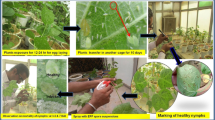
Abstract
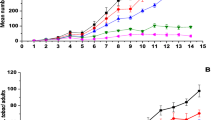
Entomopathogens (EPFs) are potential alternatives to chemical insecticides for managing Bemisia tabaci (Genn.), an invasive pest of the cotton crop. EPFs alone may not always provide enough insect pest control, but combining EPFs with pesticides, provided both components are compatible, can make an integrated pest management program considerably more effective. Hence, the bioefficacy of EPFs against whitefly, their compatibility with pesticides, and the factors responsible for determining compatibility were studied. The highest nymphal mortality was recorded with the Beauveria bassiana strains Bb-4511 (95.1%) and Bb-4565 (89.9%), and Metarhizium anisopliae Ma-1299 (86.7%) at 1 × 106 conidia ml−1. Lower LC50 values were observed for Cordyceps javanica Cj-089 and Bb-4511, 0.2 × 104 and 0.5 × 104 conidia ml−1, respectively. The toxicity index values in insecticide sensitivity assays ranged from 19.4 to 119.6% among all the EPFs. Comparatively, all the EPFs except Bb-4543 and Bb-4565 showed compatible to moderately toxic reactions to neonicotinoids and spinosyns. Organophosphates (ethion) and pyrethrins (bifenthrin) were toxic to very toxic to all the EPFs except Bb-4511, Fv-083, and Ma-1299. Cj-102 and Cj-089 were compatible with 50% of the average recommended dose of bifenthrin and ethion, and the average recommended dose for the field application of neonicotinoids and spinosyns. Principal component analysis showed that spore production and toxicity index values correlate with each other and are responsible for determining the EPF compatibility with insecticides. The EPF spore production and toxicity index are important factors for determining chemical compatibility. Compatible EPFs can be used individually or in combination as promising and compatible biological alternatives to insecticides in the management of whitefly in cotton.




Similar content being viewed by others
References
Alves SB, Moino A Jr, Almeida JEM (1998) Produtos fitossanitarios entomopatogenos. In: Alves SB (ed) Controle Microbiano de Insetos. Piracicaba, FEALQ, Portuguese, pp 217–238.
Anderson TE, Roberts DW (1983) Compatibility of Beauveria bassiana isolate with insecticide formulations used in Colorado Potato Beetle (Coleoptera: Chrysomelidae) control. J Econ Entomol 76:1437–1441
Anonymous (2021) Central Insecticide Board & Registration Committee. Available: http://cibrc.nic.in/. Accessed 2 Dec 2021
Ashfaq M, Hebert PDN, Mirza MS, Khan AM, Mansoor S, Shah GS, Zafar Y (2014) DNA barcoding of Bemisia tabaci complex (Hemiptera: Aleyrodidae) reveals southerly expansion of the dominant whitefly species on cotton in Pakistan. PLoS ONE 9:e104485
Basit MS, Saeed MA, Saleem I, Denholm, Shah M (2013) Detection of resistance, cross-resistance, and stability of resistance to new chemistry insecticides in Bemisia tabaci (Homoptera: Aleyrodidae). J Econ Entomol 106:1414–1422
Bhattacharya S, Subrata D, Tapamay D (2004) In vitro compatibility of different entomopathogens to pesticides, plant growth regulators and micronutrients. Annals Pl Prot Sci 12:199–202
Biswas KK, Bhattacharyya UK, Palchoudhury S, Balram N, Kumar A, Arora R, Sain SK, Kumar P, Khetarpal RK, Sanyal A, Mandal PK (2020) Dominance of recombinant cotton leaf curl Multan-Rajasthan virus associated with cotton leaf curl disease outbreak in northwest India. PLoS ONE 15:e0231886
Bitsadze N, Jaronski S, Khasdan V, Abashidze E, Abashidze M, Latchininsky A, Samadashvili D, Sokhadze I, Rippa M, Ishaaya I, Horowitz AR (2013) Joint action of Beauveria bassiana and the insect growth regulators diflubenzuron and novaluron, on the migratory locust, Locusta migratoria. J Pest Sci 86:293–300
Borisade OA, Mahan N (2015) Resilience and relative virulence of strains of entomopathogenic fungi under interactions of abiotic and stress. African J Microb Res 9:988–1000
Chandler D, Heale JB, Gillespie AT (1994) Effect of osmotic potential on the germination of conidia and colony growth of Verticillium lecanii. Mycol Res 98:384–388
Crowder DW, Horowitz AR, De Barro PJ (2010) Mating behavior, life history and adaptation to insecticides determine species exclusion between whiteflies. J Anim Ecol 79:563–570
