Abstract
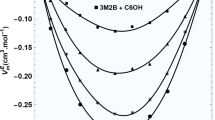
A green 1,3-propanediamine (PDA)-based deep eutectic solvent (DES) was obtained using 1,4-butanediol (BDO) as the H bond donor (HBD), in which the basic physical and chemical properties of the binary system, such as density, viscosity and surface tension, were systematically studied under atmospheric pressure at T = 293.15–318.15 K. The excess Gibbs free energy (GE), apparent molar volumes (Vφ,1 and Vφ,2), partial molar volumes (V1 and V2) and isobaric thermal expansion coefficient (αp) of the binary system were also calculated. After that, the molecular configurations and intermolecular interaction mode of the BDO + PDA-based deep eutectic solvent were simulated and calculated by the Gaussian software; then, the intermolecular interaction between the BDO and PDA was explored by spectral technologies. Based on these discussions, it is confirmed that there is a hydrogen bond (HB) between the H atom of BDO and the N atom of PDA.












Similar content being viewed by others
Change history
18 November 2021
A Correction to this paper has been published: https://doi.org/10.1007/s13738-021-02452-0
References
J.P. Zhang, Z.W. Li, Z.H. Zhang, R. Liu, B.Z. Chu, B. Yan, ACS Sustain. Chem. Eng. 8, 18062–18070 (2020)
K.R. Gurney, Y. Song, J.M. Liang, G. Roest, Environ. Sci. Technol. 54, 9896–9907 (2020)
L.W. Sun, S.K. Tang, Korean J. Chem. Eng. 36, 1708–1715 (2019)
M.A. Kareem, F.S. Mjalli, M.A. Hashim, I.M. Alnashef, J. Chem. Eng. Data 55, 4632–4637 (2010)
A. Krishnan, K.P. Gopinath, D.-V.N. Vo, R. Malolan, V.M. Nagarajan, J. Arun, Environ. Chem. Lett. 18, 2031–2054 (2020)
U. Saeed, A.L. Khan, M.A. Gilani, M. Aslam, A.U. Khan, Environ. Sci. Pollut. R. (2020). https://doi.org/10.1007/s11356-020-10260-x
M.B. Haider, D. Jha, B.M. Sivagnanam, R. Kumar, J. Chem. Eng. Data 63, 2671–2680 (2018)
A.P. Abbott, G. Capper, D.L. Davies et al., Chem. Commun. 9, 70–71 (2003)
W. Bi, M. Tian, K.L. Row, J. Chromatogr. A 1285, 22–30 (2013)
N. Azizi, E. Batebi, Catal. Sci. Technol. 2, 2445 (2012)
F. Aydin, E. Yilmaz, M. Soylak, Microchem. J. 132, 280–285 (2017)
E. Yilmaz, M. Soylak, Talanta 160, 680–685 (2016)
X. Li, M. Hou, B. Han, X. Wang, L. Zou, J. Chem. Eng. Data 53, 548–550 (2008)
G. Garcia, S. Aparicio, R. Ullah, M. Atilhan, Energ. Fuel. 29, 2616–2644 (2015)
G. Li, D. Deng, Y. Chen, H. Shan, N. Ai, J. Chem. Thermodyn. 75, 58–62 (2014)
P.A. Bhawna, S. Pandey, ChemistrySelect 2, 11422–11430 (2017)
S. Sarmad, J.P. Mikkola, X. Ji, Chemsuschem 10, 324–352 (2017)
O.V. Grineva, V.I. Zhuravlev, Densities and dielectric permittivities of three butanediols and their solutions with 10 mol% water. J. Chem. Eng. Data 41, 604–607 (1996)
E. Zorebski, M. Dzida, J. Chem. Thermodyn. 54, 100–107 (2012)
J. Abolghasem, K. Maryam, V.G. Zahra, Chem. Pharm. Bull. 53, 519–523 (2005)
B. Hawrylak, S. Andrecyk, C. Gabriel, J. Solut. Chem. 27, 827–841 (1998)
