Abstract
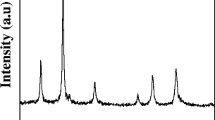
We present an eco-friendly solvothermal method for synthesis of superparamagnetic α-Fe2O3 and Fe3O4 nanoparticles without precipitation agent. The α-Fe2O3 nanoparticles were synthesized with Fe(NO3)3·9H2O as the iron source and absolute ethanol as the solvent at 200 °C. To synthesize Fe3O4 nanoparticles, ascorbic acid was added as the reductant and then the synthesis was performed at 180 (M3 sample) and 200 °C (M4 sample), while other synthesis conditions were fixed. In the case of α-Fe2O3 sample, non-uniform nanoparticles with nearly spherical shapes and rough surfaces were fabricated under solvothermal conditions. Redox reaction between absolute ethanol and NO3¯ from iron source provided a substantial concentration of OH¯ ions needed for the formation of Fe2+ and Fe3+ hydroxides. The average particle sizes of α-Fe2O3, M3, and M4 nanoparticles were obtained to be 18.6, 23.1, and 15.8 nm, respectively.







Similar content being viewed by others
References
X. Li, W. Wei, S. Wang, L. Kuai, B. Geng, Nanoscale 3, 718 (2011)
N.M.A. Rashid, C. Haw, W. Chiu, N.H. Khanis, A. Rohaizad, P. Khiew, S.A. Rahman, CrystEngComm 18, 4720 (2016)
H. Liu, C. Di Valentin, J. Phys. Chem. C 121, 25736 (2017)
J. Chen, L.N. Xu, W.Y. Li, X.L. Gou, Adv. Mater. 17, 582 (2005)
W.T. Qiu, M.S. Balogun, Y. Luo, K.Q. Chen, Y.K. Zhu, X.J. Xiao, X.H. Lu, P. Liu, Y.X. Yong, Electrochim. Acta 193, 32 (2016)
M.S. Balogun, Z. Wu, Y. Luo, W. Qiu, X. Fan, B. Long, M. Huang, P. Liu, Y. Tong, J. Power Sour. 308, 7 (2016)
F. Zhang, X. Yin, W. Zhang, J. Alloys Compd. 657, 809 (2016)
X.J. Chen, J.Q. Wei, X.Y. Wang, P.F. Wang, J. Alloys Compd. 705, 138 (2017)
J. Chang, Q. Ma, J. Ma, H. Ma, Ceram. Int. 42, 11827 (2016)
N. Wang, Y. Du, W. Ma, P. Xu, X. Han, Environ. Appl. Catal. B 210, 23 (2017)
T. Liang, H. Wang, D. Xu, K. Liao, R. Wang, B. He, Y. Gong, C. Yan, Nanoscale 10, 17814 (2018)
L. Wang, Z. Lou, J. Deng, R. Zhang, T. Zhang, A.C.S. Appl, Mater. Interfaces 7, 13098 (2015)
M. Kumari, C.U. Pittman Jr., D. Mohan, J. Colloid, Interface Sci. 442, 120 (2015)
J. Cai, S. Ding, G. Chen, Y. Sun, Q. Xie, Appl. Surf. Sci. 456, 302 (2018)
F. Li, H. Chen, X.Y. Liu, S.J. Zhu, J.Q. Jia, C.H. Xu, F. Dong, Z.Q. Wen, Y.X. Zhang, J. Mater. Chem. A 4, 2096 (2016)
N. Bombuwala-Dewage, A.S. Liyanage, C.U. Pittman Jr., D. Mohan, T. Mlsna, Bioresour. Technol. 263, 258 (2018)
Z. Wu, H. Yu, L. Kuai, H. Wang, T. Pei, B. Geng, J. Colloid, Interface Sci. 426, 83 (2014)
V. Nithya, N.S. Arul, J. Power Sour. 327, 297 (2016)
S. Erogul, S.Z. Bas, M. Ozmen, S. Yildiz, Electrochim. Acta 186, 302 (2015)
D. Chen, Z. Liu, M. Zhou, P. Wu, J. Wei, J. Alloys Compd. 742, 918 (2018)
W. Zhang, K. Banerjee-Ghosh, F. Tassinari, R. Naaman, ACS Energy Lett. 3, 2308 (2018)
D. Trpkov, M. Panjan, L. Kopanja, M. Tadic, Appl. Surf. Sci. 457, 427 (2018)
J.L. Huang, L.Q. Fan, Y. Gu, C.L. Geng, H. Luo, Y.F. Huang, J.M. Lin, J.H. Wu, J. Alloys Compd. 788, 1119 (2019)
Z. Li, C. Lu, Z. Xia, Y. Zhou, Z. Luo, Carbon 45, 1686 (2007)
J.B. Lian, X.C. Duan, J.M. Ma, T.I. Kim, W.J. Zheng, ACS Nano 3, 3749 (2009)
W. Chiu, P. Khiew, M. Cloke, D. Isa, H. Lim, T. Tan, N. Huang, S. Radiman, R. Abd-Shukor, M.A.A. Hamid, C. Chia, J. Phys. Chem. C 114, 8212 (2010)
D.L. Huber, Small 1, 482 (2005)
S. Si, C. Li, X. Wang, D. Yu, Q. Peng, Y.D. Li, Cryst. Growth Des. 5, 391 (2005)
T. Liu, N. Liu, S. Zhai, S. Gao, Z. Xiao, Q. An, D. Yang, J. Alloys Compd. 779, 831 (2019)
M. Zhu, Y. Wang, D. Meng, X. Qin, G. Diao, J. Phys. Chem. C 116, 16276 (2012)
X.F. Sun, X.Q. Qin, L.P. Li, G.S. Li, Inorg. Chem. 47, 4146 (2008)
T. Yan, R.Y. Li, T.T. Yang, Z.J. Li, Electrochim. Acta 152, 530 (2015)
Acknowledgements
The financial support of this study was provided by Sari Agricultural Sciences and Natural Resources University in the form of research project (Projected Number: 03–1398–04).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kazemi, S.Y., Biparva, P. & Pourfaraj, R. Self-synthesis of superparamagnetic α-Fe2O3 and Fe3O4 nanoparticles: an eco-friendly and economic process. J IRAN CHEM SOC 18, 3399–3406 (2021). https://doi.org/10.1007/s13738-021-02277-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02277-x