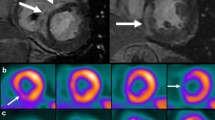
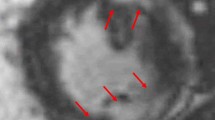
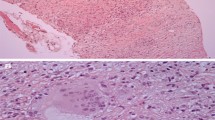
Abstract
Purpose of Review
Sarcoidosis is a systemic inflammatory disorder affecting multiple organ systems, with heterogenous clinical sequelae. Cardiac sarcoidosis (CS) is an underrecognized manifestation with significant clinical implications. Advances in cardiac imaging and new therapeutic options are rapidly changing the clinical approach to CS.
Recent Findings
Cardiac magnetic resonance imaging (CMR), 18Fluorodeoxyglucose positron emission tomography (FDG-PET), and hybrid CMR/FDG-PET imaging have emerged as powerful diagnostic and prognostic tools for CS and provide a means to monitor disease activity. Therapeutic options are similarly changing, with steroid-sparing agents and biologic therapy showing clinical promise, though ongoing clinical trials will potentially provide necessary evidence to definitively guide treatment.
Summary
In this review, we critically evaluate a variety of diagnostic and therapeutic options for CS. Additionally, we propose algorithms for the diagnostic evaluation and treatment of CS, which incorporate current guidelines as well as recent advances in CS.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Rivera NV, et al. High-density genetic mapping identifies new susceptibility variants in sarcoidosis phenotypes and shows genomic-driven phenotypic differences. Am J Respir Crit Care Med. 2016;193(9):1008–22.
Grunewald J, et al. Immunogenetics of disease-causing inflammation in sarcoidosis. Clin Rev Allergy Immunol. 2015;49(1):19–35.
Newman LS, et al. A case control etiologic study of sarcoidosis: environmental and occupational risk factors. Am J Respir Crit Care Med. 2004;170(12):1324–30.
Cozier YC, et al. Sarcoidosis in black women in the United States: data from the Black Women’s Health Study. Chest. 2011;139(1):144–50.
Rybicki BA, et al. Racial differences in sarcoidosis incidence: a 5-year study in a health maintenance organization. Am J Epidemiol. 1997;145(3):234–41.
Arkema EV, Cozier YC. Sarcoidosis epidemiology: recent estimates of incidence, prevalence and risk factors. Curr Opin Pulm Med. 2020;26(5):527–34.
Sève P, et al. Sarcoidosis: a clinical overview from symptoms to diagnosis. Cells. 2021;10(4).
Cacoub P, et al. Cardiac sarcoidosis: a long term follow up study. PLoS One. 2020;15(9):e0238391.
Hamzeh N, et al. Pathophysiology and clinical management of cardiac sarcoidosis. Nat Rev Cardiol. 2015;12(5):278–88.
Uusimaa P, et al. Ventricular tachyarrhythmia as a primary presentation of sarcoidosis. Europace. 2008;10(6):760–6.
•• Birnie DH, et al. HRS expert consensus statement on the diagnosis and management of arrhythmias associated with cardiac sarcoidosis. Heart Rhythm. 2014;11(7):305–23. HRS Guidelines for cardiac sarcoidosis.
Webb M, et al. Cardiac involvement in sarcoidosis deaths in Wayne County, Michigan: a 20-year retrospective study. Acad Forensic Pathol. 2018;8(3):718–28.
Iwai K, et al. Pathological studies on sarcoidosis autopsy. I. Epidemiological features of 320 cases in Japan. Acta Pathol Jpn. 1993;43(7–8):372–6.
Matsui Y, et al. Clinicopathological study of fatal myocardial sarcoidosis. Ann N Y Acad Sci. 1976;278:455–69.
Iwai K, et al. Racial difference in cardiac sarcoidosis incidence observed at autopsy. Sarcoidosis. 1994;11(1):26–31.
Okada DR, et al. Isolated cardiac sarcoidosis: a focused review of an under-recognized entity. J Nucl Cardiol. 2018;25(4):1136–46.
