Abstract
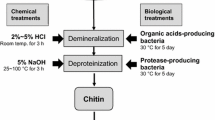
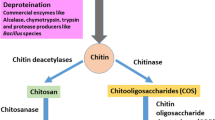
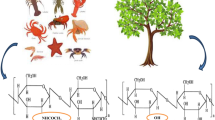
Unscientific dumping of shellfishery waste is a major environmental concern worldwide and a serious threat to the coastal area. The shell wastes constitute of many commercially valuable products, such as, chitin, calcium carbonate, proteins, and carotenoids. Processing of shell wastes is a source of wealth. Chitin is one of the constituents of the shell wastes and also the most abundant biopolymer next to the cellulose. Calcium carbonate and proteins are the other valuable constituents of the shell wastes and can serve as a better animal feed supplement. Extraction of chitin and other valuable products from a complex mixture is cumbersome and requires consecutive pretreatment processes. Both chemical and fermentation routes of pretreatment strategies have been adopted to extract the chitin from the shell wastes, which are critically reviewed with their merits and demerits. The application of process intensification techniques, such as ultrasonication and microwave radiation, is also emphasized along with the recent advancement in the field of pretreatment processes. Pleiotropic industrial applications of chitin and its analogues are also discussed.


Reprinted with permission from (Devi and Dhamodharan 2018). Copyright@American Chemical Society


Reprinted with permission from Wang et al. (2018). Copyright@American Chemical Society

Reprinted with permission from Wang et al. (2018). Copyright@American Chemical Society

Reprinted with permission from Sathiyabama and Manikandan (2018). Copyright@American Chemical Society

Reprinted with permission from Sharma and Singh (2017). Copyright@American Chemical Society

Reprinted with permission from—Nascimento et al. (2014). Copyright@American Chemical Society
Similar content being viewed by others
References
Aam BB, Heggset EB, Norberg AL, Sorlie M, Varum KM, Eijsink VG (2010) Production of chitooligosaccharides and their potential applications in medicine. Mar Drugs 8:1482–1517
Adnadjevic B, Jovanovic J (2011) The effects of microwave heating on the isothermal kinetics of chemicals reactions and physicochemical processes. In: Advances in induction and microwave heating of mineral and organic materials. InTech
Ahn CW, Nam HS, Lee HJ, Shin YC (1994) Decolorization of shrimp chitin using sodium hypochlorite. Korean J Food Sci Technol 26:787–790
Alves HJ et al (2017) Effect of shrimp shells milling on the molar mass of chitosan. Polímeros 27:41–47
Aranaz I et al (2009) Functional characterization of chitin and chitosan. Curr Chem Biol 3:203–230
Arbia W, Adour L, Amrane A, Lounici H (2013a) Optimization of medium composition for enhanced chitin extraction from Parapenaeus longirostris by Lactobacillus helveticus using response surface methodology. Food Hydrocolloids 31:392–403
Arbia W, Arbia L, Adour L, Amrane A (2013b) Chitin extraction from crustacean shells using biological methods–a review. Food Technol Biotechnol 51:12–25
Arvanitoyannis I (2008) The use of chitin and chitosan for food packaging applications. Environmentally compatible food packaging, vol 137
Aye KN, Stevens WF (2004) Improved chitin production by pretreatment of shrimp shells. J Chem Technol Biotechnol 79:421–425
Aytekin O, Elibol M (2010) Cocultivation of Lactococcus lactis and Teredinobacter turnirae for biological chitin extraction from prawn waste. Bioprocess Biosyst Eng 33:393–399
Badawy ME, Rabea EI (2011) A biopolymer chitosan and its derivatives as promising antimicrobial agents against plant pathogens and their applications in crop protection. Int J Carbohydr Chem 2011:1–29
