Abstract
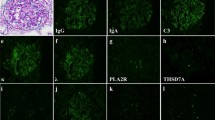
Interferon-beta (IFN-β) subtypes are widely used as immunomodulatory agents for relapsing–remitting multiple sclerosis (MS). Although generally well tolerated, a growing number of reports have recently shown association of long-term IFN-β therapy with several types of glomerulonephritis. Here, we present the case of a 42-year-old woman with MS who developed nephrotic-range proteinuria after taking IFN-β1b for nine years. Initially, due to the presence of histological features consistent with immunoglobulin A (IgA) nephropathy (granular IgA deposits in mesangial lesions), a tonsillectomy plus steroid pulse therapy was performed. However, proteinuria did not significantly decrease after these treatments. Therefore, a second renal biopsy was performed after three years, revealing a membranoproliferative glomerulonephritis-like pattern without immune complex. Further immunofluorescence analysis showed attenuated IgA staining. Consequently, IFN-β1b was replaced with dimethyl fumarate, resulting in complete remission, with proteinuria decreasing to the level of 0.2 g/day. Although it is a rare adverse effect, physicians should pay careful attention to the symptoms and findings of nephritis during the follow-up of patients under treatment with this agent.




Similar content being viewed by others
References
Helen ML, John WS, Michael SD. Shared and distinct functions of type I and type III interferons. Immunity. 2019;50:907–23.
The IFNβ Multiple Sclerosis Study Group. Interferon beta-1b is effective in relapsing-remitting sclerosis. I.Clinical results of a multicenter, randomized, double-blind, placebo-controlled trial. Neurology. 1993;43:655–61.
Jacobs LD, Cookfair DL, Rudick RA, et al. Intramuscular interferon beta-1a for disease progression in relapsing multiple sclerosis. The multiple sclerosis collaborative research group (MSCRG). Ann Neurol. 1996;39:285–94.
Markowitz GS, Nasr SH, Stokes MB, et al. Treatment with IFN-{alpha},-{beta}, or-{gamma} is associated with collapsing focal segmental glomerulosclerosis. Clin J Am Soc Nephrol. 2010;5:607–15.
Raanani P, Ben-Bassat I. Immune-mediated complications during interferon therapy in hematological patients. Acta Haematol. 2002;107:133–44.
Nishio H, Tsukamoto T, Matsubara T, et al. Thrombotic microangiopathy caused by interferon β-1b for multiple sclerosis: a case report. CEN Case Rep. 2016;5:179–83.
Wang Z, Zhang X, Han W, et al. Immune characteristics of renal allograft donors with mesangial IgA deposition. Int Immunopharmacol. 2021;91: 107282.
Sethi S, Fervenza FC. Membranoproliferative glomerulonephritis–a new look at an old entity. N Engl J Med. 2012;366:1119–31.
Sethi S, Fervenza FC. Membranoproliferative glomerulonephritis: pathogenetic heterogeneity and proposal for a new classification. Semin Nephrol. 2011;31:341–8.
Goldberg RJ, Nakagawa T, Johnson RJ, et al. The role of endothelial cell injury in thrombotic microangiopathy. Am J Kidney Dis. 2010;56:1168–74.
Noris M, Remuzzi G. Atypical hemolytic-uremic syndrome. N Engl J Med. 2009;361:1676–87.
Markowitz GS, Nasr SH, Barry S, et al. Treatment with IFN-{alpha}, -{beta}, or -{gamma} is associated with collapsing focal segmental glomerulosclerosis. Clin J Am Soc Nephrol. 2010;5:607–15.
Nakao K, Sugiyama H, Makino E, et al. Minimal change nephrotic syndrome developing during postoperative interferon-beta therapy for malignant melanoma. Nephron. 2002;90:498–500.
Kumasaka R, Nakamura N, Shirato K, et al. Nephrotic syndrome associated with interferon-beta-1b therapy for multiple sclerosis. Clin Exp Nephrol. 2006;10:222–5.
Dressler D, Wright JR, Houghton JB, et al. Another case of focal segmental glomerulosclerosis in an acutely uraemic patient following interferon therapy. Nephrol Dial Transplant. 1999;14:2049–50.
Shah M, Jenis EH, Mookerjee BK, et al. Interferon-alpha-associated focal segmental glomerulosclerosis with massive proteinuria in patients with chronic myeloid leukemia following high dose chemotherapy. Cancer. 1998;83:1938–46.
Mahe J, Meurette A, Moreau A, et al. Renal thrombotic microangiopathy caused by interferon beta-1a treatment for multiple sclerosis. Drug Des Dev Ther. 2013;7:723–8.
Ikeda K, Okamoto T, Yamamura T, et al. Nephrotic syndrome in multiple sclerosis patients who had undergone long-term interferon β-1b therapy. Rinsho Shinkeigaku. 2013;53:19–23.
Wallbach M, Grone HJ, Kitze B, et al. Nephrotic syndrome in a multiple sclerosis patient receiving long-term interferon beta therapy. Am J Kidney Dis. 2013;61:786–9.
Wu WZ, Sun HC, Shen YF, et al. Interferon alpha 2a down-regulate VEGF expression through PI3 kinase and MAP kinase signaling pathways. J Cancer Res Clin Oncol. 2005;131:169–78.
Arrambide G. Thrombotic thrombocytopenic purpura-hemolytic uremic syndrome in relapsing-remitting multiple sclerosis patients on high-dose interferon beta. Mult Scler. 2014;20:1788–9.
Ferrara N. Vascular endothelial growth factor: basic science and clinical progress. Endocr Rev. 2004;25:581–611.
Sidky YA, Borden EC. Inhibition of angiogenesis by interferons: effects on tumor-and lymphocyte-induced vascular responses. Cancer Res. 1987;47:5155–61.
Aaronson DS, Horvath CM. A road map for those who don’t know JAK-STAT. Science. 2002;296:1653–5.
Wu S, Kim C, Baer L, et al. Bevacizumab increases risk for severe proteinuria in cancer patients. J Am Soc Nephrol. 2010;21:1381–9.
Frangie C, Lefaucheur C, Medioni J, et al. Renal thrombotic microangiopathy caused by anti-VEGF-antibody treatment for metastatic renal-cell carcinoma. Lancet Oncol. 2007;8:177–8.
Hanna RM, Lopez E, Wilson J, et al. Minimal change disease onset observed after bevacizumab administration. Clin Kidney J. 2016;9:239–44.
Dauvergne M, Buob D, Rafat C, et al. Renal diseases secondary to interferon-β treatment: a multicentre clinico-pathological study and systematic literature review. Clin Kidney J. 2021;14:2563–72.
Buob D, Decambron M, Gnemmi V, et al. Collapsing glomerulopathy is common in the setting of thrombotic microangiopathy of the native kidney. Kidney Int. 2016;90:1321–31.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Informed consent
Informed consent was obtained from the patient whose case in reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Irifuku, T., Okimoto, K., Masuzawa, N. et al. Nephrotic-range proteinuria and membranoproliferative glomerulonephritis-like pattern caused by interferon-β1b in a patient with multiple sclerosis. CEN Case Rep 12, 275–280 (2023). https://doi.org/10.1007/s13730-022-00745-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13730-022-00745-8