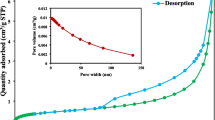
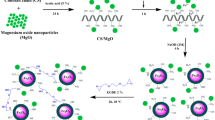
Abstract
Pectin/chitosan magnetic sponge was fabricated using inotropic gelation method and evaluated for the removal of methylene blue dye from aqueous samples. The influence of different factors including pH, methylene blue initial concentration and amount of adsorbent on MB adsorption was assessed using Box–Behnken design. Quadratic model was selected as fitting mathematical model to the studied response. The pH and methylene blue initial concentration positively affected the capacity of adsorption. Alternatively, adsorbent amount showed a negative effect on the adsorption process. For better comprehension for the mechanisms of adsorption process, adsorption isotherms were calculated by fitting Freundlich, Sips and Langmuir models, on the experimental data. It was indicated that Sips isotherm was the most adequate model to explain methylene blue adsorption on pectin/chitosan magnetic sponge with correlation coefficient of 0.9937 and the lowest Chi-square among the tested isotherms. The maximum capacity of adsorption, calculated using Sips equation, had a high value of 174 mg methylene blue per g adsorbent. In addition, the pseudo-second-order equation well described the kinetics of adsorption. Finally, pectin/chitosan magnetic sponge introduced very good potential in the management of methylene blue wastewater.






Similar content being viewed by others
References
Ai L, Zhang C, Chen Z (2011) Removal of methylene blue from aqueous solution by a solvothermal-synthesized graphene/magnetite composite. J Hazard Mater 192(3):1515–1524. https://doi.org/10.1016/j.jhazmat.2011.06.068
APHA (2005) Standard methods for the examination of water and wastewater. 21st ed, American Public Health Association (APHA), American Water Works Association (AWWA) and Water Environment Federation (WEF)
Attallah OA, Al-Ghobashy MA, Nebsen M, Salem MY (2016) Removal of cationic and anionic dyes from aqueous solution with magnetite/pectin and magnetite/silica/pectin hybrid nanocomposites: kinetic, isotherm and mechanism analysis. RSC Adv 6(14):1. https://doi.org/10.1039/c5ra23452b
Attallah OA, Al-Ghobashy MA, Nebsen M, Salem MY (2017) Adsorptive removal of fluoroquinolones from water by pectin-functionalized magnetic nanoparticles: process optimization using a spectrofluorimetric assay. ACS Sustain Chem Eng 5(1):133–145. https://doi.org/10.1021/acssuschemeng.6b01003
Boudouaia N, Bengharez Z, Jellali S (2019) Preparation and characterization of chitosan extracted from shrimp shells waste and chitosan film: application for Eriochrome black T Removal from aqueous solutions. Appl Water Sci 9(4):91. https://doi.org/10.1007/s13201-019-0967-z
Bulut E, Özacar M, Şengil İA (2008) Equilibrium and kinetic data and process design for adsorption of Congo Red onto bentonite. J Hazard Mater 154(1):613–622
Calimli MH, Nas MS, Burhan H, Mustafov SD, Demirbas Ö, Sen F (2020) Preparation, characterization and adsorption kinetics of methylene blue dye in reduced-graphene oxide supported nanoadsorbents. J Mol Liq 309:113171. https://doi.org/10.1016/j.molliq.2020.113171
Chang J, Ma J, Ma Q, Zhang D, Qiao N, Mengxiao H, Ma H (2016) Adsorption of methylene blue onto Fe3O4/activated montmorillonite nanocomposite. Appl Clay Sci 119(January):132–140. https://doi.org/10.1016/j.clay.2015.06.038
Cho DW, Lee J, Ok YS, Kwon EE, Song H (2016) Fabrication of a novel magnetic carbon nanocomposite adsorbent via pyrolysis of sugar. Chemosphere 163(November):305–312. https://doi.org/10.1016/j.chemosphere.2016.08.025
Cooper P (2008) Removing colour from dyehouse waste waters: a critical review of technology. J Soc Dyers Colour 109(3):97–100. https://doi.org/10.1111/j.1478-4408.1993.tb01536.x
