Abstract
Fitness of a single species is often measured in terms of the number of viable offspring produced. We explore the relationship between the number of ovarioles and the size of females in five species of Simuliidae in two páramo regions of Colombia. Individuals of seven species of female blackflies that landed on animals were collected from mules in the Ucumarí Regional Park (RPNU) and from cattle in the Chingaza Natural National Park (ChNNP). The ovarioles of the five most abundant species were dissected out and counted, and a one-factor ANOVA was performed to explore differences in the mean number of ovarioles produced by different female size categories. Simulium ignescens Roubaud and S. ignescens-like species were collected in RPNU and S. ignescens, Simulium muiscorum Bueno et al, Simulium cormonsi Wygodzinsky and Simulium pautense Coscarón & Takaoka, in ChNNP. In addition, we also analyzed the Pearson product-moment correlation between the mean number of ovarioles per blackfly and female size within species using those which more than 20 individuals were collected. Ovarioles were meroistic-polytrophic type. A multiple range test (least significant difference) indicated that the largest size group had the largest mean number of ovarioles per female. In the other size groups, there were approximately equal numbers of ovarioles per female. Pearson’s correlation coefficient was separately measured and indicated a 50% correlation between female size and ovariole number in S. ignescens. Although ovariole number is probably a genetically driven feature, other environmental and physiological conditions, including infections, can influence the number of eggs.


Similar content being viewed by others
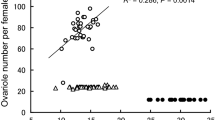
References
Adler PH, Crosskey RW (2014) World blackflies (Diptera:Simuliidae): a comprehensive revision of the taxonomic and geographical inventory. http://www.clemson.edu/cafls/biomia/pdfs/blackflyinventory.pdf. Accessed 15 Apr 2014.
Adler PH, McCreadie JW (2004) In: Mullen G, Durden L (eds) Medical and veterinary entomology. Academic, Amsterdam, Boston, pp 185–202
Bader CA, Williams CR (2012) Mating ovariole number and sperm production of the dengue vector mosquito Aedes aegypti (L.) in Australia: broad thermal optima provide the capacity for survival in a changing climate. Physiol Entomol 37:137–144
Baum JS, St George JP, McCall K (2005) Programmed cell death in the germline. Cell Dev Biol 16:245–259
Begon M, Townsend CR, Harper JL (2006) Ecology. From individuals to ecosystem. 4th edn. Blackwell publishing. Malden Oxford Victoria, p 739
Carder (2008) Plan de manejo Ucumarí. http://www.carder.gov.co/web/es/pnr-ucumar. Accesed 23 Jan 2014
Chaiwong T, Sukontason K, Chaisri U, Kuntalue B (2012) Ovarian ultrastructure and development of the blow fly, Chrysomyia megacephala (Diptera: Calliphoridae). Int J Parasitol Res 4(1):65–70
Cheke RA, Garms R, Kerner M (1982) The fecundity of Simulium damnosum s.l. in northern Togo and infections with Onchocerca spp. Ann Trop Med Parasitol 76:561–568
Chutter FM (1970) A preliminary study of factors influencing the number of oocytes present in newly emerged blackflies (Diptera: Simuliidae) in Ontario. Can J Zool 48:1389–1400
Colbo MH (1982) Size and fecundity of adult Simuliidae (Diptera) as a function of stream habitat, year and parasitism. Can J Zool 60:2507–2513
Coscarón S, Coscarón-Arias C (2007) Neotropical Simuliidae (Diptera: Insecta). In: Adis J, Arias JR, Rueda-Delgado G, Wantzen KM (eds). Aquatic biodiversity in Latin America. Vol. 3 edn Pensoft, Sofía Moscú, p 686
Crosskey RW (1990) The natural history of blackflies. London, The British Museum of Natural History. John Wiley and Sons, p 711
Fox CH, Czesak ME (2000) Evolutionary ecology of progeny size in arthropods. Ann Rev Entomol 45:341–369
