Abstract
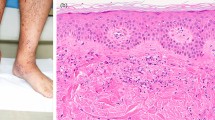
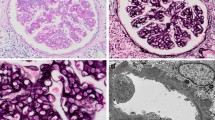
We report the case of an 80 year-old woman who developed bilateral lower extremity purpura and renal impairment with proteinuria a few days after a transient fever (day 0). High levels of both anti-streptolysin-O antibody (ASO) and anti-streptokinase antibody (ASK), as well as low levels of coagulation factor XIII in serum were noted. Skin biopsy was performed and showed a leukocytoclastic vasculitis with deposition of IgA and C3 in the cutaneous small vessels, indicating IgA vasculitis in the skin. After initiation of oral prednisolone, the skin lesions showed significant improvement. However, renal function and proteinuria gradually worsened from day 12. Kidney biopsy was performed on day 29, which demonstrated a necrotizing and crescentic glomerulonephritis with mesangial deposition of IgA and C3. In addition, the deposition of galactose-deficient IgA1 (Gd-IgA1) was positive on glomeruli and cutaneous small vessels, indicating that the purpura and glomerulonephritis both shared the same Gd-IgA1-related pathogenesis. In addition, the association between the acute streptococcal infection and the IgA vasculitis was confirmed by the deposition of nephritis-associated plasmin receptor (NAPlr) in glomeruli. The patient was treated with steroid pulse and intravenous cyclophosphamide, in addition to the oral prednisolone treatment. Renal function and proteinuria gradually improved, but did not completely recover, as is typically seen with courses of IgA vasculitis in the elderly. In this case, the streptococcal infectionrelated IgA vasculitis was confirmed pathologically by the deposition of both NAPlr and Gd-IgA1 in glomeruli, as well as Gd-IgA1 in the cutaneous small vessels.





Similar content being viewed by others
References
Sugino H, Sawada Y, Nakamura M. IgA vasculitis: etiology, treatment, biomarkers and epigenetic changes. Int J Mol Sci. 2021. https://doi.org/10.3390/ijms22147538.
Hastings MC, Rizk DV, Kiryluk K, Nelson R, Zahr RS, Novak J, et al. IgA vasculitis with nephritis: update of pathogenesis with clinical implications. Pediatr Nephrol. 2021. https://doi.org/10.1007/s00467-021-04950-y.
Neufeld M, Molyneux K, Pappelbaum KI, Mayer-Hain S, von Hodenberg C, Ehrchen J, et al. Galactose-deficient IgA1 in skin and serum from patients with skin-limited and systemic IgA vasculitis. J Am Acad Dermatol. 2019;81(5):1078–85. https://doi.org/10.1016/j.jaad.2019.03.029.
Masuda M, Nakanishi K, Yoshizawa N, Iijima K, Yoshikawa N. Group A streptococcal antigen in the glomeruli of children with Henoch-Schönlein nephritis. Am J Kidney Dis. 2003;41(2):366–70. https://doi.org/10.1053/ajkd.2003.50045.
Yumura W, Kobayashi S, Suka M, Hayashi T, Ito S, Nagafuchi H, et al. Assessment of the Birmingham vasculitis activity score in patients with MPO-ANCA-associated vasculitis: sub-analysis from a study by the Japanese Study Group for MPO-ANCA-associated vasculitis. Mod Rheumatol. 2014;24(2):304–9. https://doi.org/10.3109/14397595.2013.854075.
Kim S, Lee NY. Asymptomatic infection by Streptococcus pyogenes in schoolchildren and diagnostic usefulness of antideoxyribonuclease B. J Korean Med Sci. 2005;20(6):938–40. https://doi.org/10.3346/jkms.2005.20.6.938.
Nasr SH, D’Agati VD. IgA-dominant postinfectious glomerulonephritis: a new twist on an old disease. Nephron Clin Pract. 2011;119(1):c18–25. https://doi.org/10.1159/000324180 (discussion c6).
Nasr SH, Fidler ME, Valeri AM, Cornell LD, Sethi S, Zoller A, et al. Postinfectious glomerulonephritis in the elderly. J Am Soc Nephrol. 2011;22(1):187–95. https://doi.org/10.1681/asn.2010060611.
Han W, Suzuki T, Watanabe S, Nakata M, Ichikawa D, Koike J, et al. Galactose-deficient IgA1 and nephritis-associated plasmin receptors as markers for IgA-dominant infection-related glomerulonephritis: a case report. Medicine. 2021;100(5):e24460. https://doi.org/10.1097/MD.0000000000024460
Yamakami K, Yoshizawa N, Wakabayashi K, Takeuchi A, Tadakuma T, Boyle MD. The potential role for nephritis-associated plasmin receptor in acute poststreptococcal glomerulonephritis. Methods. 2000;21(2):185–97. https://doi.org/10.1006/meth.2000.0990.
Oda T, Yoshizawa N, Yamakami K, Sakurai Y, Takechi H, Yamamoto K, et al. The role of nephritis-associated plasmin receptor (NAPlr) in glomerulonephritis associated with Streptococcal infection. J Biomed Biotechnol. 2012;2012: 417675. https://doi.org/10.1155/2012/417675.
Oda T, Yamakami K, Omasu F, Suzuki S, Miura S, Sugisaki T, et al. Glomerular plasmin-like activity in relation to nephritis-associated plasmin receptor in acute poststreptococcal glomerulonephritis. J Am Soc Nephrol. 2005;16(1):247–54. https://doi.org/10.1681/asn.2004040341.
Suzuki H, Yasutake J, Makita Y, Tanbo Y, Yamasaki K, Sofue T, et al. IgA nephropathy and IgA vasculitis with nephritis have a shared feature involving galactose-deficient IgA1-oriented pathogenesis. Kidney Int. 2018;93(3):700–5. https://doi.org/10.1016/j.kint.2017.10.019.
Tang M, Zhang X, Li X, Lei L, Zhang H, Ling C, et al. Serum levels of galactose-deficient IgA1 in Chinese children with IgA nephropathy, IgA vasculitis with nephritis, and IgA vasculitis. Clin Exp Nephrol. 2021;25(1):37–43. https://doi.org/10.1007/s10157-020-01968-8.
Acknowledgements
We thank Mr. Takashi Arai, Ms. Kyoko Wakamatsu, and Ms. Naomi Kuwahara for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Research involving human and animal participation
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from the patient described.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Inoue, T., Takeuchi, K., Ishikawa, A. et al. A case of pathologically confirmed streptococcal infection-related IgA vasculitis with associated glomerulonephritis and leukocytoclastic cutaneous vasculitis. CEN Case Rep 11, 391–396 (2022). https://doi.org/10.1007/s13730-022-00684-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13730-022-00684-4