Bansal V, Sharma PK, Sharma N, Pal OP, Malviya R (2011) Applications of chitosan and chitosan derivatives in drug delivery. Adv Biol Res 5:28–37
Barikani M, Oliaei E, Seddiqi H, Honarkar H (2014) Preparation and application of chitin and its derivatives: a review. Iran Polym J 23:307–326
Binod P, Pusztahelyi T, Nagy V, Sandhya C, Szakács G, Pócsi I, Pandey A (2005) Production and purification of extracellular chitinases from Penicillium aculeatum NRRL 2129 under solid-state fermentation. Enzyme Microb Technol 36:880–887
Bourbon AI, Pinheiro AC, Cerqueira MA, Rocha CM, Avides MC, Quintas MA, Vicente AA (2011) Physico-chemical characterization of chitosan-based edible films incorporating bioactive compounds of different molecular weight. J Food Eng 106:111–118
Bustos RO, Healy MG Microbial deproteinisation of waste prawn shell. In: Anon (ed) Proceedings of the 2nd international symposium on environmental biotechnology, 1994. Institution of Chemical Engineers Symposium Series. Publ by Inst of Chemical Engineers, Rugby, United Kingdom, pp 13–15
Charoenvuttitham P, Shi J, Mittal GS (2006) Chitin extraction from black tiger shrimp (Penaeus monodon) waste using organic acids. Sep Sci Technol 41:1135–1153
Chen J, Wang Q, Hua Z, Du G (2007) Research and application of biotechnology in textile industries in China. Enzyme Microb Technol 40:1651–1655
Choi C-R et al (2012) Chitooligosaccharides decreases plasma lipid levels in healthy men. Int J Food Sci Nutr 63:103–106
Coral G, Huberman A, de la Lanza G, Monroy-Ruiz J (1998) Muscle pigmentation of rainbow trout (Oncorhynchus mykiss) fed on oil-extracted pigment from langostilla (Pleuroncodes planipes) compared with two commercial sources of astaxanthin. J Aquat Food Prod Technol 7:31–45
Correia R, Magalhaes M, Marques P, Senos A (1996) Wet synthesis and characterization of modified hydroxyapatite powders. J Mater Sci Mater Med 7:501–505
Dash M, Chiellini F, Ottenbrite R, Chiellini E (2011) Chitosan—A versatile semi-synthetic polymer in biomedical applications. Prog Polym Sci 36:981–1014
Devi R, Dhamodharan R (2018) Pretreatment in hot glycerol for facile and green separation of chitin from prawn shell waste. Acs Sustain Chem Eng 6:846–853. https://doi.org/10.1021/acssuschemeng.7b03195
Díaz-Rojas EI, Argüelles-Monal WM, Higuera-Ciapara I, Hernández J, Lizardi-Mendoza J, Goycoolea FM (2006) Determination of chitin and protein contents during the isolation of chitin from shrimp waste. Macromol Biosci 6:340–347
Du J, Tan E, Kim HJ, Zhang A, Bhattacharya R, Yarema KJ (2014) Comparative evaluation of chitosan, cellulose acetate, and polyethersulfone nanofiber scaffolds for neural differentiation. Carbohydr Polym 99:483–490
Dutta PK, Dutta J, Tripathi V (2004) Chitin and chitosan: chemistry, properties and applications. J Sci Ind Res 63:20–31
Dutta J, Tripathi S, Dutta P (2012) Progress in antimicrobial activities of chitin, chitosan and its oligosaccharides: a systematic study needs for food applications. Revista de Agaroquimica y Tecnologia de Alimentos 18:3–34
El Hadrami A, Adam LR, El Hadrami I, Daayf F (2010) Chitosan in plant protection. Mar Drugs 8:968–987
El Knidri H, El Khalfaouy R, Laajeb A, Addaou A, Lahsini A (2016) Eco-friendly extraction and characterization of chitin and chitosan from the shrimp shell waste via microwave irradiation. Process Saf Environ Prot 104:395–405
FAO (2017) Increased production of farmed shrimp leads to improved international trade. GLOBEFISH—analysis and information on world fish trade http://www.fao.org/in-action/globefish/market-reports/resource-detail/en/c/989543/. Accessed 10 July 2017
Fernandes JC et al (2012) Inhibition of bladder tumor growth by chitooligosaccharides in an experimental carcinogenesis model. Mar Drugs 10:2661–2675
