Abstract
De novo domestication has received recent attention because of the potential to produce new crop species with additional agroecosystem functions and useful products for climate-resilient agricultural systems of the future. However, there are often traits in wild species that make them difficult to domesticate. One key domestication trait selected by early farmers and modern plant breeders in many crops is the ability to self-pollinate. Benefits include higher seed set and more reliable seed production, as well as more efficient selection during breeding because it allows for unmasking of recessive traits and enforcement of favorable gene interactions. Similarly, interspecific hybridization has been used to add to the genetic diversity of many crop species. We evaluated self-pollination and interspecific hybridization in Silphium integrifolium and S. perfoliatum to learn the extent of possible gene flow between the species and its usefulness in cultivar development and the potential for fixation of favorable domestication genes by self-pollination. Our results indicate that Silphium interspecific hybrids can be easily developed, potentially facilitating movement of otherwise invariable traits from one species to another. Further, we showed, for the first time, that self-pollination is also possible in both species and their interspecific hybrids, but the rate varies among genotypes. This has profound implications in optimizing plant breeding methods for the study and improvement of these species and adds more evidence to our understanding of mating systems in Asteraceae, an underutilized family of plants with great potential for additional domesticated species.
Similar content being viewed by others
1 Introduction
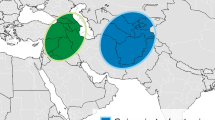
While many crops were long ago domesticated by humans, interest remains in the scientific community to domesticate additional species to fit specific agricultural niches, sustainability objectives, or functions to benefit humanity (reviewed in DeHaan et al. 2016). In particular, domestication of perennial species into grain crops has been proposed to reduce or reverse negative environmental impacts of large-scale agriculture. Compared with annual crops, perennials have the potential to reduce soil erosion and better manage water and nutrient use, preventing losses or excesses. Carbon sequestration is increased by taking advantage of a longer growing season each year, which may increase biomass accumulation (Glover et al. 2010). While perennial options currently exist for some crop niches, such as fruits, there are very few perennial alternatives to the annual cereal, oilseed, and pulse grain crops that form the foundation of modern food systems. Two contribal wild relatives of the domesticated sunflower (Helianthus annuus L.), cup plant (Silphium perfoliatum L.), and whole-leaf rosinweed (Silphium integrifolium Michx.) have been identified as potential candidates for domestication to provide edible oils and other products (Fig. 1; van Tassel et al. 2017). However, for it to be a successful crop in the near future, breeders need to rapidly improve yield, grain/seed quality, and fitness for managed environments while maintaining the important ecosystem services or other novel functions that inspired breeders’ interest in the first place. At least at first, this will need to be accomplished without the well-developed scientific tools and community resources available to high-performing annual crops.
Photographs of research plots consisting of cup plant (Silphium perfoliatum L.; left panel) and whole-leaf rosinweed (Silphium integrifolium Michx.; right panel). S. perfoliatum is a generally lodging resistant, rust disease resistant, high biomass species with opposite leaves that fuse into a cup that often retains water. S. integrifolium, of the two species, tends to produce more seeds and less leaf material. Both species have interesting traits that are unique but largely invariable within them
Both S. integrifolium and S. perfoliatum are perennial members of the Heliantheae (sunflower) tribe of the Asteraceae family. They are native to the prairies of North America, from the Rocky Mountains eastward to Appalachia and from Canada to the Gulf of Mexico (USDA-NRCS 2019). S. perfoliatum has been cultivated on a limited basis as a forage crop since at least 1957 and more recently has been explored as a bioenergy crop (Zilverberg et al. 2016). Work on S. integrifolium was initiated in the early 2000s independently in Poland for biomass production and Kansas for oilseed production (Kowalski and Wiercinski 2004; van Tassel et al. 2014). Oil quality and quantity characteristics in Silphium vary considerably among wild accessions, but some plant collections are close to wild sunflower in composition and oil content (Reinert et al. 2019). At this time, well-established breeding systems have not been developed for Silphium, and most breeding efforts rely on half-sib-based evaluation because of its well-known tendency to outcross. Unfortunately, half-sib-based evaluation only provides a weak estimate of additive genetic variance, thereby diminishing trait heritability and, on a practical level, limiting the potential of genetic progress from selection (reviewed in Hallauer 2007).
