Abstract
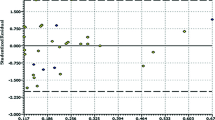
Hepatitis C virus (HCV) is a main cause of liver disease worldwide and a potential cause of considerable malady and mortality in the future. The docking and molecular dynamics simulation (MDs) have been carried out on a series of indole-based derivatives into the cavity of HCV-NS5B polymerase. The docking and MDs led to recognition of the best conformers of inhibitors and also determination of the key interacting amino acids of the binding site of the protein packet. In addition, the conformers are very similar to bioactive ones which in turn will guarantee the most reliable and predictive models in 2D and 3D-QSAR studies. To find the correlation between inhibitory activities of ligands against HCV-NS5B, a comparative molecular field analysis (CoMFA) study, as a 3D-QSAR method, was carried out and the corresponding contour maps of electrostatic and steric fields were computed. Furthermore, 2D descriptors were calculated utilizing the optimal conformers. In the case of 2D-QSAR, a variable selection was applied by genetic algorithm (GA), followed by model building using partial least squares (GA-PLS) and support vector machine (GA-SVM) regression methods. The predictive ability of the proposed models was validated by a structurally diverse test set of 22 compounds that had not been included in the training step. The q 2 and the \(r_{\text{pred}}^{2}\) values for CoMFA, GA-PLS and GA-SVM models were 0.521, 0.655, and 0.746 and 0.659, 0.881, and 0.967, respectively. Moreover, the molecular interactions of these inhibitors with the HCV-NS5B’s active site residues were properly discussed.





Similar content being viewed by others
References
C. Hepatitis, Available via World Health Organization (2013), http://www.who.int/mediacentre/factsheets/fs164/en/. Accessed 2013
T. Ishida, T. Suzuki, S. Hirashima, K. Mizutani, A. Yoshida, I. Ando, S. Ikeda, T. Adachi, H. Hashimoto, Bioorg. Med. Chem. Lett. 16(7), 1859–1863 (2006)
M. Wang, K. Wang, A. Yan, C. Yu, Int. J. Mol. Sci. 13(4), 4033–4047 (2012)
D.C. Myles, Curr. Opin. Drug Discov. Devel. 4(4), 411–416 (2001)
K. Koike, J. Infect Chemother. 12(5), 227–232 (2006)
S. Zeuzem, S.V. Feinman, J. Rasenack, E.J. Heathcote, M-Y Lai, E. Gane, J. O’Grady, J. Reichen, M. Diago, A. Lin, J. Hoffman, M.J. Brunda, New Eng. J. Med. 343(23), 1666–1672 (2000)
Y.H. Yen, C.H. Hung, T.H. Hu, C.H. Chen, C.M. Wu, J.H. Wang, S.N. Lu, C.M. Lee, Aliment Pharmacol. Ther. 27(1), 72–79 (2008)
M. Masarone, M. Persico, Expert Rev. Anti infect. Ther. 9(5), 535–543 (2011)
M. Leise, W.R. Kim, Hepatology 50(4), 1307–1309 (2009)
V.K. Agrawal, R. Sohgaura, P.V. Khadikar, Bioorg. Med. Chem. 10(9), 2919–2926 (2002)
R. Khosrokhavar, J.B. Ghasemi, F. Shiri, Int. J. Mol. Sci. 11(9), 3052–3068 (2010)
P.V. Khadikar, A. Phadnis, A. Shrivastava, Bioorg. Med. Chem. 10(4), 1181–1188 (2002)
H. Sun, D.O. Scott, Chem. Bio. Drug. Des. 75(1), 3–17 (2010)
P.D. Patel, M.R. Patel, N. Kaushik-Basu, T.T. Talele, J. Chem. Inf. Model 48(1), 42–55 (2007)
S. Gharaghani, T. Khayamian, F. Keshavarz, Struct. Chem. 23(2), 341–350 (2012)
K.X. Chen, C.A. Lesburg, B. Vibulbhan, W. Yang, T-Y Chan, S. Venkatraman, F. Velazquez, Q. Zeng, F. Bennett, G.N. Anilkumar, J. Duca, Y. Jiang, P. Pinto, L. Wang, Y. Huang, O. Selyutin, S. Gavalas, H. Pu, S. Agrawal, B. Feld, H-C Huang, C. Li, K-C Cheng, N-Y Shih, J.A. Kozlowski, S.B. Rosenblum, F.G. Njoroge, J. Med. Chem. 55(5), 2089–2101 (2012)
K.X. Chen, B. Vibulbhan, W. Yang, M. Sannigrahi, F. Velazquez, T-Y Chan, S. Venkatraman, G.N. Anilkumar, Q. Zeng, F. Bennet, Y. Jiang, C.A. Lesburg, J. Duca, P. Pinto, S. Gavalas, Y. Huang, W. Wu, O. Selyutin, S. Agrawal, B. Feld, H-C Huang, C. Li, K-C Cheng, N-Y Shih, J.A. Kozlowski, S.B. Rosenblum, F.G. Njoroge, J. Med. Chem. 55(2), 754–765 (2011)
S. Harper, S. Avolio, B. Pacini, M. Di Filippo, S. Altamura, L. Tomei, G. Paonessa, M. Di, A. Carfi, C. Giuliano, J. Padron, F. Bonelli, G. Migliaccio, R. De Francesco, R. Laufer, M. Rowley, F. Narjes, J. Med. Chem. 48(14), 4547–4557 (2005)