Finkelstein A, Wohlt J, Emanuele S, Tweed S (1993) Composition and nutritive value of ground sea clam shells as calcium supplements for lactating holstein cows1. J Dairy Sci 76:582–589
Flores-Albino B, Arias L, Gómez J, Castillo A, Gimeno M, Shirai K (2012) Chitin and L (+)-lactic acid production from crab (Callinectes bellicosus) wastes by fermentation of Lactobacillus sp. B2 using sugar cane molasses as carbon source. Bioprocess Biosyst Eng 35:1193–1200
Foster A, Webber J (1961) Chitin. Advances in carbohydrate chemistry 15:371–393
Frydenberg RP, Hammershøj M, Andersen U, Wiking L (2013) Ultrasonication affects crystallization mechanisms and kinetics of anhydrous milk fat. Cryst Growth Des 13:5375–5382
Fu Y, Xiao C (2017) A facile physical approach to make chitosan soluble in acid-free water. Int J Biol Macromol 103:575–580
Funahashi R, Ono Y, Qi Z-D, Saito T, Isogai A (2017) Molar masses and molar mass distributions of chitin and acid-hydrolyzed chitin. Biomacromolecules 18:4357–4363
Gao Y, Cranston R (2008) Recent advances in antimicrobial treatments of textiles. Text Res J 78:60–72
Gautier S, Xhauflaire-Uhoda E, Gonry P, Piérard G (2008) Chitin–glucan, a natural cell scaffold for skin moisturization and rejuvenation. Int J Cosmet Sci 30:459–469
Gerry R (1980) Ground dried whole mussels as a calcium supplement for chicken rations. Poult Sci 59:2365–2368
Ghorbel-Bellaaj O, Jellouli K, Younes I, Manni L, Salem MO, Nasri M (2011) A solvent-stable metalloprotease produced by Pseudomonas aeruginosa A2 grown on shrimp shell waste and its application in chitin extraction. Appl Biochem Biotechnol 164:410–425
Ghorbel-Bellaaj O, Younes I, Maâlej H, Hajji S, Nasri M (2012) Chitin extraction from shrimp shell waste using Bacillus bacteria. Int J Biol Macromol 51:1196–1201
Ghorbel-Bellaaj O, Hajji S, Younes I, Chaabouni M, Nasri M, Jellouli K (2013) Optimization of chitin extraction from shrimp waste with Bacillus pumilus A1 using response surface methodology. Int J Biol Macromol 61:243–250
Gortari MC, Hours RA (2013) Biotechnological processes for chitin recovery out of crustacean waste: a mini-review. Electron J Biotechnol 16:1–14
Guinotte F, Nys Y, De Monredon F (1991) The effects of particle size and origin of calcium carbonate on performance and ossification characteristics in broiler chicks. Poult Sci 70:1908–1920
Gupta D (2007) Antimicrobial treatments for textiles. Indian J Fibre Text Res 32:254–263
Haddar A, Hmidet N, Ghorbel-Bellaaj O, Fakhfakh-Zouari N, Sellami-Kamoun A, Nasri M (2011) Alkaline proteases produced by Bacillus licheniformis RP1 grown on shrimp wastes: application in chitin extraction, chicken feather-degradation and as a dehairing agent. Biotechnol Bioprocess Eng 16:669
Hajji S, Ghorbel-Bellaaj O, Younes I, Jellouli K, Nasri M (2015) Chitin extraction from crab shells by Bacillus bacteria. Biological activities of fermented crab supernatants. Int J Biol Macromol 79:167–173
Han F-S, Yang S-J, Lin M-B, Chen Y-Q, Yang P, Xu J-M (2016) Chitooligosaccharides promote radiosensitivity in colon cancer line SW480. World J Gastroenterol 22:5193
Harkin C, Brück W, Lynch C (2015) Isolation & identification of bacteria for the treatment of brown crab (Cancer pagurus) waste to produce chitinous material. J Appl Microbiol 118:954–965
Heu M-S, Kim J-S, Shahidi F (2003) Components and nutritional quality of shrimp processing by-products. Food Chem 82:235–242
Higuera-Ciapara I, Felix-Valenzuela L, Goycoolea F (2006) Astaxanthin: a review of its chemistry and applications. Crit Rev Food Sci Nutr 46:185–196
Horst MN, Walker AN, Klar E (1993) The pathway of crustacean chitin synthesis. The crustacean integument morphology and biochemistry, pp 113–149
