Abstract
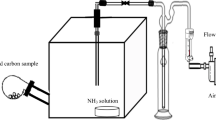
Indoor air pollution is becoming more prominent, which requires an urgent need to develop cost-effective adsorption materials. In this work, a novel and bimodal composite of diatomite (Dt) and activated carbon (AC) was developed. Then copper was impregnated on the surface of the diatomite-activated carbon composite (Cu/Dt-AC) for the adsorption of indoor air pollutants (Trimethylamine (TMA) and NH3). The Dt-AC composites contained the inherent characteristics of Dt and AC, showing the bimodal and hierarchically porous structures. The mass ratio of Dt-AC can significantly affect BET (Brunauer Emmet and Teller) surface area and pore size of the Dt-AC composite. Cu-loading can generate many surface functional groups (especially oxygen-containing functional groups –OH, –C–O, O–Si–O, SiO–H), thereby greatly improving the adsorption efficiency. The optimum copper proportion on TMA adsorption performance was 22 wt.% regardless of the ratios of Dt-AC whereas the best NH3 adsorption performance was obtained with a ratio of 1:3 and 22 wt.% of copper. The maximum adsorption capacities of TMA and NH3 were 185.59 and 37.48 mg/g, respectively. The adsorption process of TMA and NH3 well followed the Langmuir isotherm and pseudo-second-order kinetics models. Furthermore, both the intraparticle diffusion and film diffusion models jointly controlled the adsorption mechanism of TMA and NH3 on the Cu/Dt-AC filter. The spent Cu/Dt-AC filter can be regenerated, and its adsorption efficiency remained above 90% for TMA and above 80% for NH3 over five cycles.











Similar content being viewed by others
Abbreviations
- AC:
-
Activated carbon
- BET:
-
Brunauer Emmet and Teller
- Cu/Dt-AC:
-
Copper impregnated diatomite-activated carbon composite
- Dt:
-
Diatomite
- Dt-AC:
-
Diatomite-activated carbon composite
- FE-SEM:
-
Field emission scanning electron microscope
- FTIR:
-
Fourier transform infrared spectroscopy
- NLDFT:
-
Nonlocal density functional theory
- PET:
-
Polyethylene terephthalate
- SEM–EDS:
-
Scanning electron microscopy with energy-dispersive spectroscopy
- TGA:
-
Thermogravimetric analysis
- TMA:
-
Trimethylamine
- VOCs:
-
Volatile organic compounds
- XRD:
-
X-ray diffraction
- XRF:
-
X-ray fluorescence
References
Activated Carbon Market, “Activated Carbon Market by Type (Powdered, Granular, Others (Pelletized, Bead)), Application (Liquid Phase (Water Treatment, Foods & Beverages, Pharmaceutical & Medical), Gaseous Phase (Industrial, Automotive)), Region—Global Forecast to 2021” https://www.marketsandmarkets.com/Market-Reports/activated-carbon-362.html, 2021 (accessed 12 January 2021)
Al-Ghouti MA, Da’ana DA (2020) Guidelines for the use and interpretation of adsorption isotherm models: a review. J Hazard Mater 393:122383. https://doi.org/10.1016/j.jhazmat.2020.122383
Anitha T, Kumar PS, Kumar KS (2016) Synthesis of nano-sized chitosan blended polyvinyl alcohol for theremoval of Eosin Yellow dye from aqueous solution. J Water Process Eng 13:127–136. https://doi.org/10.1016/j.jwpe.2016.08.003
Bedolla PO, Feldbauer G, Wolloch M, Eder SJ, Dorr N, Mohn P, Redinger J, Vernes A (2014) Effects of van der Waals Interactions in the Adsorption of Isooctane and Ethanol on Fe (100) surfaces. J Phys Chem C Nanomater Interfaces 118(31):17608–17615. https://doi.org/10.1021/jp503829c
Boraphech P, Thiravetyan P (2015) Trimethylamine (fishy odor) adsorption by biomaterials: effect of fatty acids, alkanes, and aromatic compounds in waxes. J Hazard Mater 284:269–277. https://doi.org/10.1016/j.jhazmat.2014.11.014
Boyd GE, Adamson AW, Myers LS (1947) The exchange adsorption of ions fromaqueous solutions by organic zeolites II. Kinetics J Am Chem Soc 69:2836–2848. https://doi.org/10.1021/ja01203a066
Celebi H (2020) Use of bioballs as an adsorbent for the removal of copper. J Chem Soc Pak 43(2):114–123
Çelebi Ö, Şimşek İ, Celebi H (2021) Escherichia coli inhibition and arsenic removal from aqueous solutions using raw eggshell matrix. Int J Environ Sci Technol 18:3205–3220. https://doi.org/10.1007/s13762-021-03216-2
Chi C, Chen W, Guo M, Weng M, Yan G, Shen X (2016) Law and features of TVOC and Formaldehyde pollution in urban indoor air. Atmospheric Environ 132:85–90. https://doi.org/10.1016/j.atmosenv.2016.02.043