Crini G, Badot P-M (2008) Application of chitosan, a natural aminopolysaccharide, for dye removal from aqueous solutions by adsorption processes using batch studies: a review of recent literature. Prog Polym Sci 33(4):399–447. https://doi.org/10.1016/J.PROGPOLYMSCI.2007.11.001
Dai J, Sun J, Xie A, He J, Li C, Yan Y (2016) Designed preparation of 3D hierarchically porous carbon material via solvothermal route and in situ activation for ultrahigh-efficiency dye removal: adsorption isotherm, kinetics and thermodynamics characteristics. RSC Adv 6(5):3446–3457. https://doi.org/10.1039/c5ra24774h
Dalaran M, Emik S, Güçlü G, İyim TB, Özgümüş S (2011) Study on a novel polyampholyte nanocomposite superabsorbent hydrogels: synthesis, characterization and investigation of removal of Indigo carmine from aqueous solution. Desalination 279(1–3):170–182. https://doi.org/10.1016/J.DESAL.2011.06.004
Dragan ES, Loghin DFA (2013) Enhanced sorption of methylene blue from aqueous solutions by semi-IPN composite cryogels with anionically modified potato starch entrapped in PAAm matrix. Chem Eng J 234(December):211–222. https://doi.org/10.1016/j.cej.2013.08.081
Elmoubarki R, Taoufik M, Ahmed Moufti H, Tounsadi FZ, Mahjoubi Y Bouabi, Qourzal S, Abdennouri M, Barka N (2017) Box–Behnken experimental design for the optimization of methylene blue adsorption onto Aleppo Pine Cones. J Mater Environ Sci 8(6):2184–2191
Freundlich HMF (1906) Uber die adsorption in losungen (adsorption in solution). Z Phys Chem 57:385–490
Fu J, Chen Z, Wang M, Liu S, Zhang J, Zhang J, Han R, Qun X (2015) Adsorption of methylene blue by a high-efficiency adsorbent (polydopamine microspheres): kinetics, isotherm, thermodynamics and mechanism analysis. Chem Eng J 259(January):53–61. https://doi.org/10.1016/j.cej.2014.07.101
Gadgil A (1998) Drinking water in developing countries. Annu Rev Energy Environ 23(1):253–286. https://doi.org/10.1146/annurev.energy.23.1.253
Hastuti B, Hadi S, Afifah SN, Mulyani B (2019) Synthesis and characterization of chitosan–pectin as adsorbent of dyes. IOP Conf Ser Mater Sci Eng 578(1):012005. https://doi.org/10.1088/1757-899x/578/1/012005
Hunter PR, MacDonald AM, Carter RC (2010) Water supply and health. PLoS Med. https://doi.org/10.1371/journal.pmed.1000361
Jeon YS, Lei J, Kim JH (2008) Dye adsorption characteristics of alginate/polyaspartate hydrogels. J Ind Eng Chem 14(6):726–731. https://doi.org/10.1016/j.jiec.2008.07.007
Jonassen H, Treves A, Kjøniksen AL, Smistad G, Hiorth M (2013) Preparation of ionically cross-linked pectin nanoparticles in the presence of chlorides of divalent and monovalent cations. Biomacromolecules 14(10):3523–3531. https://doi.org/10.1021/bm4008474
Karada E, Kundakci S, Üzüm ÖB, Karada E, Kundakci S, Üzüm ÖB (2011) Investigation of swelling/sorption characteristics of highly swollen AAm/AMPS hydrogels and semi IPNs with PEG as biopotential sorbent. J Encapsul Adsorpt Sci 1(1):7–22. https://doi.org/10.4236/JEAS.2011.11002
Kyzas GZ, Jie F, Matis KA (2013) The change from past to future for adsorbent materials in treatment of dyeing wastewaters. Materials 6(11):5131–5158. https://doi.org/10.3390/ma6115131
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40(9):1361–1403. https://doi.org/10.1021/ja02242a004
Lian S, Wang E, Kang Z, Bai Y, Gao L, Jiang M, Changwen H, Lin X (2004) Synthesis of magnetite nanorods and porous hematite nanorods. Solid State Commun 129(8):485–490. https://doi.org/10.1016/j.ssc.2003.11.043
Ma Z, Guan Y, Liu H (2005) Synthesis and characterization of micron-sized monodisperse superparamagnetic polymer particles with amino groups. J Polym Sci Part A Polym Chem 43(15):3433–3439. https://doi.org/10.1002/pola.20803