Hou Y, Shavandi A, Carne A, Bekhit AA, Ng TB, Cheung RCF, Bekhit AE-DA (2016) Marine shells: potential opportunities for extraction of functional and health-promoting materials. Crit Rev Environ Sci Technol 46:1047–1116
Huang R, Mendis E, Rajapakse N, Kim S-K (2006) Strong electronic charge as an important factor for anticancer activity of chitooligosaccharides (COS). Life Sci 78:2399–2408
Ingeç T, Tekin T (2004) Effect of ultrasound on the production reaction kinetics of sodium thiosulfate. Chem Eng Technol Ind Chem Plant Equip Process Eng Biotechnol 27:150–153
Ivancic A, Macaev F, Aksakal F, Boldescu V, Pogrebnoi S, Duca G (2016) Preparation of alginate–chitosan–cyclodextrin micro-and nanoparticles loaded with anti-tuberculosis compounds. Beilstein J Nanotechnol 7:1208
Jeong HW et al (2012) Chitooligosaccharide induces mitochondrial biogenesis and increases exercise endurance through the activation of Sirt1 and AMPK in rats. PLoS ONE 7:e40073
Jianglian D, Shaoying Z (2013) Application of chitosan based coating in fruit and vegetable preservation: a review. J Food Process Technol 4:1–4
Ju C, Yue W, Yang Z, Zhang Q, Yang X, Liu Z, Zhang F (2010) Antidiabetic effect and mechanism of chitooligosaccharides. Biol Pharm Bull 33:1511–1516
Jung W, Kuk J, Kim K, Park R (2005) Demineralization of red crab shell waste by lactic acid fermentation. Appl Microbiol Biotechnol 67:851–854
Kafetzopoulos D, Martinou A, Bouriotis V (1993) Bioconversion of chitin to chitosan: purification and characterization of chitin deacetylase from Mucor rouxii. Proc Natl Acad Sci 90:2564–2568
Kamjumphol W, Chareonsudjai P, Chareonsudjai S (2018) Antibacterial activity of chitosan against Burkholderia pseudomallei. MicrobiologyOpen 7:1–8
Kang N-H et al (2012) Modulation of lipid metabolism by mixtures of protamine and chitooligosaccharide through pancreatic lipase inhibitory activity in a rat model. Lab Animal Res 28:31–38
Katiyar D, Hemantaranjan A, Singh B, Bhanu AN (2014) A future perspective in crop protection: chitosan and its oligosaccharides. Adv Plants Agric Res 1:1–8
Kaya M, Baran T, Karaarslan M (2015) A new method for fast chitin extraction from shells of crab, crayfish and shrimp. Nat Prod Res 29:1477–1480
Khanafari A, Marandi R, Sanatei S (2008) Recovery of chitin and chitosan from shrimp waste by chemical and microbial methods. J Environ Health Sci Eng 5:1–24
Khorrami M, Najafpour G, Younesi H, Amini G (2011) Growth kinetics and demineralization of shrimp shell using Lactobacillus plantarum PTCC 1058 on various carbon sources. Iran J Energy Environ 2:320–325
Khoushab F, Yamabhai M (2010) Chitin research revisited. Mar Drugs 8:1988–2012
Kim E-K et al (2012) Chitooligosaccharides induce apoptosis in human myeloid leukemia HL-60 cells. Bioorg Med Chem Lett 22:6136–6138
Kjartansson GT, Zivanovic S, Kristbergsson K, Weiss J (2006a) Sonication-assisted extraction of chitin from North Atlantic shrimps (Pandalus borealis). J Agric Food Chem 54:5894–5902
Kjartansson GT, Zivanovic S, Kristbergsson K, Weiss J (2006b) Sonication-assisted extraction of chitin from shells of fresh water prawns (Macrobrachium rosenbergii). J Agric Food Chem 54:3317–3323
Kumar RS (2016) Textiles for industrial applications. CRC Press, Boca Raton
Leal ALG, de Castro PF, de Lima JPV, de Souza Correia E, de Souza Bezerra R (2010) Use of shrimp protein hydrolysate in Nile tilapia (Oreochromis niloticus L.) feeds. Aquacult Int 18:635–646
Lee M, Nah J-W, Kwon Y, Koh JJ, Ko KS, Kim SW (2001) Water-soluble and low molecular weight chitosan-based plasmid DNA delivery. Pharm Res 18:427–431