ChungLee K-HK-Y (2009) Removal of trimethylamine by adsorption over zeolite catalysts and deodorization of fish oil. J Hazard Mater 172:922–927. https://doi.org/10.1016/j.jhazmat.2009.07.081
Deng L, Du P, Yu W, Yuan P, Annabi-Bergaya F, Liu D, Zhou J (2019) Novel hierarchically porous allophane/diatomite nanocomposite for benzene adsorption. Appl Clay Sci 168:155–163. https://doi.org/10.1016/j.clay.2018.11.007
Diatomite price, 2021 Diatomite Price Manufactures & supplier https://www.made-in-china.com/products-search/hot-china-products/Diatomite_Price.html, 2021 (accessed 10 January 2021)
Dobor J, Perenyi K, Varga I, Varga M (2015) A new carbonediatomite earth composite adsorbent for removal of heavy metals from aqueous solutions and a novel application idea. Microporous Mesoporous Mater 217:63–70. https://doi.org/10.1016/j.micromeso.2015.06.004
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:385–471
Gebreegziabher TB, Wang S, Nam H (2019) Adsorption of H2S, NH3 and TMA from indoor air using porous corncob activated carbon: isotherm and kinetics study. J Environ Chem Eng 7:103234. https://doi.org/10.1016/j.jece.2019.103234
Hadjar H, Hamdi B, Ania CO (2011) Adsorption of p-cresol on novel diatomite/carbon composites. J Hazard Mater 188:304–310. https://doi.org/10.1016/j.jhazmat.2011.01.108
Halim AA, Aziz HA, Johari MAM, Ariffin KS (2010) Comparison study of ammonia and COD adsorption on zeolite, activated carbon and composite materials in landfill leachate treatment. Desalination 262:31–35. https://doi.org/10.1016/j.desal.2010.05.036
Hu R, Liu G, Zhang H, Xue H, Wang X (2018) Levels, characteristics and health risk assessment of VOCs in different functional zones of Hefei. Ecotoxicol Environ Saf 160:301–307. https://doi.org/10.1016/j.ecoenv.2018.05.056
Kayranli B, Gok O, Yilmaz T, Gok G, Celebi H, Seckin IY, Kalat D (2021) Zinc removal mechanisms with recycled lignocellulose: from fruit residual to biosorbent then soil conditioner. Water Air Soil Pollut 232(8):311. https://doi.org/10.1007/s11270-021-05260-7
Kim S-M, Jeong H-R, Wang S, Nam H and Nam K-W, (2017) “Odor removal by diatomite. In: Proceeding of The Korean Society for Power System Engineering 2017 Spring Annual Meeting, 130–131
Kutluay S, Baytar O, Sahin O (2019) Equilibrium, kinetic and thermodynamic studies for dynamic adsorption of benzene in gas phase onto activated carbon produced from elaeagnus angustifolia seeds. J Environ Chem Eng 7:102947. https://doi.org/10.1016/j.jece.2019.102947
Lee S-W, Daud WMAW, Lee M-G (2010) Adsorption characteristics of methyl mercaptan, dimethyl disulfide, and trimethylamine on coconut-based activated carbons modified with acid and base. J Ind Eng Chem 16:973–977. https://doi.org/10.1016/j.jiec.2010.04.002
Li Y, Yin S, Yu S, Yuan M, Dong Z, Zhang D, Yang L, Zhang R (2020) Characteristics, source apportionment and health risks of ambient VOCs during high ozone period at an urban site in central plain. China Chemosph 250:126283. https://doi.org/10.1016/j.chemosphere.2020.126283
Liang X, Sun X, Xu J, Ye D (2020) Improved emissions inventory and VOCs speciation for industrial OFP estimation in China. Sci Total Environ 745:140838. https://doi.org/10.1016/j.scitotenv.2020.140838
Liu P, He H, Wei G, Liu D, Liang X, Chen T, Zhu J, Zhu R (2017) An efficient catalyst of manganese supported on diatomite for toluene oxidation: manganese species, catalytic performance, and structureactivity relationship. Microporous Mesoporous Mater 239:101–110. https://doi.org/10.1016/j.micromeso.2016.09.053
Liu Y, Song M, Liu X, Zhang Y, Hui L, Kong L, Zhang Y, Zhang C, Qu Y, An J, Ma D, Tan Q, Feng M (2020) Characterization and sources of volatile organic compounds (VOCs) and their related changes during ozone pollution days in 2016 in Beijing. China Environ Pollut 257:113599. https://doi.org/10.1016/j.envpol.2019.113599
Mochizuki T, Kubota M, Matsuda H, Camacho LFD (2016) Adsorption behaviors of ammonia and hydrogen sulfide on activated carbon prepared from petroleum coke by KOH chemical activation. Fuel Process Technol 144:164–169. https://doi.org/10.1016/j.fuproc.2015.12.012
Nam H, Wang S, Jeong H-R (2018) TMA and H2S gas removals using metal loaded on rice husk activated carbon for indoor air purification. Fuel 213:186–194. https://doi.org/10.1016/j.fuel.2017.10.089
