021.119595
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Editorial responsibility: Jing Chen.
Rights and permissions
About this article
Cite this article
Sedaghatian, AR., Marjani, A., Joshaghani, A.H. et al. Synthesis of magnetic graphene quantum dots–chitosan nanocomposite: an efficient adsorbent for removal of Pb2+ from aqueous solution. Int. J. Environ. Sci. Technol. 19, 11447–11458 (2022). https://doi.org/10.1007/s13762-022-04368-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-04368-5