Madrakian T, Afkhami A, Ahmadi M (2012) Adsorption and kinetic studies of seven different organic dyes onto magnetite nanoparticles loaded tea waste and removal of them from wastewater samples. Spectrochim Acta Part A Mol Biomol Spectrosc 99:102–109
Mikhaylova M, Kim DK, Bobrysheva N, Osmolowsky M, Semenov V, Tsakalakos T, Muhammed M (2004) Superparamagnetism of magnetite nanoparticles: dependence on surface modification. Langmuir 20(6):2472–2477. https://doi.org/10.1021/la035648e
Nas MS, Calimli MH, Burhan H, Yılmaz M, Mustafov SD, Sen F (2019) Synthesis, characterization, kinetics and adsorption properties of Pt-Co@GO nano-adsorbent for methylene blue removal in the aquatic mediums using ultrasonic process systems. J Mol Liq 296:112100. https://doi.org/10.1016/j.molliq.2019.112100
Nsom MV, Etape EP, Tendo JF, Namond BV, Chongwain PT, Yufanyi MD, William N (2019) A green and facile approach for synthesis of starch-pectin magnetite nanoparticles and application by removal of methylene blue from textile effluent. J Nanomater 2019(August):1–12. https://doi.org/10.1155/2019/4576135
Oki T, Kanae S (2006) Global hydrological cycles and world water resources. Science 313:1068–1072. https://doi.org/10.1126/science.1128845
Piaskowski K, Świderska-Dąbrowska R, Zarzycki PK (2018) Dye removal from water and wastewater using various physical, chemical, and biological processes. J AOAC Int 101(5):1371–1384. https://doi.org/10.5740/jaoacint.18-0051
Pimentel D, Houser J, Preiss E, White O, Fang H, Mesnick L et al (1997) Water resources: agriculture, the environment, and society. Bioscience 47(2):97–106. https://doi.org/10.2307/1313020
Rocher V, Bee A, Siaugue JM, Cabuil V (2010) Dye removal from aqueous solution by magnetic alginate beads crosslinked with epichlorohydrin. J Hazard Mater 178(1–3):434–439. https://doi.org/10.1016/j.jhazmat.2010.01.100
Saleh SM (2005) HPLC determination of four textile dyes and studying their degradation using spectrophotometric technique. MSc desertation, An-Najah National University, Nablus, Palestine
Sips R (1948) On the structure of a catalyst surface. J Chem Phys 16(5):490. https://doi.org/10.1063/1.1746922
Sivashankar R, Sathya AB, Vasantharaj K, Sivasubramanian V (2014) Magnetic composite an environmental super adsorbent for dye sequestration: a review. Environ Nanotechnol Monit Manag 1–2(November):36–49. https://doi.org/10.1016/j.enmm.2014.06.001
Thomas PC, Cipriano BH, Raghavan SR (2011) Nanoparticle-crosslinked hydrogels as a class of efficient materials for separation and ion exchange. Soft Matter 7(18):8192. https://doi.org/10.1039/c1sm05369h
Yin W, Hao S, Cao H (2017) Solvothermal synthesis of magnetic CoFe2O4/RGO nanocomposites for highly efficient dye removal in wastewater. RSC Adv 7(7):4062–4069. https://doi.org/10.1039/C6RA26948F
Zhou L, Huang J, He B, Zhang F, Li H (2014) Peach gum for efficient removal of methylene blue and methyl violet dyes from aqueous solution. Carbohyd Polym 101(January):574–581. https://doi.org/10.1016/J.CARBPOL.2013.09.093
Acknowledgement
The authors acknowledge the financial support received from the American University in Cairo (AUC) through Faculty Support Research Grants.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Editorial responsibility: Fatih Şen.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Attallah, O.A., Mamdouh, W. Development and optimization of pectin/chitosan magnetic sponge for efficient cationic dyes removal using Box–Behnken design. Int. J. Environ. Sci. Technol. 18, 131–140 (2021). https://doi.org/10.1007/s13762-020-02828-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-020-02828-4