One of the key traits that would immediately improve yield as well as breeding efficiency is the ability to self-pollinate. Yield is improved because selfing ability provides reproductive assurance (Eckert et al. 2010) in the absence of adequate pollinators, which is already a problem and expected to become increasingly limiting (Nazzi and Pennacchio 2014). Additionally, self-compatibility allows intentional inbreeding, increasing a breeder’s ability to produce consistent genetic lines, purge deleterious alleles, and identify the genetic basis of valuable traits (Muñoz-Sanz et al. 2020). These features all improve the efficiency and effectiveness of modern breeding.
Because of these clear benefits, knowledge of self-pollination and outcrossing ability is considered one of the key reproductive biology traits to factor into the decision to domesticate a new species (DeHaan et al. 2016). Flexibility in cross-pollination and self-pollination has dictated the crop improvement trajectory of many major crops, as illustrated by the classic example of maize (Zea mays L.). The transition from open-pollinated maize (with near zero yearly genetic gain in yield) to the single-cross hybrid system that provided circa 120 kg ha−1 y−1 genetic gain, happened over the course of about 20 years and is one of the great success stories in scientific breeding. It transformed maize as a crop. This was only possible because the species was able to both self-pollinate and cross-pollinate with ease and predictability, as needed by the breeder (Crow 1998). Genetically uniform inbred lines resulted in uniform hybrids, and both quantitative and single gene traits were more efficiently maintained in inbred lines and more efficiently evaluated as targets of selection in either inbred lines (pedigree selection) or hybrids (testcross selection), compared with the half-sib or mass selection methods of the past (Hallauer 2007). Many herbaceous perennials that are of interest for grain/seed production at this time have either near complete self-incompatibility or, in the case of Silphium, are presumed to be self-incompatible (van Tassel and DeHaan 2013), which limits the ability of the breeder to capture and maintain rare gene variants and traits and efficiently exercise selection, limiting genetic gain (Hallauer 2007). However, it is known that wild plant species exist on a spectrum of near complete self-compatibility to near complete self-incompatibility, with some species exhibiting variability among populations (Whitehead et al. 2018). Genetic gain in Silphium would be limited versus competing hybrid annual crops, such as maize or sunflower, if self-incompatibility is a strong characteristic in the genus.
Self-incompatibility is the inability of a healthy plant to develop seeds when self-pollinated. In self-incompatible systems, stigmas can discriminate between pollen grains which are genetically similar or dissimilar to the flower and prevent fertilization by similar pollen (Silva and Goring 2001). While gametophytic self-incompatibility is the most common form in flowering plants, identified in approximately 60 different families (Kao and McCubbin 1996), only the sporophytic system has ever been observed in the Asteraceae (Gonthier et al. 2013). A further constraint on self-pollination in Silphium, which can be overcome easily by physical intervention of a pollinator, is the spatial separation between the female florets (arising from the ray petals) and the male florets (arising from the disk florets; Fig. 2).
The components of a Silphium flower, illustrated by a color photograph. The capitulum is denoted by (A), the ligule or ray petal floret by (B), the stigma by (C), and the stamen by (D). Note the separation of stigma and stamen to different florets, which is a different arrangement from the better-known contribal species sunflower (Helianthus annuus), in which stigma and stamen occur on the same floret. The arrangement on Silphium has implications for self- and cross-pollination, which is important for understanding its population biology and potential for crop development through breeding
A second key consideration for domesticating a new species is availability of genetic resources (DeHaan et al. 2016). Maximizing functional genetic diversity of the base population of a new crop through exploration of secondary gene pools, such as congeneric species, allows for adaptation of a new crop to managed systems. For example, modern sunflower is the result of multiple introgressions of new functional diversity from wild congenerics, which increased gene number in the domesticate by 1.5% and resulted in improved domestication traits (Hübner et al. 2019). Both Silphium species display characteristics which could be combined to make an interspecific hybrid an excellent option for a final domesticated cultivar or as a source of genes for introgression from one species to the other. S. perfoliatum and S. integrifolium can successfully hybridize, but casual observation of these hybrids suggests reduced fertility (Jessee and Yatskievych 2011; Van Tassel et al. 2017). This is most likely due to chromosome structural differences between parents of each species, as interspecific hybrids form chromatin bridges during meiosis (Settle and Fisher 1972). Cross-incompatibility can be either unilateral or bilateral, opening the possibility that some species may be usable as female but not male parents or vice versa.