•• Terasaki F, et al. JCS 2016 guideline on diagnosis and treatment of cardiac sarcoidosis - digest version. Circ J, 2019;83(11):2329–2388. JCS Guidelines for cardiac sarcoidosis.
Judson MA, et al. The WASOG Sarcoidosis Organ Assessment Instrument: an update of a previous clinical tool. Sarcoidosis Vasc Diffuse Lung Dis. 2014;31(1):19–27.
Nagao S, et al. Electrocardiographic abnormalities and risk of developing cardiac events in extracardiac sarcoidosis. Int J Cardiol. 2015;189:1–5.
Crouser ED, et al. Diagnosis and detection of sarcoidosis. An Official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;201(8):e26–e51.
Ohira H, et al. Underdiagnosis of cardiac sarcoidosis by ECG and echocardiography in cases of extracardiac sarcoidosis. ERJ Open Res. 2022;8(2).
Kurmann R, Mankad SV, Mankad R. Echocardiography in Sarcoidosis. Curr Cardiol Rep. 2018;20(11):118.
Darlington P, et al. Diagnostic approach for cardiac involvement in sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2019;36(1):11–7.
Di Stefano C, et al. Diagnostic and predictive value of speckle tracking echocardiography in cardiac sarcoidosis. BMC Cardiovasc Disord. 2020;20(1):21.
Schouver ED, et al. Early detection of cardiac involvement in sarcoidosis with 2-dimensional speckle-tracking echocardiography. Int J Cardiol. 2017;227:711–6.
Bera D, et al. Serum angiotensin converting enzyme, erythrocyte sedimentation rate and high sensitive-C reactive protein levels in diagnosis of cardiac sarcoidosis- where do we stand? Indian Pacing Electrophysiol J. 2020;20(5):184–8.
Handa T, et al. Significance of plasma NT-proBNP levels as a biomarker in the assessment of cardiac involvement and pulmonary hypertension in patients with sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2010;27(1):27–35.
Kiko T, et al. A multiple biomarker approach in patients with cardiac sarcoidosis. Int Heart J. 2018;59(5):996–1001.
Kobayashi Y, et al. Association of high serum soluble interleukin 2 receptor levels with risk of adverse events in cardiac sarcoidosis. ESC Heart Fail. 2021;8(6):5282–92.
Kolluri N, et al. Routine laboratory biomarkers as prognostic indicators of cardiac sarcoidosis outcomes. Sarcoidosis Vasc Diffuse Lung Dis. 2022;39(3):e2022023.
Markatis E, et al. Cardiac sarcoidosis: diagnosis and management. Rev Cardiovasc Med. 2020;21(3):321–38.
Freeman AM, et al. Predictors of cardiac sarcoidosis using commonly available cardiac studies. Am J Cardiol. 2013;112(2):280–5.
Mehta D, et al. Cardiac involvement in patients with sarcoidosis: diagnostic and prognostic value of outpatient testing. Chest. 2008;133(6):1426–35.
Padala SK, et al. Impact of early initiation of corticosteroid therapy on cardiac function and rhythm in patients with cardiac sarcoidosis. Int J Cardiol. 2017;227:565–70.
Elwazir MY, Bois JP, Chareonthaitawee P. Utilization of cardiac imaging in sarcoidosis. Expert Rev Cardiovasc Ther. 2022;20(4):253–66.
Smedema JP, et al. Evaluation of the accuracy of gadolinium-enhanced cardiovascular magnetic resonance in the diagnosis of cardiac sarcoidosis. J Am Coll Cardiol. 2005;45(10):1683–90.
•• Greulich S, et al. Hybrid cardiac magnetic resonance/fluorodeoxyglucose positron emission tomography to differentiate active from chronic cardiac sarcoidosis. JACC Cardiovasc Imaging. 2022;15(3):445–456. Study showing diagnostic utility in hybrid CMR/FDG-PET in evaluation of cardiac sarcoidosis.
