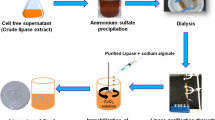
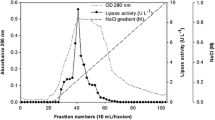
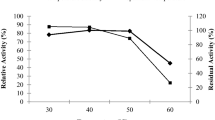
Abstract
The lipA gene, encoding a solvent-tolerant extracellular lipase from Proteus sp. SW1, was displayed on the cell surface of Escherichia coli by fusing it to an antigen 43 anchoring motif. The display of LipA on the Escherichia coli cell surface was directly confirmed by immunofluorescence microscopy and flow cytometry. After 6 days of incubation in media containing 1 % used cooking oil, an Escherichia coli strain expressing surface displayed lipase was able to degrade 27 % of the oil. The biosurfactant, pseudopyronine B, was purified from culture supernatants of Pseudomonas sp. SL31. Its critical micelle concentration was determined to be 1400 mg/l, and the surfactant was stable within a temperature range from 0 to 120 °C and a pH range of 3–11. Pseudopyronine B-containing crude media extracts efficiently removed up to 51 % of the cadmium from contaminated water. We demonstrated the oil degradation ability of the mixed culture of four bacterial strains, namely the recombinant Escherichia coli expressing cell surface displayed lipase (pKKJlipA), His-tagged lipase (pETlipA), extracellular lipase-producing Proteus sp. SW1, and pseudopyronine B-producing Pseudomonas sp. SL31 by culturing in LB media containing 1 % oil. The consortium degraded 29 % of oil in one day and reached 84 % after 7 days.








Similar content being viewed by others
References
Abalos A et al (2004) Enhanced biodegradation of casablanca crude oil by a microbial consortium in presence of a rhamnolipid produced by Pseudomonas aeruginosa AT10. Biodegradation 15(4):249–260
Gurung N et al (2013) A broader view: microbial enzymes and their relevance in industries, medicine, and beyond. BioMed Res Int 2013:1–18
Andersen O (1984) Chelation of cadmium. Environ Health Perspect 54:249–266
Becker S et al (2005) A generic system for the Escherichia coli cell-surface display of lipolytic enzymes. FEBS Lett 579(5):1177–1182
Bodour AA, Drees KP, Maier RM (2003) Distribution of biosurfactant-producing bacteria in undisturbed and contaminated arid southwestern soils. Appl Environ Microbiol 69(6):3280–3287
Cammarota MC, Freire DMG (2006) A review on hydrolytic enzymes in the treatment of wastewater with high oil and grease content. Bioresour Technol 97(17):2195–2210
Cerqueira VS et al (2011) Biodegradation potential of oily sludge by pure and mixed bacterial cultures. Bioresour Technol 102:11003–11010
Chang JS et al (2004) Enhancement of phenanthrene solubilization and biodegradation by trehalose lipid biosurfactants. Environ Toxicol Chem 23(12):2816–2822
Chang SH et al (2005) Remediation of metal-contaminated soil by an integrated soil washing-electrolysis process. Soil Sediment Contam 14(6):559–569
Chu M et al (2002) Structure of sch 419560, a novel α-pyrone antibiotic produced by Pseudomonas fluorescens. J Antibiot 55(2):215–218
Desai JD, Banat IM (1997) Microbial production of surfactants and their commercial potential. Microbiol Mol Bio Rev 61(1):47–64
Dharmsthiti S, Kuhasuntisuk B (1998) Lipase from Pseudomonas aeruginosa LP602: biochemical properties and application for wastewater treatment. J Ind Microbiol Biotechnol 21(1–2):75–80
Giddens AC et al (2008) Natural product inhibitors of fatty acid biosynthesis: synthesis of the marine microbial metabolites pseudopyronines A and B and evaluation of their anti-infective activities. Tetrahedron Lett 64(7):1242–1249
Gupta R et al (2015) Molecular and functional diversity of yeast and fungal lipases: their role in biotechnology and cellular physiology. Prog Lipid Res 57:40–54
Hasman H, Chakraborty T, Klemm P (1999) Antigen-43-mediated autoaggregation of Escherichia coli is blocked by fimbriation. J Bacteriol 181(16):4834–4841
Henderson IR, Owen P (1999) The major phase-variable outer membrane protein of Escherichia coli structurally resembles the immunoglobulin A1 protease class of exported protein and is regulated by a novel mechanism involving dam and OxyR. J Bacteriol 181(7):2132–2141
Jorfi S et al (2013) Application of biosurfactants produced by Pseudomonas aeruginosa SP4 for bioremediation of soils contaminated by pyrene. Soil Sediment Contam 22(8):890–911
Jose J, Maas RM, Teese MG (2012) Autodisplay of enzymes—molecular basis and perspectives. J Biotechnol 161(2):92–103
Juwarkar AA et al (2007) Biosurfactant technology for remediation of cadmium and lead contaminated soils. Chemosphere 68(10):1996–2002
Kim J, Vipulanandan C (2006) Removal of lead from contaminated water and clay soil using a biosurfactant. J Environ Eng 132:777–786