Reichenberg D (1953) Properties of ion exchange resins in relation to their structure. III. Kinetics of exchange. J Am Chem Soc 75:589–597. https://doi.org/10.1021/ja01099a022
Saha D, Deng S (2010a) Adsorption equilibrium and kinetics of CO2, CH4, N2O, and NH3 on ordered mesoporous carbon. J Colloid Interface Sci 345:402–409. https://doi.org/10.1016/j.jcis.2010.01.076
Saha D, Deng S (2010b) Characteristics of ammonia adsorption on activated alumina. J Chem Eng 55:5587–5593. https://doi.org/10.1021/je100405k
Santawee N, Treesubauntorn C, Thiravetyan P (2019) Lignin and holocellulose from coir pith involved in trimethylamine (fishy odor) adsorption. J Environ Sci (china) 79:43–53. https://doi.org/10.1016/j.jes.2018.10.009
Sato T, Abe S, Ito S, Abe T (2019) Silk fibroin fiber for selective palladium adsorption: Kinetic, isothermal and thermodynamic properties. J Environ Chem Eng 7:102958. https://doi.org/10.1016/j.jece.2019.102958
Shuang Y, Cui Y, Shi R, Yang P, Wang J, Wang Y (2019) Regenerated WO272 nanowires with superb fast and selective adsorption for cationic dye: Kinetics, isotherm, thermodynamics, mechanism. J Hazard Mater 379:120834. https://doi.org/10.1016/j.jhazmat.2019.120834
Sun X, He J, Yang X (2017) Human breath as a source of VOCs in the built environment, Part I: a method for sampling and detection species. Build Environ 125:565–573. https://doi.org/10.1016/j.buildenv.2017.06.038
Tang L, Li L, Chen R, Wang C, Ma W, Ma X (2016) Adsorption of acetone and isopropanol on organic acid modified activated carbons. J Environ Chem Eng 4:2045–2051. https://doi.org/10.1016/j.jece.2016.03.031
Tzvetkova PG, Nickolov RN, Tzvetkova CT, Bozhkov OD, Voykova DK (2016) Diatomite/carbon adsorbent for phenol removal. J Chem Technol Metall 51(2):202–209
Wang S, Nam H, Nam H (2019a) Utilization of cocoa activated carbon for TMA and H2S gas removals in a confined space and its techno-economic analysis and life cycle analysis. Environ Prog Sustain Energy 38:e13241. https://doi.org/10.1002/ep.13241
Wang S, Lee Y-N, Nam H, Nam H, Kim H-K (2019b) Chemical activation of porous diatomite ceramic filter for the adsorption of TMA, H2S, CH3COOH and NH3: isotherm and kinetic studies. J Environ Chem Eng 7:103481. https://doi.org/10.1016/j.jece.2019.103481
Wang S, Nam H, Gebreegziabher TB, Nam H (2020a) Adsorption of acetic acid and hydrogen sulfide using NaOH impregnated activated carbon for indoor air purification. Engineering Reports 2:e12083. https://doi.org/10.1002/eng2.12083
Wang S, Nam H, Nam H (2020b) Preparation of activated carbon from peanut shell with KOH activation and its application for H2S adsorption in confined space. J Environ Chem Eng 8:103683. https://doi.org/10.1016/j.jece.2020.103683
Wang S, Nam H, Kim H, Nam K (2016) Cocoa activated carbon to remove VOCs (TMA, H2S). In: 13th International Symposium on the Genetics of Industrial Microorganisms (GIM2016 WUHAN), 277
Weber WJ, Morris JC, (1962) Proc. Int. Conf. Water pollution symposium, Pergamon, Oxford, 2: 231–266
Xie F, Wu F, Liu G, Mu Y, Feng C, Wang H, Giesy JP (2014) Removal of phosphate from eutrophic lakes through adsorption by in situ formation of magnesium hydroxide from diatomite. Environ Sci Technol 48:582–590. https://doi.org/10.1021/es4037379
Zhou K, Ma W, Zeng Z, Ma X, Xu X, Guo Y, Li H, Li L (2019) Experimental and DFT study on the adsorption of VOCs on activated carbon/metal oxides composites. Chem Eng J 372:1122–1133. https://doi.org/10.1016/j.cej.2019.04.218
Zou W, Gao B, Ok YS, Dong L (2019) Integrated adsorption and photocatalytic degradation of volatile organic compounds (VOCs) using carbon-based nanocomposites: A critical review. Chemosphere 218:845–859. https://doi.org/10.1016/j.chemosphere.2018.11.175
Acknowledgements
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
All authors consented to publish this research article.
Additional information
Editorial responsibility: Maryam Shabani.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, S., Nam, H., Nam, H. et al. Hierarchically porous diatomite-activated carbon composite for TMA and NH3 removal in confined spaces. Int. J. Environ. Sci. Technol. 19, 11217–11232 (2022). https://doi.org/10.1007/s13762-022-03922-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-03922-5