The study of self-incompatibility and interspecies hybrids in Silphium can better inform the next steps and speed of domestication within the genus, as well as for other Heliantheae and Asteraceae species. Sunflower, for instance, has benefitted from interspecific gene introgression and a flexible reproductive biology (i.e., it can self- and cross-pollinate easily). Our goal is to understand the limitations of a comparison of Silphium to Helianthus, in terms of self-pollination and interspecific crosses, and develop approaches based on this knowledge that would facilitate modern breeding of Silphium.
2 Materials and methods
2.1 Germplasm
Experiments were conducted on the Prosper, ND, worksite of the North Dakota Agricultural Experiment Station, North Dakota State University, and the St. Paul campus of the Minnesota Agricultural Experiment Station, University of Minnesota. All plant material was obtained from The Land Institute breeding program (Table 1). This program was initiated from wild collections of S. integrifolium and S. perfoliatum from across the United States, and the S. integrifolium populations used in this study have undergone no more than 4 cycles of selection for presumptive domestication traits, primarily to increase the number of female florets per capitulum. Of these, families 7 and 49 are each full-sibling families with half-sibling parents, while the remaining half-sibling families and other stocks are not known to have undergone any inbreeding. Estimations of inbreeding and relatedness were based solely on pedigree records.
2.2 Pollen viability and germination experiments
Pollen from two S. integrifolium genotypes, two S. perfoliatum genotypes, and two S. integrifolium x S. perfoliatum hybrid genotypes were assessed for viability and germination rate. Pollen viability was estimated using the Alexander staining technique (Alexander 1969). Pollen grains were placed on a microscopic slide and one drop of Alexander stain buffer (95% ethanol, 10 ml; Malachite green (1% in 95% ethanol), 1 ml; Fuchsin acid (1% in water), 5 ml; Orange G (1% in water), 0.5 ml; phenol, 5 g; chloral hydrate, 5 g; glacial acetic acid, 2 ml; glycerol, 25 ml; and distilled water, 50 ml was added. The stained pollen grains were incubated for 1 h at 25– 27 °C. Stained pollen grains were observed with a Carl Zeiss™Axio Lab™ A1 Upright Laboratory Microscope with a top-mounted AxioCam Erc 5 s camera and 10x magnification. Pollen grains stained dark (dark blue or purple) were counted as viable.
To assess pollen germination rate, capitula were collected from plants during mid- to late anthesis. Petals and bracts were separated, and anthers and stigmas were isolated from capitula. Pollen grains and stigmas were stored on ice in small glass vials. The stigmas were ground using a mortar and pestle to generate stigma extract. The Silphium pollen growth media (PGM) was prepared according to the Keshava Murthy and colleagues (Keshava Murthy et al. 1994) protocol for sunflower PGM production. First, stigma extract was made by grinding two fresh stigmas of each genotype in 100 μl PGM stock solution at room temperature for approximately 120 s. Fifty microliters of the extract were added to an aqueous mixture of sucrose (150 g L−1), boric acid (200 mg L−1), potassium nitrate (200 mg L−1), magnesium sulphate (200 mg L−1), calcium nitrate (200 mg L−1), and PEG 6000 (223.6 g L−1). Stigma extract was added to the PGM because it has been shown to increase in vitro pollen germination rate. Seven different PGMs were prepared: one each containing stigma extract from the six Silphium genotypes and one with no stigma extract as control.
An aliquot of 80 uL of PGM was dropped on the microscope slide and pollen grains were sprinkled on to the PGM. Samples were incubated overnight in darkness at 25–27 °C. Pollen tube growth was observed with a Carl Zeiss™Axio Lab™ A1 Upright Laboratory Microscope with a top mounted AxioCam Erc 5 s camera and 10x magnification. We considered a pollen grain germinated when the pollen tube was at least equivalent to the diameter of the pollen grain. Germination rate in percentage was determined by dividing the number of germinated pollen grains by the total number of counted pollen grains. Three slides were observed per treatment combination.