Liu M et al (2016) Efficient mucus permeation and tight junction opening by dissociable “mucus-inert” agent coated trimethyl chitosan nanoparticles for oral insulin delivery. J Controll Release 222:67–77
Lopata AL, Kleine-Tebbe J, Kamath SD (2017) Allergens and molecular diagnostics of shellfish allergy. In: Molecular allergy diagnostics. Springer, pp 399–414
Luo Z, Dong X, Ke Q, Duan Q, Shen L (2014) Downregulation of CD147 by chitooligosaccharide inhibits MMP-2 expression and suppresses the metastatic potential of human gastric cancer. Oncol Lett 8:361–366
Mahdavi B, Rahimi A (2013) Seed priming with chitosan improves the germination and growth performance of ajowan (Carum copticum) under salt stress. EurAsian J BioSci 7:69–76
Mahmoud N, Ghaly A (2015) Effect of particle size on the kinetic parameters of the deproteinization process of galactose supplemented shrimp shells by Aspergillus niger. J Adv Biotechnol Bioeng 3:14–31
Mahmoud NS, Ghaly A unconventional demineralization of crustacean waste for the production of chitin. In: 2006 ASAE annual meeting, 2006. American Society of Agricultural and Biological Engineers, p 1
Mao X, Guo N, Sun J, Xue C (2017) Comprehensive utilization of shrimp waste based on biotechnological methods: a review. J Clean Prod 143:814–823
Mayne ST (1996) Beta-carotene, carotenoids, and disease prevention in humans. FASEB J 10:690–701
Mezzomo N, Ferreira SRS (2016) Carotenoids functionality, sources, and processing by supercritical technology: a review. J Chem 2016:1–16
Mizani M, Aminlari M, Khodabandeh M (2005) An effective method for producing a nutritive protein extract powder from shrimp-head waste. Food Sci Technol Int 11:49–54
Moattari M, Moattari F, Kaka G, Kouchesfehani HM, Sadraie HMN (2018) Application of chitosan in textiles. Ann Mater Sci Eng 3:1032
Morganti P (2009) Chitin-nanofibrils in skin treatment. J Appl Cosmetol 27:251–270
Morganti P, Fabrizi G, Palombo P, Palombo M, Ruocco E, Cardillo A, Morganti G (2008) Chitin-nanofibrils: a new active cosmetic carrier. J Appl Cosmetol 26:113–128
Morganti P, Morganti G, Morganti A (2011) Transforming nanostructured chitin from crustacean waste into beneficial health products: a must for our society. Nanotechnol Sci Appl 4:123
Mourya V, Inamdar NN, Tiwari A (2010) Carboxymethyl chitosan and its applications. Adv Mater Lett 1:11–33
Muir FV, Harris PC, Gerry RW (1976) The comparative value of five calcium sources for laying hens. Poult Sci 55(3):1046–1051
Muzzarelli R, Muzzarelli C (2005) Chitosan chemistry: relevance to the biomedical sciences. In: Polysaccharides I. Springer, pp 151–209
Nascimento AV, Singh A, Bousbaa H, Ferreira D, Sarmento B, Amiji MM (2014) Mad2 checkpoint gene silencing using epidermal growth factor receptor-targeted chitosan nanoparticles in non-small cell lung cancer model. Mol Pharm 11:3515–3527
Neves AC, Zanette C, Grade ST, Schaffer JV, Alves HJ, Arantes MK (2017) Optimization of lactic fermentation for extraction of chitin from freshwater shrimp waste. Acta Scientiarum Technol 39:125–133
Ngo D-N, Lee S-H, Kim M-M, Kim S-K (2009) Production of chitin oligosaccharides with different molecular weights and their antioxidant effect in RAW 264.7 cells. J Funct Foods 1:188–198
Nisha S, Seenivasan A, Vasanth D Chitin and its derivatives: structure, production, and their applications. In: International conference on signal processing, communication, power and embedded system (SCOPES), Paralakhemundi, Odisha, India, 2016. vol 3. IEEE, pp 127–130
No HK, Hur EY (1998) Control of foam formation by antifoam during demineralization of crustacean shell in preparation of chitin. J Agric Food Chem 46:3844–3846
Oh K-T, Kim Y-J, Jung W-J, Park R-D (2007) Demineralization of crab shell waste by Pseudomonas aeruginosa F722. Process Biochem 42:1069–1074