T. Sun, R.M. DiGuilio, A.S. Teja, J. Chem. Eng. Data 37, 246–249 (1992)
A.K. Nain, Fluid Phase Equilib. 265, 46–56 (2008)
C.S. Yang, S.P. Ma, Q. Zhou, J. Chem. Eng. Data 49, 582–587 (2004)
H.Z. Zhang, J.R. Rustad, J.F. Banfield, J. Phy. Chem. A 111, 5008–5014 (2007)
X. Esteve, A. Conesa, A. Coronas, J. Chem. Eng. Data 48, 392–397 (2003)
H.C. Ku, C.H. Tu, J. Chem. Eng. Data 45, 391–394 (2000)
H. Kiaee, A.A. Rostami, D. Farmanzadeh, J. Mol. Liq. 231, 242–248 (2017)
A. Pal, H. Kumar, Fluid Phase Equilib. 181, 17–32 (2001)
S.J. Zhang, X. Li, H.P. Chen, J.F. Wang, M.I. Zhang, J. Chem. Eng. Data 49, 760–764 (2004)
J. Nath, J.G. Pandey, J. Chem. Eng. Data 42, 1137–1139 (1997)
M.D.C. Grande, J.A. Julia, M. Garcı, C.M. Marschoff, J. Chem. Thermodyn. 39, 1049–1056 (2007)
J.V. Srinivasua, T.S. Krishna, K. Narendra, G. SrinivasaRao, B. SubbaRao, J. Mol. Liq. 236, 27–37 (2017)
C.S. Yang, G.H. He, Y.F. He, P.S. Ma, J. Chem. Eng. Data 53, 1211–1215 (2008)
N. Ohtori, Y. Kondo, Y. Ishii, J. Mol. Liq. 314, 113764 (2020)
O. Ciocirlan, O. Iulian, Serb. Chem. Soc. 74, 317–329 (2009)
A. Baskar, A.M. Siddiq, M.S. Alam, J. Mol. Liq. 251, 458–469 (2018)
E. Zorebski, P. Góralski, B. Godula, M. Zorebski, J. Chem. Thermodyn. 68, 145–152 (2014)
C. Yang, G. He, Y. He, P. Ma, J. Chem. Eng. Data 53, 1639–1642 (2008)
M. Moosavi, F. Khashei, A. Sharifi, M. Mirzaei, J. Chem. Thermodyn. 107, 1–7 (2017)
D.M. Bajiac, G.R. Ivanis, Z.P. Visak, E.M. Zivkovic, S.P. Serbanovic, M.K. Kijevcanin, J. Chem. Thermodyn. 57, 510–529 (2013)
D. Urszula, K. Merta, K. Walczak, Colloids Surf. A Physicochem Eng. Asp. 436, 504–511 (2013)
S.V. Dzyuba, R.A. Bartsch, Chem. Phys. Chem. 3, 161–166 (2002)
D. Urszula, K. Merta, K. Walczak, Colloid Surf. A 436, 504–511 (2013)
S.L. Yu, Y.W. Li, X.Q. Xia, Spectral Analysis (Chongqing University Press, Chongqing, 1994)
N. Calvar, B. González, E. Gómez, J. Canosa, J. Chem. Eng. Data 54, 1334–1339 (2009)
C.S. Yang, P.S. Ma, F.M. Jing, D.Q. Tang, J. Chem. Eng. Data 48, 836–840 (2003)
L. Zhao, Q. Li, L. Ma, C. Liu, F. Sha, J.B. Zhang, J. Mol. Liq. 241, 374–385 (2017)
G.P. Dubey, M. Sharma, N. , J. Chem. Thermodyn. 40, 309–320 (2008)
G.P. Dubey, K. Kumar, J. Chem. Eng. Data 55, 1700–1703 (2010)
C.S. Yang, W. Xu, P.S. Ma, J. Chem. Eng. Data 49, 1794–1801 (2004)
C.S. Srikanth, S.S.C. Chuang, J. Phys. Chem. C 117, 9196–9205 (2013)
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21666027), the Program for Grassland Excellent Talents of Inner Mongolia Autonomous Region and the Inner Mongolia Science and Technology Key Projects.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, K., Li, B., Zhang, R. et al. Density, viscosity, surface tension, spectroscopic properties and computational chemistry of the 1, 4-butanediol + 1, 3-propanediamine-based deep eutectic solvent. J IRAN CHEM SOC 19, 1203–1217 (2022). https://doi.org/10.1007/s13738-021-02371-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02371-0