Abstract
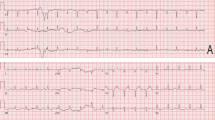
Osimertinib-induced cardiotoxicity is a well-known but rare disorder. An 84-year-old woman was diagnosed with recurrence of lung adenocarcinoma showing an epidermal growth factor receptor mutation of exon 19 deletion, which was initially treated by curative-intent thoracic radiotherapy 4 years prior. She started taking osimertinib (80 mg/day). She had no history of heart disease and showed no signs of cardiac problems. However, 2 months later she presented with symptoms of cardiac failure and QT prolongation on electrocardiogram. Cardiac enzyme levels were not elevated and coronary computed tomography angiography showed no significant stenosis. On admission, sudden-onset torsade de pointes required electrocardioversion. Thus, drug-induced cardiac failure was strongly suspected and we stopped osimertinib therapy. Cardiac function and the electrocardiogram abnormality improved. To our knowledge, this is the third case of coincidence of cardiac failure and QT prolongation and the second case of sudden-onset torsade de pointes associated with osimertinib treatment. In our case, osimertinib-induced cardiac failure with QT prolongation was recovered by stopping the drug treatment. The potential for cardiotoxicity should be considered with osimertinib treatment.



Similar content being viewed by others
References
Anand K, Ensor J, Trachtenberg B et al (2019) Osimertinib-induced cardiotoxicity: a retrospective review of the FDA adverse events reporting system (FAERS). JACC Cardio Oncol 1(2):172–178
Kunimasa K, Kamada R, Oka T et al (2020) Cardiac adverse events in EGFR-mutated non-small cell lung cancer treated with osimertinib. JACC Cardio Oncol 2(1):1–10
Babar T, Blomberg C, Hoffner E et al (2014) Anti-HER2 cancer therapy and cardiotoxicity. Curr Pharm Des 20(30):4911–4919
Balduzzi S, Mantarro S, Guarneri V et al (2014) (2014) Trastuzumab-containing regimens for metastatic breast cancer. Cochrane Database Syst Rev 6:Cd006242
Ewer MS, Patel K, O’Brien D et al (2015) Cardiac safety of afatinib: a review of data from clinical trials. Cardio Oncol 1(1):3
Nuvola G, Dall'Olio FG, Melotti B et al (2019) Cardiac toxicity from afatinib in EGFR-mutated NSCLC: a rare but possible side effect. J Thorac Oncol 14(7):e145–e146
Bian S, Tang X, Lei W (2020) A case of torsades de pointes induced by the third-generation EGFR-TKI, osimertinib combined with moxifloxacin. BMC Pulmonary Med 20(1):1–5
Acknowledgements
We thank Edanz Group (https://en-author-services.edanzgroup.com/ac) for editing a draft of this manuscript.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
For this type of study formal consent is not required.
Informed consent
Informed consent was obtained from the patient.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Ikebe, S., Amiya, R., Minami, S. et al. Osimertinib-induced cardiac failure with QT prolongation and torsade de pointes in a patient with advanced pulmonary adenocarcinoma. Int Canc Conf J 10, 68–71 (2021). https://doi.org/10.1007/s13691-020-00450-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13691-020-00450-2