Abstract
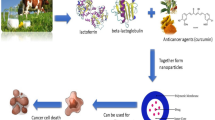
Today, fabrication of new materials in nano scale based on protein-polysaccharide for targeted drug delivery by oral way is unresolved question for researchers in drug delivery system field. In this work, nanoparticles were made by β-lactoglobulin (β-LG), as a globular milk whey protein, complex with low methoxyl pectin (LMP) for chemotherapy agent (oxali-palladium) delivery. Affinity of β-LG to chemotherapy agent was investigated based on the intrinsic fluorescence intensity at 25 °C, so that β-LG binds to chemotherapy agent with molar ratio of 1:1. Hence, the nanoparticles were synthesized at various pH at ambient temperature. The results indicated that the nanoparticles were fabricated smaller than 200 nm at optimal pH approximately close to isoelectric point of β-LG with stability and solubility properties at ambient temperature. Also, scanning electron microscopy results demonstrated that nanoparticles were formed spherical so that were homogeneously distributed. Moreover, the results of dissolution profiles of drug release from β-LG nanoparticle-LMP complex at 37 °C show that in acidic conditions, nanoparticles are stable and there is no significant drug release but in alkaline conditions drug is released from nanoparticles. Therefore, we suggest β-LG nanoparticles-LMP complex are biodegradable and biocompatible and might be useful as targeted oral drug delivery system.





Similar content being viewed by others
Abbreviations
- β-LG:
-
Β-Lactoglobulin
- LMP:
-
Low methoxyl pectin
- DLS:
-
Dynamic light scattering
- SEM:
-
Scanning electron microscopy
References
R. Santipanichwong, M. Suphantharika, J. Weiss, D.J. Mcclements, Core-shell biopolymer nanoparticles produced by electrostatic deposition of beet pectin onto heat-denatured β-lactoglobulin aggregates. J. Food Sci. 73, 23 (2008)
W. Chanasattru, O.G. Jones, E.A. Decker, D.J. McClements, Impact of cosolvents on formation and properties of biopolymer nanoparticles formed by heat treatment of β-lactoglobulin-pectin complexes. Food Hydrocoll. 23, 2450 (2009)
Y. Zhang, H.F. Chan, K.W. Leong, Advanced materials and processing for drug delivery: the past and the future. Adv. Drug Deliv. Rev. 65, 104 (2013)
A. Kumari, S.K. Yadav, S.C. Yadav, Biodegradable polymeric nanoparticles based drug delivery systems. Coll. Surf. B 75, 1 (2010)
J. Essemine, I. Hasni, R. Carpentier, T.J. Thomas, H.A. Tajmir-Riahi, Binding of biogenic and synthetic polyamines to β-lactoglobulin. Int. J. Biol. Macromol. 49, 201 (2011)
L. Liang, V. Tremblay-Hébert, M. Subirade, Characterisation of the β-lactoglobulin/α-tocopherol complex and its impact on α-tocopherol stability. Food Chem. 126, 821 (2011)
A. Taheri-Kafrani, E. Asgari-Mobarakeh, Abdol-Khalegh Bordbar, T. Haertlé, Structure-function relationship of β-lactoglobulin in the presence of dodecyltrimethyl ammonium bromide. Coll. Surf. B 75, 268 (2010)
A. Divsalar, A.A. Saboury, H. Mansoori-Torshizi, A.A. Moosavi-Movahedi, Binding properties of a new anti-TUMOR component (2,2′-bipyridin octylglycinato Pd(II) nitrate) with bovine β-lactoglobulin-A and -B. J. Biomol. Struct. Dyn. 25, 173 (2007)
A. Divsalar, A.A. Saboury, A.A. Moosavi-Movahedi, H. Mansoori-Torshizi, Comparative studies on the interaction between bovine b-lacto globulin type A and B and a new designed Pd(II) complex with anti-tumor activity at different temperatures by spectrophotometric methods. J. Biomol. Struct. Dyn. 26, 586 (2009)
A. Divsalar, A.A. Saboury, R. Yousefi, A.A. Moosavi-Movahedi, H. Mansoori-Torshizi, Spectroscopic and cytotoxic studies of the novel designed palladium (II) complexes: β-Lactoglobulin and K562 as the targets. Int. J. Biol. Macromol. 40, 381 (2007)
A. Divsalar, A.A. Saboury, H. Mansoori-Torshizi, B. Hemmatinejad, Comparative and structural analysis of the interaction between β-lactoglobulin type A and B with a new anticancer component (2,2′-Bipyridin n-hexyl dithiocarbamato Pd(II) nitrate). Bull. Kor. Chem. Soc. 27, 1801 (2006)