Oh K-T, Kim Y-J, Van Nguyen N, Jung W-J, Park R-D (2008) Effect of crab shell size on bio-demineralization with lactic acid-producing bacterium, Lactobacillus paracasei subsp. tolerans KCTC-3074. Biotechnol Bioprocess Eng 13:566–570
Oso A, Idowu A, Niameh O (2011) Growth response, nutrient and mineral retention, bone mineralisation and walking ability of broiler chickens fed with dietary inclusion of various unconventional mineral sources. J Animal Physiol Animal Nutr 95:461–467
Pachapur VL, Guemiza K, Rouissi T, Sarma SJ, Brar SK (2016) Novel biological and chemical methods of chitin extraction from crustacean waste using saline water. J Chem Technol Biotechnol 91:2331–2339
Pacheco N, Garnica-Gonzalez M, Gimeno M, Bárzana E, Trombotto S, David L, Shirai K (2011) Structural characterization of chitin and chitosan obtained by biological and chemical methods. Biomacromol 12:3285–3290
Paul T et al (2015) Production of chitin and bioactive materials from Black tiger shrimp (Penaeus monodon) shell waste by the treatment of bacterial protease cocktail 3. Biotech 5:483–493
Peniston QP, Johnson EL (1978) Process for demineralization of crustacea shells. United State Patent
Percot A, Viton C, Domard A (2003) Characterization of shrimp shell deproteinization. Biomacromol 4:1380–1385
Pichyangkura R, Chadchawan S (2015) Biostimulant activity of chitosan in horticulture. Sci Hortic 196:49–65
Pillai C, Paul W, Sharma CP (2009) Chitin and chitosan polymers: chemistry, solubility and fiber formation. Prog Polym Sci 34:641–678
Ploydee E, Chaiyanan S (2014) Production of high viscosity chitosan from biologically purified chitin isolated by microbial fermentation and deproteinization. Int J Polym Sci 2014:1–8
Prameela K, Mohan CM, Smitha P, Hemalatha K (2010) Bioremediation of shrimp biowaste by using natural probiotic for chitin and carotenoid production an alternative method to hazardous chemical method. Int J Appl Sci Biol Chem Technol 1:903–910
Prashanth KH, Tharanathan R (2007) Chitin/chitosan: modifications and their unlimited application potential—an overview. Trends Food Sci Technol 18:117–131
Qin C, Li H, Xiao Q, Liu Y, Zhu J, Du Y (2006) Water-solubility of chitosan and its antimicrobial activity. Carbohydr Polym 63:367–374
Quan H, Zhu F, Han X, Xu Z, Zhao Y, Miao Z (2009) Mechanism of anti-angiogenic activities of chitooligosaccharides may be through inhibiting heparanase activity. Med Hypotheses 73:205–206
Ramalingam P, Ko YT (2016) Improved oral delivery of resveratrol from N-trimethyl chitosan-g-palmitic acid surface-modified solid lipid nanoparticles. Colloids Surf B 139:52–61
Ramírez M, Rodríguez AT, Alfonso L, Peniche C (2010) Chitin and its derivatives as biopolymers with potential agricultural applications. Biotecnol Apl 27:270–276
Rao M, Munoz J, Stevens W (2000) Critical factors in chitin production by fermentation of shrimp biowaste. Appl Microbiol Biotechnol 54:808–813
Rawway M, Beltagy EA, Abdul-Raouf UM, Elshenawy MA, Kelany MS (2018) Optimization of process parameters for chitinase production by a marine Aspergillus flavus MK20
Rinaudo M (2006) Chitin and chitosan: properties and applications. Prog Polym Sci 31:603–632
Sabu T, Visakh P, Mathew A (2013) Advances in natural polymers: composites and nanocomposites, vol 18. Advanced Structured Materials. Springer, Berlin
Sahariah P, Masson M (2017) Antimicrobial chitosan and chitosan derivatives: a review of the structure-activity relationship. Biomacromolecules 18:3846–3868
Sahu BB, Sahu U, Nagesh Kumar Barik AA, Paikaray A, Mohapatra S, Sahu JK (2017) Bio-refinery products from shell fish processing waste: application of Chitin, Chitosan, Chitooligo saccharides and derivatives in organic agriculture. Int J Fish Aquat Res 2:27–31