Aitken M, et al. Diagnostic accuracy of cardiac MRI versus FDG PET for cardiac sarcoidosis: a systematic review and meta-analysis. Radiology. 2022;304(3):566–79.
Greulich S, et al. CMR imaging predicts death and other adverse events in suspected cardiac sarcoidosis. JACC Cardiovasc Imaging. 2013;6(4):501–11.
Yoshida A, et al. Direct comparison of the diagnostic capability of cardiac magnetic resonance and endomyocardial biopsy in patients with heart failure. Eur J Heart Fail. 2013;15(2):166–75.
Kouranos V, et al. Complementary role of CMR to conventional screening in the diagnosis and prognosis of cardiac sarcoidosis. JACC Cardiovasc Imaging. 2017;10(12):1437–47.
Sobol I, et al. Assessment of unexplained cardiomyopathy: clinical utility of delayed-enhancement cardiac magnetic resonance compared to endomyocardial biopsy. J Am Coll Cardiol. 2012;59(13, Supplement):E1553.
Flamée L, et al. Prognostic value of cardiovascular magnetic resonance in patients with biopsy-proven systemic sarcoidosis. Eur Radiol. 2020;30(7):3702–10.
Coleman GC, et al. Prognostic value of myocardial scarring on CMR in patients with cardiac sarcoidosis. JACC Cardiovasc Imaging. 2017;10(4):411–20.
Hulten E, et al. Presence of late gadolinium enhancement by cardiac magnetic resonance among patients with suspected cardiac sarcoidosis is associated with adverse cardiovascular prognosis: a systematic review and meta-analysis. Circ Cardiovasc Imaging. 2016;9(9):e005001.
Crouser ED, et al. Improved detection of cardiac sarcoidosis using magnetic resonance with myocardial T2 mapping. Am J Respir Crit Care Med. 2014;189(1):109–12.
Greulich S, et al. Comprehensive cardiovascular magnetic resonance assessment in patients with sarcoidosis and preserved left ventricular ejection fraction. Circ Cardiovasc Imaging. 2016;9(11).
Dill T. Contraindications to magnetic resonance imaging. Heart. 2008;94(7):943–8.
Sobic-Saranovic D, Artiko V, Obradovic V. FDG PET imaging in sarcoidosis. Semin Nucl Med. 2013;43(6):404–11.
Soussan M, et al. Functional imaging in extrapulmonary sarcoidosis: FDG-PET/CT and MR features. Clin Nucl Med. 2014;39(2):e146–59.
Agrawal T, et al. Diagnosis of cardiac sarcoidosis: a primer for non-imagers. Heart Fail Rev. 2022;27(4):1223–33.
Kim SJ, Pak K, Kim K. Diagnostic performance of F-18 FDG PET for detection of cardiac sarcoidosis; a systematic review and meta-analysis. J Nucl Cardiol. 2020;27(6):2103–15.
Youssef G, et al. The use of 18F-FDG PET in the diagnosis of cardiac sarcoidosis: a systematic review and metaanalysis including the Ontario experience. J Nucl Med. 2012;53(2):241–8.
Bekki M, et al. Localization of myocardial FDG uptake for prognostic risk stratification in corticosteroid-naïve cardiac sarcoidosis. J Nucl Cardiol. 2022;29(5):2132–44.
Ning N, et al. Serial cardiac FDG-PET for the diagnosis and therapeutic guidance of patients with cardiac sarcoidosis. J Card Fail. 2019;25(4):307–11.
Coulden RA, et al. Utility of FDG PET and cardiac MRI in diagnosis and monitoring of immunosuppressive treatment in cardiac sarcoidosis. Radiol Cardiothorac Imaging. 2020;2(4):e190140.
Saric P, et al. PET imaging in cardiac sarcoidosis: a narrative review with focus on novel PET tracers. Pharmaceuticals (Basel). 2021;14(12).
Christopoulos G, et al. Suppressing physiologic 18-fluorodeoxyglucose uptake in patients undergoing positron emission tomography for cardiac sarcoidosis: the effect of a structured patient preparation protocol. J Nucl Cardiol. 2021;28(2):661–71.