E. Gao, C. Liu, M. Zhu, H. Lin, Q. Wu, L. Liu, Current development of Pd(II) complexes as potential antitumor agents. Anti Cancer Agents Med. Chem. 9, 356 (2009)
L. van den Berg, Y. Rosenberg, M.A.J.S. van Boekel, M. Rosenberg, F. van de Velde, Microstructural features of composite whey protein/polysaccharide gels characterized at different length scales. Food Hydrocoll. 23, 1288 (2009)
O.G. Jones, E.A. Decker, D.J. McClements, Formation of biopolymer particles by thermal treatment of β-lactoglobulin-pectin complexes. Food Hydrocoll. 23, 1312–1321 (2009)
O.G. Jones, E.A. Decker, D.J. McClements, Thermal analysis of β-lactoglobulin complexes with pectins or carrageenan for production of stable biopolymer particles. Food Hydrocoll. 24, 239 (2010)
O.G. Jones, U. Lesmes, P. Dubin, D.J. McClements, Effect of polysaccharide charge on formation and properties of biopolymer nanoparticles created by heat treatment of β-lactoglobulin-pectin complexes. Food Hydrocoll. 24, 374 (2010)
S.M.H. Hosseini, Z. Emam-Djomeh, S.H. Razavi, A.A. Moosavi-Movahedi, A.A. Saboury, M.A. Mohammadifar, A. Farahnaky, M.S. Atri, P.V. Meeren, Complex coacervation of β-lactoglobulin–κ-Carrageenan aqueous mixtures as affected by polysaccharide sonication. Food Chem. 141, 215 (2013)
B. Ghalandari, A. Divsalar, A.A. Saboury, T. Haertlé, K. Parivar, R. Bazl, M. Eslami-Moghadam, M. Amanlou, Spectroscopic and theoretical investigation of oxali–palladium interactions with β-lactoglobulin. Spectrochim. Acta. 118, 1038 (2014)
J.C. Souder, W.C. Ellenbogen, Control of d-amphetamine sulphate sustained release capsule. Drug Stand. 26, 77 (1995)
L. Stryer, Intramolecular resonance transfer of energy in proteins. Biochim. Biophys. Acta. 35, 242 (1959)
J. Chakraborty, N. Das, U.C. Halder, Unfolding diminishes fluorescence resonance energy transfer (FRET) of lysine modified b-lactoglobulin: relevance towards anti-HIV binding. J. Photochem. Photobiol. B Biol. 102, 1 (2011)
Y. Sun, S. Wei, C. Yin, L. Liu, C. Hu, Y. Zhao, Y. Ye, X. Hu, J. Fan, Synthesis and spectroscopic characterization of 4-butoxyethoxy-N-octadecyl-1,8-naphthalimide as a new fluorescent probe for the determination of proteins. Bioorg. Med. Chem. Lett. 21, 3798 (2011)
J. Ding, L. Yuan, L. Gao, J. Chen, Fluorescence quenching of a rhodamine derivative: selectively sensing Cu2+ in acidic aqueous media. J. Lumin. 132, 1987 (2012)
M.F. Zhang, Z.Q. Xu, Y.S. Ge, F.L. Jiang, Y. Liu, Binding of fullerol to human serum albumin: spectroscopic and electrochemical approach. J. Photochem. Photobiol. B Biol. 108, 34 (2012)
S. Tabassum, W.M. Al-Asbahy, M. Afzal, F. Arjmand, Synthesis, characterization and interaction studies of copper based drug with human serum albumin (HSA): spectroscopic and molecular docking investigations. J. Photochem. Photobiol. B Biol. 114, 132 (2012)
J. Moue´coucou, C. Villaume, C. Sanchez, L. Me´jean, β-Lactoglobulin/polysaccharide interactions during in vitro gastric and pancreatic hydrolysis assessed in dialysis bags of different molecular weight cut-offs. Biochim. Biophys. Acta. 1670, 105 (2004)
C.M.G.C. Renard, J.F. Thibault, Degradation of pectins in alkaline conditions: kinetics of demethylation. Carbohyd. Res. 286, 139 (1996)
J. Chamani, A.A. Moosavi-Movahedi, O. Rajabi, M. Gharanfoli, M. Momen-Heravi, G.H. Hakimelahi, A. Neamati-Baghsiah, A.R. Varasteh, Cooperative α-helix formation of β-lactoglobulin induced by sodium n-alkyl sulfates. J. Coll. Inter. Sci. 293, 52 (2006)
N. Taulier, T.V. Chalikian, Characterization of pH-induced transitions of β-Lactoglobulin: ultrasonic, densimetric, and spectroscopic studies. J. Mol. Biol. 314, 873 (2001)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghalandari, B., Divsalar, A., Saboury, A.A. et al. β-Lactoglobulin nanoparticle as a chemotherapy agent carrier for oral drug delivery system. J IRAN CHEM SOC 12, 613–619 (2015). https://doi.org/10.1007/s13738-014-0519-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-014-0519-2