Salah R et al (2013) Anticancer activity of chemically prepared shrimp low molecular weight chitin evaluation with the human monocyte leukaemia cell line, THP-1. Int J Biol Macromol 52:333–339
Santo Pereira AE, Silva PM, Oliveira JL, Oliveira HC, Fraceto LF (2017) Chitosan nanoparticles as carrier systems for the plant growth hormone gibberellic acid. Colloids Surf B 150:141–152
Santos-Moriano P et al (2018) Enzymatic production of fully deacetylated chitooligosaccharides and their neuroprotective and anti-inflammatory properties. Biocatal Biotransform 36:57–67
Sathiyabama M, Manikandan A (2018) Application of copper–chitosan nanoparticles stimulate growth and induce resistance in Finger Millet (Eleusine coracana Gaertn.) plants against blast disease. J Agric Food Chem 66:1784–1790
Sawai J, Shiga H (2006) Kinetic analysis of the antifungal activity of heated scallop-shell powder against Trichophyton and its possible application to the treatment of dermatophytosis. Biocontrol Sci 11:125–128
Sawai J, Satoh M, Horikawa M, Shiga H, Kojima H (2001a) Heated scallop-shell powder slurry treatment of shredded cabbage. J Food Prot 64:1579–1583
Sawai J, Shiga H, Kojima H (2001b) Kinetic analysis of the bactericidal action of heated scallop-shell powder. Int J Food Microbiol 71:211–218
Sedaghat F, Yousefzadi M, Toiserkani H, Najafipour S (2016) Chitin from Penaeus merguiensis via microbial fermentation processing and antioxidant activity. Int J Biol Macromol 82:279–283
Sethulekshmi C (2014) Chitin and its benefits. Int J Adv Res Biol Sci 1:171–175
Shahidi F (1994) Seafood processing by-products. In: Seafoods: chemistry, processing technology and quality. Springer, pp 320–334
Shahidi F, Botta JR (2012) Seafoods: chemistry, processing technology and quality. Springer, Berlin
Shahidi F, Synowiecki J (1991) Isolation and characterization of nutrients and value-added products from snow crab (Chionoecetes opilio) and shrimp (Pandalus borealis) processing discards. J Agric Food Chem 39:1527–1532
Sharma D, Singh J (2017) Synthesis and characterization of fatty acid grafted chitosan polymer and their nanomicelles for nonviral gene delivery applications. Bioconjug Chem 28:2772–2783
Sinha S, Chand S, Tripathi P (2014) Microbial degradation of chitin waste for production of chitosanase and food related bioactive compounds. Appl Biochem Microbiol 50:125
Sini TK, Santhosh S, Mathew PT (2007) Study on the production of chitin and chitosan from shrimp shell by using Bacillus subtilis fermentation. Carbohydr Res 342:2423–2429
Stoykov YM, Pavlov AI, Krastanov AI (2015) Chitinase biotechnology: production, purification, and application. Eng Life Sci 15:30–38
Suginta W, Khunkaewla P, Schulte A (2013) Electrochemical biosensor applications of polysaccharides chitin and chitosan. Chem Rev 113:5458–5479
Tanha N, Karimzadeh K, Zahmatkesh A (2017) A study on the antimicrobial activities of chitin and chitosan extracted from freshwater prawn shells (Macrobrachium nipponense). Int J Health Stud 3:16–19
Varshosaz J, Hassanzadeh F, Aliabadi HS, Khoraskani FR, Mirian M, Behdadfar B (2016) Targeted delivery of doxorubicin to breast cancer cells by magnetic LHRH chitosan bioconjugated nanoparticles. Int J Biol Macromol 93:1192–1205
Vo T-S, Kim S-K (2010) Potential anti-HIV agents from marine resources: an overview. Mar Drugs 8:2871–2892
Wahyuntari B, Setyahadi S (2011) Process design of microbiological chitin extraction. Microbiol Indones 5:7
Wang S-L, Chio S-H (1998) Deproteinization of shrimp and crab shell with the protease of Pseudomonas aeruginosa K-187. Enzyme Microb Technol 22:629–633