H.J.C. Berendsen, D. van der Spoel, R. van Drunen, Comp. Phys. Comm. 91(1–3), 43–56 (1995)
D. van Der Spoel, E. Lindahl, B. Hess, G. Groenhof, A.E. Mark, H.J.C. Berendsen, J. Comp. Chem. 26(16), 1701–1718 (2005)
E. Lindahl, B. Hess, D. van der Spoel, J. Mol. Model. 7(8), 306–317 (2001)
W.F. van Gunsteren, S.R. Billeter, A.A. Eising, P.H. Hünenberger, P. Krüger, A.E. Mark, W.R.P. Scott, I.G. Tironi, Biomolecular Simulation: The GROMOS96 manual and userguide. Hochschuleverlag AG an der ETH Zürich. 1996 (citeulike:8062915)
T. Darden, D. York, L. Pedersen, J. Chem. Phys. 98(12), 10089–10092 (1993)
U. Essmann, L. Perera, M.L. Berkowitz, T. Darden, H. Lee, L.G. Pedersen, J. Chem. Phys. 103(19), 8577–8593 (1995)
W.C. Swope, H.C. Andersen, P.H. Berens, K.R. Wilson, J. Chem. Phys. 76(1), 637–649 (1982)
E.H. Kennard (McGraw-Hill, 1938)
K. Huang, Statistical mechanics (Wiley, New York, 1963)
H.J.C. Berendsen, J.P.M. Postma, W.F. van Gunsteren, J. Hermans, Intermol. Forces. 14, 331–342 (1981)
H.C. Andersen, J. Chem. Phys. 72(4), 2384–2393 (1980)
H.J.C. Berendsen, J.P.M. Postma, W.F. van Gunsteren, A. DiNola, J.R. Haak, J. Chem. Phys. 81(8), 3684–3690 (1984)
M. Thompson, Molecular docking using ArgusLab, an efficient shape-based search algorithm and the AScore scoring function. In: ACS meeting, Philadelphia (2004)
Accelrys Software Inc. San Diego, CA 92121
R. Todeschini, V. Consonni, A. Mauri, M. Pavan, Dragon Software: Calculation of Molecular Descriptors, Department of Environmental Sciences, University of Milano-Bicocca, and Talete, srl. (2003)
A. Mauri, V. Consonni, M. Pavan, R. Todeschini, DRAGON software: an easy approach to molecular descriptor calculations. Match 56(2), 237–248 (2006)
S. Wold, A. Ruhe, H. Wold, I.W. Dunn, SIAM J. SciStat. Comp. 5(3), 735–743 (1984)
M. Clark, R.D. Cramer, Quant. Struct. Act. Rel. 12(2), 137–145 (1993)
B. Bush, R. Nachbar Jr, J. Comput. Aided Mol. Des. (1993). doi:10.1007/bf00124364
R.D. Cramer, J.D. Bunce, D.E. Patterson, I.E. Frank, Quant. Struct. Act. Rel. 7(1), 18–25 (1988)
V. Vapnik, The Nature of Statistical Learning Theory (Springer, 2000)
V. Vapnik, Statistical learning theory (Wiley, New York, 1998)
C. Cortes, V. Vapnik, Support vector machine. Mach. Learn. 20(3), 273–297 (1995)
S. Wold, Technometrics (1978). doi:10.1080/00401706.1978.10489693
A. Golbraikh, A. Tropsha, J. Mol. Graph Model 20(4), 269–276 (2002)
P.P. Roy, K. Roy, QSAR Comb. Sci. 27(3), 302–313 (2008)
A. Golbraikh, M. Shen, Z. Xiao, Y.-D. Xiao, K.-H. Lee, A. Tropsha, J. Comput. Aided Mol. Des. 17(2–4), 241–253 (2003)
M.L. Barreca, N. Iraci, G. Manfroni, R. Gaetani, C. Guercini, S. Sabatini, O. Tabarrini, V. Cecchetti, J Chem Inf Model 54(2), 481–497 (2014)
M. Bohm, J. Sturzebecher, G. Klebe, J. Med. Chem. 42, 458–477 (1999)
M. Daszykowski, B. Walczak, D.L. Massart, Anal. Chim. Acta. 468(1), 91–103 (2002)
R.W. Kennard, LA, Technometrics 11(1), 137–148 (1969)
J.H. Holland, Sci. Am. 267(1), 66–72 (1992)
D.W. Salt, S. Ajmani, R. Crichton, D.J. Livingstone, J. Chem. Inf. Model 47(1), 143–149 (2006)
R. Todeschini, V. Consonni, R. Mannhold, H. Kubinyi, H. Timmerman, Handbook of molecular descriptors (Wiley, 2008)
Z. Mihalic, S. Nikolic, N. Trinajstic, J. Chem. Inf. Comp. Sci. 32(1), 28–37 (1992)
M. Fernández, J. Caballero, A. Tundidor-Camba, Bioorg. Med. Chem. 14(12), 4137–4150 (2006)
G. Moreau, P. Broto, Nouv. J. Chim. 4, 757–764 (1980)
J. Galvez, M. Galvez-Llompart, R. Garcia-Domenech, Green Chem. 12(6), 1056–1061 (2010)
H. Cao, R. Cao, H. Zhang, X. Zheng, D. Gao, Curr. Med. Chem. 15, 1462–1477 (2008)
I. Musmuca, A. Caroli, A. Mai, N. Kaushik-Basu, P. Arora, R. Ragno, J. Chem. Inf. Model 50, 662–676 (2010)
H. Kubinyi, Handbook of chemoinformatics (Wiley-VCH Verlag GmbH, 2008)
J.B. Ghasemi, and H. Tavakoli, Sci. Pharm. 80(3), 547–566 (2012)
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghasemi, J.B., Nazarshodeh, E. & Abedi, H. Molecular docking, 2D and 3D-QSAR studies of new indole-based derivatives as HCV-NS5B polymerase inhibitors. J IRAN CHEM SOC 12, 1789–1799 (2015). https://doi.org/10.1007/s13738-015-0654-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-015-0654-4