De Barro PJ, Liu SS, Boykin LM, Dinsdale AB (2011) Bemisia tabaci: a statement of species status. Annu Rev Entomol 56:1–19
de Olivera RC (2004) Neves PMOJ, Compatibility of Beauveria bassiana with Acaricides. Neotropical Entomol 33:353–358
Dinsdale A, Cook L, Riginos C, Buckley YM, Barro PD (2010) Refined global analysis of Bemisia tabaci (Gennadius) (Hemiptera: Sternorrhyncha: Aleyrodidae) mitochondrial CO1 to identify species level genetic boundaries. Ann Entomol Soc Am 103:196–208
Duarte A, Menendez JM, Triguero N (1992) Preliminary study on the compatibility of Metarhizium anisopliae with some chemical pesticides. Rev Baracoa 22:31–39
Ellango R, Singh ST, Rana VS, Gayatri Priya N, Raina H, Chaubey R, Naveen NC, Mahmood R, Ramamurthy VV, Asokan R, Rajagopal R (2015) Distribution of Bemisia tabaci genetic groups in India. Environ Entomol 44:1258–1264
Faraji S, Shadmehri AD, Mehrvar A (2016) Compatibility of entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae with some pesticides. J Entomol Soc Iran 36:137–146
Faria M, Hotchkiss JH, Hajek AE, Wraight SP (2010) Debilitation in conidia of the entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae and implication with respect to viability determinations and mycopesticide quality assessments. J Invertebr Pathol 105:74–83
Faria M, Wraight SP (2001) Biological control of Bemisia tabaci with fungi. Crop Protect 20:767–778
Finney DJ (1971) Probit analysis. 3. Aufl. Cambridge University Press, Cambridge. XV, 333 S., 41 Rechenbeispiele, 20 Diagr., 8 Tab., 231 Lit., L 5.80
Goettel M, Inglis GD (1997) Fungi: Hyphomycetes. In: Lacey L (ed) Manual of techniques in insect pathology. Academic Press, San Diego, pp 213–248
Huang X, Madan A (1999) CAP3: A DNA sequence assembly program. Genome Res 9:868–877
Humber RA (2012) Identification of entomopathogenic fungi. In: Lacey LA (ed) Manual of Techniques in Invertebrate Pathology. Academic Press London, pp 151–187
James RR, Elzen GW (2001) Antagonism between Beauveria bassiana and Imidacloprid when combined for Bemisia argentifolii control. J Econ Ent 94:357–361
Kanakala S, Ghanim M (2019) Global genetic diversity and geographical distribution of Bemisia tabaci and its bacterial endosymbionts. PLoS ONE 14:e0213946
Karthikeyan A, Selvanarayanan V (2011) In vitro efficacy of Beauveria bassiana (Bals.) Vuill. and Verticillium lecanii (Zimm.) Viegas against selected insect pests of cotton. Recent Res Sci Tech 3:142–143
Khun KK, Ash GJ, Stevens MM, Huwer RK, Wilson BAL (2021a) Interactions of fungal entomopathogens with synthetic insecticides for the control of Kuschelorhynchus macadamiae (Coleoptera: Curculionidae). J Appl Entomol 145. https://doi.org/10.1111/jen.12879
Khun KK, Ash GJ, Stevens MM, Huwer RK, Wilson BAL (2021b) Transmission of Metarhizium anisopliae and Beauveria bassiana to adults of Kuschelorhynchus macadamiae (Coleoptera: Curculionidae) from infected adults and conidiated cadavers. Sci Rep 11:2188. https://doi.org/10.1038/s41598-021-81647-0
Lacey LA, Grywacz D, Shapiro-Iln DI, Frutos R, Brownbridge M, Goettel MS (2015) Insect pathogens as biological control agents: back to the future. J Invertebr Pathol 132:1–41
Longhurst C, Babcock JM, Denholm I, Gorman K, Thomas JD, Sparks TC (2013) Cross-resistance relationships of the sulfoximine insecticide sulfoxaflor with neonicotinoids and other insecticides in the whiteflies Bemisia tabaci and Trialeurodes vaporariorum. Pest Manag Sci 69:809–813
Lu S, Chen M, Li J, Shi Y, Gu Q, Yan F (2019) Changes in Bemisia tabaci feeding behaviors caused directly and indirectly by cucurbit chlorotic yellows virus. Virol J 16:1–14
Mascarin GM (2013) The virulence of entomopathogenic fungi against Bemisia tabaci biotype B (Hemiptera: Aleyrodidae) and their conidial production using solid substrate fermentation. Biol Control 66:209–218
Moino A Jr, Alves SB (1998) Effects of imidacloprid and fipronil on Beauveria bassiana (Bals.) Vuill. and Metarhizium anisopliae (Metsch.) Sorok. and on the grooming behavior of Heterotermes tenuis (Hagen) An. Soc Entomol Brasil 27:611–619
Monga D (2014) Cotton leaf curl virus diseases. Technical Bulletin No.2/2014. Central Institute for Cotton Research, Regional Station, Sirsa, India, pp 1–34