Mahmood F, Crans WJ (1998) Ovarian development and parity termination in Culiseta melanura (Diptera: Culicidae). J Med Entomol 35(6):980–988
Malmqvist B, Adler PH, Strasevicius D (2004a) Testing hypotheses on egg number and size in black flies (Diptera: Simuliidae). J Vector Ecol 29(2):248–256
Malmqvist B, Strasevicius D, Hellgren O, Adler PH, Bensch S (2004b) Vertebrate host specificity of wild caught blackflies revealed by mictochondrial DNA in blood. Proc R Soc B 271(S4):S152–S155
Mantilla JS, Moncada LI, Matta NE, Adler P (2013) Two new species of Black Flies (Diptera: Simuliidae) from the high Andes of Colombia. Zootaxa 3700(3):423–434
Mokry JE (1980) Laboratory studies on blood feeding of black flies (Diptera: Simuliidae). 1. Factors affecting fecundity. Tropenmed Parasitol 21:374–380
Moncada LI, Mantilla JS, Lotta I, Matta N, Adler P (2013) Simulium cormonsi Wygodzinsky (Diptera: Simuliidae) in Colombia: first record egg and larval description and bionomics. Proc Entomol Soc Wash 1156(1):103–106
Muñoz de Hoyos P, Coscarón S (1999) Claves para la identificación de Simúlidos (Diptera: Simuliidae) presentes entre las vertientes Magdalenense y Orinocense, en un sector al centro de Colombia. Rev Acad Colom Cien 23:181–214
Papaj D (2000) Ovarian dynamics and host use. Ann Rev Entomol 45:423–448
Petry F, Lozovei AL, Chahad-Ehiers S (2006) Eggs fertility and biological aspects of the life cycle in natural populations of Simulium (Diptera, Simuliidae). Braz Arch Biol Tech 49(5):799–805
Quiroga SF (2013) Plan estratégico para el ecoturismo en las áreas protegidas de la cuenca alta del río Otún. http://www.pereiraculturayturismo.gov.co/files/descargasTurismo/Diagnostico_proyecto_FOCA_version_final.pdf. Accesed 25 Jan 2014
R’ kha S, Moreteau B, Coyne JA, David JR (1997) Evolution of a lesser fitness trait: egg production in the specialist Drosophila sechellia. Genet Res 69(1):17–23
Raikhel AS, Dhadialla TS (1992) Accumulation of yolk proteins in insect oocytes. Ann Rev Entomol 37:217–251
Smith CC, Fretwell SD (1974) The optimal balance between size and number of offspring. Am Nat 108:499–506
Snodgrass RE (1935) Principles of insect morphology. McGrawHill, New York, p 513
Steinwascher K (1984) Egg size variation in Aedes aegypti relationship to body size and other variables. Am Mid Nat 112(1):76–84
Vargas O, Pedraza P (2004) El Parque Natural Nacional Chingaza. Departamento de Biología. Facultad de Ciencias. Universidad Nacional de Colombia. Colciencias, UAESPNN, EAAB. Bogotá DC, p 197
Vásquez VJ, Serrano MA (2009) Las Áreas Naturales Protegidas de Colombia. Conservación Internacional—Colombia & Fundación Biocolombia. Panamericana Formas e Impresos. Bogotá, Colombia. pp 308–309
West-Eberhard MJ (2003) Developmental plasticity and evolution. Oxford University Press. p 367
Xu L, Zhou C, Xiao Y, Zhang P, Tang Y, Xu Y (2012) Insect oviposition plasticity in response to host availability: the case of the tephritid fruit fly Bactrocerca dorsalis. Ecol Entomol 37:446–452
Acknowledgments
Financial support for this work from Ecopetrol, Colciencias, and the Universidad Nacional de Colombia is gratefully acknowledged. We are also very grateful to Parques Nacionales, Aguas y Aguas of Pereira, the students of Medical Entomology Laboratory of Universidad Nacional de Colombia, LEMUN, and the Relation Parasite-Host team at the Universidad Nacional de Colombia.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by Eunice AB Galati – FSP/USP
Rights and permissions
About this article
Cite this article
Esquivel, L.A., Moncada, L.I., Panza, C.A. et al. The Relationship Between Ovarioles Number and Female Size in Blackflies of the High Andes of Colombia. Neotrop Entomol 44, 92–97 (2015). https://doi.org/10.1007/s13744-014-0253-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-014-0253-6