Wang J, Jiang M, Xin Y, Geng N, Li X, Xuan S (2016) Effect of chitooligosaccharide on hepatic triglyceride metabolism and related mechanisms. Chin J Hepatol 24:220–224
Wang H, Qian J, Ding F (2018) Emerging chitosan-based films for food packaging applications. J Agric Food Chem 66:395–413
Watanabe T, Fujimoto R, Sawai J, Kikuchi M, Yahata S, Satoh S (2014) Antibacterial characteristics of heated scallop-shell nano-particles. Biocontrol Sci 19:93–97
Wild LG, Lehrer SB (2005) Fish and shellfish allergy. Current allergy and asthma reports 5:74–79
Wooten J, Singer NS (2003) Method of extracting chitin from the shells of exoskeletal animals. Google Patents
Wysokowski M, Petrenko I, Stelling AL, Stawski D, Jesionowski T, Ehrlich H (2015) Poriferan chitin as a versatile template for extreme biomimetics. Polymers 7:235–265
Xia W, Liu P, Zhang J, Chen J (2011) Biological activities of chitosan and chitooligosaccharides. Food Hydrocolloids 25:170–179
Yan N, Chen X (2015) Don’t waste seafood waste: turning cast-off shells into nitrogen-rich chemicals would benefit economies and the environment. Nature 524:155–158
Younes I, Rinaudo M (2015) Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar Drugs 13:1133–1174
Younes I, Ghorbel-Bellaaj O, Nasri R, Chaabouni M, Rinaudo M, Nasri M (2012) Chitin and chitosan preparation from shrimp shells using optimized enzymatic deproteinization. Process Biochem 47:2032–2039
Younes I, Hajji S, Frachet V, Rinaudo M, Jellouli K, Nasri M (2014) Chitin extraction from shrimp shell using enzymatic treatment. Antitumor, antioxidant and antimicrobial activities of chitosan. Int J Biol Macromol 69:489–498
Zeng D, Mei X, Wu J (2010) Effects of an environmentally friendly seed coating agent on combating head smut of corn caused by Sphacelotheca reiliana and corn growth. J Agric Biotechnol Sustain Dev 2:108–112
Zeng D, Luo X, Tu R (2012) Application of bioactive coatings based on chitosan for soybean seed protection. Int J Carbohydr Chem 2012:1–5. https://doi.org/10.1155/2012/104565
Zhang H, Jin Y, Deng Y, Wang D, Zhao Y (2012) Production of chitin from shrimp shell powders using Serratia marcescens B742 and Lactobacillus plantarum ATCC 8014 successive two-step fermentation. Carbohydr Res 362:13–20
Zhang J, Xl D, Jiang Z-F (2013) Research progress in functions of bioactive chitooligosaccharides in prevention and treatment of Alzheimer’s disease. Food Sci 11:066
Zhang H, Yang S, Fang J, Deng Y, Wang D, Zhao Y (2014) Optimization of the fermentation conditions of Rhizopus japonicus M193 for the production of chitin deacetylase and chitosan. Carbohydr Polym 101:57–67
Zhao Y, Park R-D, Muzzarelli RA (2010) Chitin deacetylases: properties and applications. Mar Drugs 8:24–46
Zhao L et al (2012) Effect of chitooligosaccharides schiff base on chlorophyll content in tobacco leaves infected by TMV. Acta Agric Jiangxi 24:113–116
Ziegler RG (1991) Vegetables, fruits, and carotenoids and the risk of cancer. Am J Clin Nutr 53:251S–259S
Acknowledgements
Nisha Suryawanshi thanks CSIR, New Delhi, for funding the fellowship for her Ph.D. Programme. We also thank National Institute of Technology for funding and support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: M. Abbaspour.
Rights and permissions
About this article
Cite this article
Suryawanshi, N., Jujjavarapu, S.E. & Ayothiraman, S. Marine shell industrial wastes–an abundant source of chitin and its derivatives: constituents, pretreatment, fermentation, and pleiotropic applications-a revisit. Int. J. Environ. Sci. Technol. 16, 3877–3898 (2019). https://doi.org/10.1007/s13762-018-02204-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-018-02204-3