Osborne MT, et al. Patient preparation for cardiac fluorine-18 fluorodeoxyglucose positron emission tomography imaging of inflammation. J Nucl Cardiol. 2017;24(1):86–99.
Quinn B, et al. Radiation dosimetry of 18F-FDG PET/CT: incorporating exam-specific parameters in dose estimates. BMC Med Imaging. 2016;16(1):41.
Divakaran S, et al. Diagnostic accuracy of advanced imaging in cardiac sarcoidosis. Circ Cardiovasc Imaging. 2019;12(6):e008975.
Dweck MR, et al. Hybrid magnetic resonance imaging and positron emission tomography with fluorodeoxyglucose to diagnose active cardiac sarcoidosis. JACC Cardiovasc Imaging. 2018;11(1):94–107.
Trivieri MG, et al. Challenges in cardiac and pulmonary sarcoidosis: JACC state-of-the-art review. J Am Coll Cardiol. 2020;76(16):1878–901.
Ramirez R, et al. Advanced imaging in cardiac sarcoidosis. J Nucl Med. 2019;60(7):892–8.
Subramanian K, et al. Access to cardiac PET/CT by sarcoidosis patients and cost-effectiveness analysis of cardiac PET/MR compared to the standard of care. Clin Imaging. 2023;94:50–5.
Ezzeddine FM, et al. Electrogram-guided endomyocardial biopsy yield in patients with suspected cardiac sarcoidosis and relation to outcomes. J Cardiovasc Electrophysiol. 2021;32(9):2486–95.
Yilmaz A, et al. Comparative evaluation of left and right ventricular endomyocardial biopsy: differences in complication rate and diagnostic performance. Circulation. 2010;122(9):900–9.
Rosario KF, et al. Cardiac sarcoidosis: current approaches to diagnosis and management. Curr Allergy Asthma Rep. 2022;22(12):171–82.
Fazelpour S, et al. Corticosteroid and immunosuppressant therapy for cardiac sarcoidosis: a systematic review. J Am Heart Assoc. 2021;10(17).e021183.
Stievenart J, et al. Cardiac sarcoidosis: systematic review of the literature on corticosteroid and immunosuppressive therapies. Eur Respir J. 2022;59(5).
Subramanian M, et al. Pre-treatment myocardial (18)FDG uptake predicts response to immunosuppression in patients with cardiac sarcoidosis. JACC Cardiovasc Imaging. 2021;14(10):2008–16.
Yazaki Y, et al. Prognostic determinants of long-term survival in Japanese patients with cardiac sarcoidosis treated with prednisone. Am J Cardiol. 2001;88(9):1006–10.
Griffin JM, et al. Management of cardiac sarcoidosis using mycophenolate mofetil as a steroid-sparing agent. J Card Fail. 2021;27(12):1348–58.
Gallegos C, et al. Non-steroidal treatment of cardiac sarcoidosis: a systematic review. Int J Cardiol Heart Vasc. 2021;34:100782.
•• Vis R, et al. Prednisone vs methotrexate in treatment naïve cardiac sarcoidosis. J Nucl Cardiol. 2023. Study comparing prednisone, methotrexate, or combination therapy showing effective suppression of FDG-PET imaging in all groups with no significant difference in clinical outcomes. Supports use of prednisone plus methotrexate or methotrexate monotherapy in treatment of CS.
Nagai S, et al. Treatment with methotrexate and low-dose corticosteroids in sarcoidosis patients with cardiac lesions. Intern Med. 2014;53(23):2761.
•• Birnie D, et al. Cardiac sarcoidosis multi-center randomized controlled trial (CHASM CS- RCT). Am Heart J. 2020;220:246–252. Ongoing multicenter randomized clinical trial comparing prednisone and prednisone/methotrexate combination therapy as initial treatment in CS.
Page RL 2nd, et al. Drugs that may cause or exacerbate heart failure: a scientific statement from the American Heart Association. Circulation. 2016;134(6):e32–69.
