Abstract
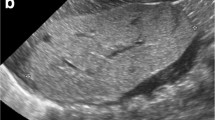
Type IV renal tubular acidosis is known to occur in obstructive uropathy with urinary tract infection. Fanconi syndrome, however, has not been described in these settings. We report two preterm infants who developed Fanconi syndrome associated with hydronephrosis and urinary tract infection. Patient 1 is a boy with 21 trisomy, bilateral renal hypoplasia and bilateral vesicoureteral reflux delivered at 35 weeks’ gestation. At postnatal day 42, he developed Fanconi syndrome after urinary tract infection, which persisted until the surgical correction of vesicoureteral reflux. Patient 2 was delivered at 35 weeks’ gestation. At postnatal day 9, he was admitted for severe dehydration. He had phimosis and ultrasonography showed left pelviectasis. Laboratory data were compatible with Fanconi syndrome, which resolved spontaneously after fluid therapy. Subsequently urine culture grew bacteria and treatment for infection and topical corticosteroid for phimosis were performed. DMSA scintigraphy performed later showed left renal scar. Tubular cell stretch, due to vesicoureteral reflux in Patient 1 and phimosis in Patient 2, and urinary tract infection in association with immaturity of tubules are thought to have caused Fanconi syndrome.
Similar content being viewed by others
References
Sirac C, Bridoux F, Essig M, et al. Toward understanding renal Fanconi syndrome: step by step advances through experimental models. Contrib Nephrol. 2011;169:247–61.
Igarashi T. Pediatric Fanconi syndrome. In: Avner E, Harmon WE, Niaudet P, Yoshikawa N, Emma F, Goldstein SL, editors. Pediatric nephrology. Berlin: Springer-Verlag; 2014.
Bogdanovic R, Stajic N, Putnik J, et al. Transient type 1 pseudo-hypoaldosteronism: report on an eight-patient series and literature review. Pediatr Nephrol. 2009;24:2167–75.
Tanaka H, Nakahata T, Ito R, et al. An infant case of bilateral small kidneys with both proximal and distal tubular dysfunction. Acta Paediatr Jpn. 1998;40:367–9.
Li C, Wang W, Kwon TH, et al. Altered expression of major renal Na transporters in rats with bilateral ureteral obstruction and release of obstruction. Am J Physiol Renal Physiol. 2003;285:F889–901.
Schmidt C, Hocherl K, Schweda F, et al. Regulation of renal sodium transporters during severe inflammation. J Am Soc Nephrol. 2007;18:1072–83.
Li Z, Langhans SA. Transcriptional regulators of Na, K-ATPase subunits. Front Cell Dev Biol. 2015;3:66.
Haycock GB, Aperia A. Salt and the newborn kidney. Pediatr Nephrol. 1991;5:65–70.
Prieur B, Cordeau-Lossouarn L, Rotig A, et al. Perinatal maturation of rat kidney mitochondria. Biochem J. 1995;305(Pt 2):675–80.
Van Why SK, Mann AS, Ardito T, et al. Expression and molecular regulation of Na(+)-K(+)-ATPase after renal ischemia. Am J Physiol. 1994;267:F75–85.
Kwon TH, Frokiaer J, Han JS, et al. Decreased abundance of major Na(+) transporters in kidneys of rats with ischemia-induced acute renal failure. Am J Physiol Renal Physiol. 2000;278:F925–939.
Ghiculescu RA, Kubler PA. Aminoglycoside-associated Fanconi syndrome. Am J Kidney Dis. 2006;48:e89–e93.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Financial disclosures
No honorarium, grant, or other form of payment was given to anyone to produce the manuscript.
Conflict of interest
The authors have declared that no conflict of interest exists.
Ethical standards
This article does not contain any studies with animals performed by any authors.
Informed consent
Informed consent was obtained from all individuals included in the study.
About this article
Cite this article
Tominaga, T., Sato, T., Ichihashi, Y. et al. Transient Fanconi syndrome in two preterm infants with hydronephrosis and urinary tract infection. CEN Case Rep 6, 88–90 (2017). https://doi.org/10.1007/s13730-017-0250-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13730-017-0250-9