Monga D, Sain SK (2021) Incidence and severity of cotton leaf curl virus disease on different BG II hybrids and its effect on the yield and quality of cotton crop. J Environ Biol 42(1):90–98. http://www.jeb.co.in/journal_issues/202101jan21/paper11.pdf
Morales FJ (2007) Tropical Whitefly IPM Project. Adv Virus Res 69:249–311
Naveen NC, Chaubey R, Kumar D, Rebijith KB, Rajagopal R, Subrahmanyam B, Subramanian S (2017) Insecticide resistance status in the whitefly, Bemisia tabaci genetic groups Asia-I, Asia-II-1 and Asia-II-7 on the Indian subcontinent. Sci Rep 7:40634
Nene YL, Thapliyal PN (2018) Fungicides in plant disease control, 4th edn. Medtech Publisher
Neves PMOJ, Hirose E, Tchujo PT (2001) Compatibility of entomopathogenic fungi with neonicotinoid insecticides. Neotropi Entomol 30:263–268
Oliveira CN, Neves PMOJ, Kawazoe LS (2003) Compatibility between the entomopathogenic fungus Beauveria bassiana and insecticides used in coffee plantations. Sci Agricola 60:663–667
Pan LL, Cui XY, Chen QF, Wang XW, Liu SS (2018) Cotton leaf curl disease: which whitefly is the vector? Phytopath 108:1172–1183
Polston JE, De Barro P, Boykin LM (2014) Transmission specificities of plant viruses with the newly identified species of the Bemisia tabaci species complex. Pest Manag Sci 70:1547–1552
Puntener W (1981) Manual for field trials in plant protection, 2nd edn. Ciba-Geigy, Ltd, Basle
Quesada-Moraga E, Maranhão EA, Valverde-Garcia P, Santiago-Álvarez C (2006) Selection of Beauveria bassiana isolates for control of the whiteflies Bemisia tabaci and Trialeurodes vaporarium on the basis of their virulence, thermal requirement and toxicogenic activity. Biol Control 36:274–287
Sain SK, Monga D, Kumar R, Nagrale DT, Hiremani NS, Kranthi S (2019a) Compatibility of entomopathogenic fungi with insecticides and their efficacy for IPM of Bemisia tabaci in cotton. J Pesti Sci 44:97–105
Sain SK, Monga D, Kumar R, Nagrale DT, Hiremani NS, Kranthi S, Kranthi KR (2019b) Comparative effectiveness of bioassay methods in identifying the most virulent entomopathogenic fungal strains to control Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae). Egypt J Biol Pest Control 29:31–1–11
Sain SK, Monga D, Hiremani NS, Nagrale DT, Kranthi S, Kumar R, Kranthi KR, Tuteja OP, Waghmare VN (2021) Evaluation of bioefficacy potential of entomopathogenic fungi against the whitefly (Bemisia tabaci Genn.) on cotton under polyhouse and field conditions. J Inverteb Patho 183:107618
Sheoran OP, Tonk DS, Kaushik LS, Hasija RC, Pannu RS (1998) Statistical Software Package for Agricultural Research Workers. In: Hooda DS, Hasija RC (eds) Recent Advances in information theory, Statistics and Computer Applications, Department of Mathematics Statistics. CCS HAU, Hisar, pp 139–143
Simon C, Frati F, Beckenbach A, Crespi B, Liu H, Flook P (1994) Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers. Ann Entomol Soc Am 87:651–701
Steinkraus DC (1996) Control of tarnished plant bug with Beauveria bassiana and interactions with imidacloprid, pp. 888–889. In Proc, Beltwide Cotton Conference, Nashville, TN. National Cotton Council, Memphis, TN (9–12 January)
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press Inc, New York, pp 315–322
Acknowledgements
The authors are thankful to ICAR—Central Institute for Cotton Research for providing the necessary laboratory, field facilities, and funding for this investigation under the institute project.
Funding
The research work was funded by the Indian Council of Agricultural Research—Central Institute for Cotton Research, Nagpur (India), institutional research project funds.
Author information
Authors and Affiliations
Contributions
SKS—planned and implemented the research experiments, compiled and analyzed the data, and wrote the manuscript; DM, SK, RK, NH, DT—planned the research experiments and edited the manuscript. All the authors read, reviewed, and edited the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Edited by Lessando Moreira Gontijo
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sain, S.K., Monga, D., Kranthi, S. et al. Evaluation of the Bioefficacy and Insecticide Compatibility of Entomopathogens for Management of Whitefly (Hemiptera: Aleyrodidae) on Upland Cotton Under Laboratory and Polyhouse Conditions. Neotrop Entomol 51, 600–612 (2022). https://doi.org/10.1007/s13744-022-00964-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-022-00964-9