Bakker ALM, et al. Effectiveness and safety of infliximab in cardiac sarcoidosis. Int J Cardiol. 2021;330:179–85.
Judson MA, et al. Outcomes of prednisone-tapering regimens for cardiac sarcoidosis: a retrospective analysis demonstrating a benefit of infliximab. Respir Med. 2022;203:107004.
Gilotra NA, et al. Clinical and imaging response to tumor necrosis factor alpha inhibitors in treatment of cardiac sarcoidosis: a multicenter experience. J Card Fail. 2021;27(1):83–91.
•• Baker MC, et al. TNF-alpha inhibition for the treatment of cardiac sarcoidosis. Semin Arthritis Rheum. 2020;50(3):546–552. Retrospective study showing effective treatment of CS with TNF-alpha inhibitors with good cardiac safety profile.
Krause ML, et al. Successful use of rituximab in refractory cardiac sarcoidosis. Rheumatology (Oxford). 2016;55(1):189–91.
Elwazir M, et al. Rituximab for the treatment of refractory cardiac sarcoidosis: a single-center experience. J Card Fail. 2022;28(2):247–58.
Sekhri V, et al. Cardiac sarcoidosis: a comprehensive review. Arch Med Sci. 2011;7(4):546–54.
Sadek MM, et al. Corticosteroid therapy for cardiac sarcoidosis: a systematic review. Can J Cardiol. 2013;29(9):1034–41.
Takaya Y, et al. Outcomes in patients with high-degree atrioventricular block as the initial manifestation of cardiac sarcoidosis. Am J Cardiol. 2015;115(4):505–9.
Al-Khatib SM, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2018;15(10):e190–252.
Desai R, et al. The burden of cardiac arrhythmias in sarcoidosis: a population-based inpatient analysis. Ann Transl Med. 2018;6(17):330.
Viles-Gonzalez JF, et al. Supraventricular arrhythmias in patients with cardiac sarcoidosis prevalence, predictors, and clinical implications. Chest. 2013;143(4):1085–90.
Locke AH, et al. Arrhythmia in cardiac sarcoidosis. Cardiol Rev. 2021;29(3):131–42.
Muser D, et al. Characterization of the electroanatomic substrate in cardiac sarcoidosis: correlation with imaging findings of scar and inflammation. JACC Clin Electrophysiol. 2018;4(3):291–303.
Weng W, et al. Atrial arrhythmias in clinically manifest cardiac sarcoidosis: incidence, burden, predictors, and outcomes. J Am Heart Assoc. 2020;9(17):e017086.
Zipse MM, Sauer WH. Cardiac sarcoidosis and consequent arrhythmias. Card Electrophysiol Clin. 2015;7(2):235–49.
Yodogawa K, et al. Effect of corticosteroid therapy on ventricular arrhythmias in patients with cardiac sarcoidosis. Ann Noninvasive Electrocardiol. 2011;16(2):140–7.
Adhaduk M, et al. The role of electrophysiology study in risk stratification of cardiac sarcoidosis patients: meta-analyses and systemic review. Int J Cardiol. 2022;349:55–61.
Kron J, et al. Efficacy and safety of implantable cardiac defibrillators for treatment of ventricular arrhythmias in patients with cardiac sarcoidosis. Europace. 2013;15(3):347–54.
AHA/ACC/HFSA Guideline for the Management of Heart Failure. J Card Fail. 2022;28(5):e1–167.
Birnie DH, et al. Cardiac sarcoidosis. J Am Coll Cardiol. 2016;68(4):411–21.
Rosenthal DG, et al. Long-term corticosteroid-sparing immunosuppression for cardiac sarcoidosis. J Am Heart Assoc. 2019;8(18):e010952.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shaver, A., Schwartz, A., Bhatt, K. et al. Diagnostic Approach and Management of Cardiac Sarcoidosis. Curr Pulmonol Rep 12, 70–79 (2023). https://doi.org/10.1007/s13665-023-00309-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13665-023-00309-w