Kjaergaard K et al (2002) Antigen 43-mediated autotransporter display, a versatile bacterial cell surface presentation system. J Bacteriol 184(15):4197–4204
Koma D et al (2003) Degradation of car engine base oil by Rhodococcus sp. NDKK48 and Gordonia sp. NDKY76A. Biosci Biotechnol Biochem 67(7):1590–1593
Lee SH, Lee SY, Park BC (2005) Cell surface display of lipase in Pseudomonas putida KT2442 using OprF as an anchoring motif and its biocatalytic applications. Appl Environ Microbiol 71(12):8581–8586
Maier RM et al (2001) Remediation of metal-contaminated soil and sludge using biosurfactant technology. Int J Occup Med Environ Health 14(3):241–248
McInerney MJ, Javaheri M, Nagle DP (1990) Properties of the biosurfactant produced by Bacillus licheniformis strain JF-2. J Ind Microbiol 5(2–3):95–101
Mongkolthanaruk W, Dharmsthiti S (2002) Biodegradation of lipid-rich wastewater by a mixed bacterial consortium. Int Biodeterior Biodegrad 50(2):101–105
Mukherjee AK, Bordoloi NK (2011) Bioremediation and reclamation of soil contaminated with petroleum oil hydrocarbons by exogenously seeded bacterial consortium: a pilot-scale study. Environ Sci Technol 18(3):471–478
Phugare SS et al (2011) Textile dye degradation by bacterial consortium and subsequent toxicological analysis of dye and dye metabolites using cytotoxicity, genotoxicity and oxidative stress studies. J Hazard Mater 186(1):713–723
Płaza GA, Zjawiony I, Banat IM (2006) Use of different methods for detection of thermophilic biosurfactant-producing bacteria from hydrocarbon-contaminated and bioremediated soils. J Pet Sci Technol 50(1):71–77
Pornsunthorntawee O et al (2008) Structural and physicochemical characterization of crude biosurfactant produced by Pseudomonas aeruginosa SP4 isolated from petroleum-contaminated soil. Bioresour Technol 99(6):1589–1595
Robichon C et al (2011) Engineering Escherichia coli BL21 (DE3) derivative strains to minimize E. coli protein contamination after purification by immobilized metal affinity chromatography. Appl Environ Microbiol 77(13):4634–4646
Rodrigues LR et al (2006) Physicochemical and functional characterization of a biosurfactant produced by Lactococcus lactis 53. Colloids Surf B 49(1):79–86
Rufino RD et al (2012) Application of the biosurfactant produced by Candida lipolytica in the remediation of heavy metals. Chem Eng Trans 27:61–66
Saeki H et al (2009) Oil spill remediation by using the remediation agent JE1058BS that contains a biosurfactant produced by Gordonia sp. strain JE-1058. Bioresour Technol 100(2):572–577
Sandrin TR, Chech AM, Maier RM (2000) A rhamnolipid biosurfactant reduces cadmium toxicity during naphthalene biodegradation. Appl Environ Microbiol 66(10):4585–4588
Singh MP et al (2003) Novel alpha-pyrones produced by a marine Pseudomonas sp. F92S91: taxonomy and biological activities. J Antibiot 56:1033–1044
Stelmack PL, Gray MR, Pickard MA (1999) Bacterial adhesion to soil contaminants in the presence of surfactants. Appl Environ Microbiol 65(1):163–168
Tajmir-Riahi HA (1989) Carbohydrate metal ion complexes. Interaction of d-glucono-1,5-lactone with Zn(II), Cd(II), and Hg(I1) ions in the solid and aqueous solution, studied by 13C-NMR, FT-IR, and X-ray powder diffraction measurements. Can J Chem 67:651–654
Tan H et al (1994) Complexation of cadmium by a rhamnolipid biosurfactant. Environ Sci Technol 28(13):2402–2406
Whangsuk W et al (2013) Gene cloning and characterization of a novel highly organic solvent tolerant lipase from Proteus sp. SW1 and its application for biodiesel production. Mol Biotechnol 53(1):55–62
Youssef NH et al (2004) Comparison of methods to detect biosurfactant production by diverse microorganisms. J Microbiol Methods 56:339–347
Acknowledgments
This research work was supported in part by a grant from the Chulabhorn Research Institute and Center of Excellence on Environmental Health and Toxicology, Thailand. Special thanks go to Piyajit Watcharasit for Texas red-conjugated anti-rabbit IgG antibody, Nuchanart Rangkadilok and Supachai Ritruechai for preparative HPLC, Amnart Khongmanee for his kind assistance with fluorescence microscopes, James Dubbs for critical reading and discussion, Pakwilai Chouichit for technical assistance, and also to Piyapol Munpiyamit for photograph preparation. We appreciate the funding by the Royal Golden Jubilee Ph.D. Program (PHD/0144/2554) to Sirinthra Thiengmag.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Thiengmag, S., Chuencharoen, S., Thasana, N. et al. Bacterial consortium expressing surface displayed, intra- and extracellular lipases and pseudopyronine B for the degradation of oil. Int. J. Environ. Sci. Technol. 13, 2067–2078 (2016). https://doi.org/10.1007/s13762-016-1034-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-016-1034-z