2.3 Self- and cross-pollination experiments
The remaining experiments required manual pollination of capitula. Female-designated capitula were covered with nylon or cotton bags before bloom to prevent pollination. Pollen donor capitula were covered at least a day prior to pollen collection to ensure that pollen would be available. Pollination was carried out when stigmas were visible and mature, namely, when the stigma branches are opened laterally. The designated females were prepared for crossing by cutting back ligules (ray petals) to better expose stigma. Figure 2 details the anatomy of a Silphium capitulum. Pollen was collected either by isolating male florets from pollen donors and storing them in glass vials or gently tapping pollen from donor capitula into plastic containers. Female flowers were either pollinated with four to six male florets directly by dusting pollen on stigmas or by brushing pollen on to stigma using wire and polyester pipe cleaners. After pollination, capitula were again covered with nylon or cotton bags to prevent uncontrolled pollination or loss due to shattering, and harvested after senescence. The number of female florets and the number of developed seeds were recorded for each harvested capitulum. Developed seeds are easily differentiated from undeveloped seeds by visual assessment. The rate at which a capitulum produced developed seeds (referred to as “seed set”) was determined by dividing the number of developed seeds in a capitulum by the total number of female florets in the capitulum (quantified by the sum of empty and filled achenes).
To quantify the ability of S. perfoliatum, S. integrifolium, and their interspecific hybrids to intermate, a full diallel cross of the six individual genotypes listed in Table 1 was conducted. This was replicated in two field seasons. Following the previously described crossing method, capitula on each plant were pollinated in triplicate with each of the five other genotypes, for a total of fifteen pollinated capitula on each plant.
In each of two field seasons, 2 to 3 capitula on each of the six single plants listed in Table 1 were self-pollinated to infer repeatability of self-pollinated seed set. To further inform the expected range of self-pollination in Silphium germplasm, active self-pollination treatments were also assigned to plants in each of the breeding families listed in Table 1. Within the breeding families, only one capitulum was used on a given plant within a family.
To assess self-pollination in the absence of a pollinator, breeding family genotypes were passively self-pollinated on different flowers from the pollinations described above and compared with the active self-pollination described in the previous paragraph. In passive self-pollination, cotton bags were placed over the capitulum prior to anthesis and left until senescence. Heads were harvested, threshed, and seed set quantified, as above.
Additionally, we assessed open-pollinated seed set of S. perfoliatum, S. integrifolium, and their interspecific hybrids by harvesting mature, open-pollinated heads from the plots of the six individual genotypes listed in Table 1. Heads were threshed individually, and seed set assessed by digital x-ray imaging. A total of 16 heads per genotype was evaluated.
2.4 Statistical analysis
We performed ANOVA with sums of squares calculated sequentially (Type 1 SS) using the “aov” function in statistical software R, version 3.4.3 (https://www.r-project.org/) in order to determine the source of differences in pollen viability, in vitro germination among different pollen-stigma combinations, the number of female florets, and seed set. Effects for female species, male species, and year were assessed as fixed effects. Genotypes within the female and male groups were also tested as nested factors. Fisher’s Least Significant Differences were used to calculate differences in seed set and floret number in crossing experiments, and t tests were used to test reciprocal effects and treatment levels to the control in the in vitro experiments.
In cases where pairwise data were non-normally distributed, a Mann-Whitney U test was used to identify differences between two-level factors. This analysis was conducted using the “dplyr” and “stats” packages of R v. 3.4.2.
Analysis of open-pollinated seed set on parent stocks and interspecific crosses was conducted using Proc Mixed of SAS v. 9.4 (https://www.sas.com/). Fixed effects included replication and genotype, with random effects of whole plot error and plant-to-plant variation within plots. Fixed effects were tested using the whole plot error effect as the denominator term in the F test.
3 Results and discussion
3.1 Assessment of inter-species cross compatibility
The evaluation of pollen viability by Alexander staining revealed a range of viable pollen among the tested genotypes and hybrids from 85 to 100%, with hybrid genotype 390 × 54 showing the lowest rate of pollen viability, and the other five tested genotypes all having pollen viability above 98% (data not shown).
With respect to in vitro pollen tube germination, the analysis of variance revealed significant stigma and pollen species effects as well as stigma and pollen genotype nested within species (p < 0.001 for all). An interaction between specific genotypes within each species was significant (p < 0.001), but this did not extend to the species level (Table 2). The pollen germination rate ranged from 0.0 (54x[390 × 54]) to 40.0% (54 × 2130). Pollen germination followed the trend from Alexander staining of 390 × 54 having the poorest pollen viability. The interspecific hybrids had significantly lower pollen germination when used as a stigma extract and as the source of pollen. However, in half of the treatments with interspecific hybrid stigma extract, pollen germination was significantly enhanced compared with the control media with no stigma extract. In addition, the use of stigma extract from S. perfoliatum to germinate S. integrifolium pollen, and vice versa, led to significantly higher pollen germination compared with the control, with only one exception, indicating floral physiology was conducive to cross pollination among species (Table 3).
We found significant differences among species and genotypes within species for cross seed set on the female side (p < 0.001), but not on the male side. However, specific pairs of species and genotypes within species were more fertile than others (p < 0.001; Tables 4 and 2). Across all observed crosses, the seed set varied from 6.1 ([390 × 2130]×54) to 95.2% (2130 × 54) (Table 4). There is no evidence of either unilateral or bilateral cross-incompatibility, as interspecific crosses in both directions successfully produced many seeds. The means for each species as a female were 17.0 (hybrid), 55.6 (S. integrifolium), and 63.6% (S. perfoliatum) (Table 4), suggesting lower seed set when hybrids are used as a female compared with either parent. In general, these results mirror the in vitro pollen experiment (Table 3). To further verify the result of lower female fertility in the interspecific hybrids, we looked at open-pollinated seed set on the same plants, in the presence of ample pollinator activity at bloom (Table 5). While total female floret number showed mid-parent values in the interspecies hybrids, there was significantly lower open-pollinated seed set on the interspecific hybrids relative to the parent stocks. The reduction is of practical significance because only 20–30% of female florets were fertilized in the presence of mixed pollen of both species and their hybrids.
3.2 Assessment of self-fertilization
In our in vitro pollen germination experiment, four of the six treatments of pollen on stigma extract from the same plant resulted in significantly higher pollen germination than the control, indicating that floral physiology was conducive to self-pollination (Table 3). We further analyzed the self-fertilization ability of S. integrifolium, S. perfoliatum, and interspecific hybrids in a series of crossing experiments. We found species (p < 0.001) was the most important influence on the rate of self-fertilization in the Silphium individuals we studied, with S. perfoliatum genotypes having the highest selfing rate (mean = 56.2%), followed by S. integrifolium (mean = 28.8%), and finally the interspecific hybrids (mean = 7.1%; Tables 4 and 2). The effect of genotypes within each species was not significant (p = 0.06). All results were stable over two field seasons (years) and multiple attempts with different capitula on the same genotypes. We also assessed the rate of self-fertility on an additional, unrelated set of 19 breeding family genotypes of S. integrifolium, which resulted in nine genotypes with non-zero rates of self-pollination and the highest genotype with 21.6% self-pollinated seed set. The results indicate that self-pollination appears to be possible in many genotypes, with some genotypes of both species capable of relatively high self-pollinated seed set, given manual pollination of stigmas with self-pollen. Many attempts at passive self-pollination, in which capitula were covered and not manually pollinated, always resulted in no seeds being produced (significantly different from manually self-pollinated seed set, Mann-Whitney U test p < 0.001).
3.3 Significance of the findings to de novo domestication of future crops
Both S. integrifolium and S. perfoliatum show promise as candidates for domestication, potentially serving as perennial agronomic crops providing forage, biomass, and vegetable oil. As shown by our crossing experiment results (Table 4), congenerics can hybridize and produce viable offspring. Pollen produced by these interspecies hybrids, while forced to initiate disomic pairing and meiosis with homeologous chromosomes, had very high pollen stainability, indicating normal pollen production. Pollen from hybrids readily germinated under in vitro test conditions, although at a lower rate than S. integrifolium or S. perfoliatum plants. Further, both the ovules and pollen of interspecies hybrids were fertile and resulted in seeds in all attempted self-pollinations and backcrosses to either parent (Table 4) and under open pollination (Table 5). This demonstrates that gene flow between the species can be easily facilitated, allowing for traits, such as resistance to rust disease, branching morphology, lodging resistance, length of bloom, and seed yield, to be transferred from one species to another. Members of the contribal genus Helianthus, including domesticated sunflower, are also known to hybridize across species boundaries. Introgressions from several Helianthus species are prevalent in both wild and domesticated H. annuus genomes and have been associated with genes and genetic loci providing important phenotypic variation (Hübner et al. 2019). The data presented here show that Silphium breeders will also be able to take advantage of these approaches, combining beneficial traits from different species, and suggest that the barriers to gene flow are low enough that hybridization could easily occur in the wild. Indeed, wild plant collectors have provided casual reports of wild Silphium plants with hybrid characteristics.
However, the utility of Silphium interspecific hybrids directly as F1 hybrid cultivars for oilseed production is unlikely, as the partial sterility we have observed in hybrids reduces seed yield (Table 5). Partial sterility in Silphium is likely the result of chromosomal structure differences between the parent species, resulting in abnormal meiosis in some pollen and ovule cells (Settle and Fisher 1972), but as the in vitro experiment mirrored our seed set results, a physiological cause cannot be ruled out. Other uses of Silphium (e.g., as a perennial forage or biofuel) would not require seed production past the F1 and thus may benefit from interspecific hybrids per se as cultivars, as yield of vegetation is generally observed to be high. While sterility of interspecific hybrids is a barrier to their use as plant varieties per se, the sterility is not too significant of a barrier to prevent hybridization as a means for broadening the genetic pool of either species through backcrossing or other methods.
Prior to this study, the presence of self-incompatibility had never been described in detail in these species, and its effects and implications had never been formally discussed. Self-incompatibility in Silphium could be an impediment to domestication, as it makes the development of homogenous, genetically stable, seed-propagated lines impossible, slowing numerous avenues of genetic research. On a more applied level, self-incompatibility would limit the development and fixation of recessive phenotypes critical for certain agronomic and consumer traits. Our results show, contrary to previous belief (van Tassel and DeHaan 2013), that some genotypes of two species of Silphium, as well as their interspecific hybrid offspring, are readily able to self-fertilize. This result was first suggested by in vitro pollen germination analysis (Table 1) and later empirically confirmed in vivo (Table 2). All six of the genotypes that were studied in-depth were able to self-fertilize to some extent in vivo. Further analysis of additional S. integrifolium genotypes showed considerable variability in self-seed set, with a large percentage showing non-zero levels of self-pollination. This implies that the strength of self-incompatibility in Silphium, and particularly S. integrifolium, is highly variable across genotypes and populations. This is in stark contrast to wild sunflower, which is an obligate cross-pollinator (Heiser and Smith 1969) except for a few rare examples of self-compatibility (Burke et al. 2002).
As a new crop, Silphium will need to rapidly catch up to the performance of its domesticated crop relatives and non-relatives, which now occupy the agricultural landscape. In order to displace some portion of existing crops, the performance of Silphium will have to balance ease of production, yield, quality, and associated economic considerations as well as carry new niche functions, such as ecosystem services (DeHaan et al. 2016). While, historically, inbreeding and F1 hybrid cultivars have been associated with reduced biodiversity on landscapes, as single plant genotypes are mass produced over large geographic areas, these techniques have and will continue to advance genetic gains of dominant crops relative to new domesticates. The need to advance genetic gain in new crops, to make them viable competitors to dominant, existing crops, necessitates intelligent use of inbred lines and hybrid cultivars in systems which provide functional biodiversity in other ways that do not limit genetic gain (e.g., polyculture [Glover et al. 2010], cover crops, or cultivar mixtures).
4 Conclusion
We show, for the first time, that self-pollination commonly occurs in the Silphium genus, a genus targeted for accelerated domestication as a source of food and energy in Europe, North America, and elsewhere. Degree of self-pollination is dependent on genotype, which will allow it to be a target for selection. Self-pollination will facilitate fixation of domestication trait genes in the germplasm pool, enhance the efficiency of selection in breeding, and allow for production of uniform cultivars and inbred lines that can be incorporated into functionally diverse landscapes. Also, we show that interspecific hybridization in Silphium is easy and common, providing sources of additional genetic variation beyond the primary gene pool for traits that are invariable within each species. The flexibility of reproductive biology to allow for maximal use of genetic pools, and efficient selection and curation of the genetic diversity, will increase the probability that Silphium is a successful crop in the future (DeHaan et al. 2016), to add biodiversity and restore ecosystem services to the landscape.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request. Tables 4 and 5 are also available through Zenodo at: https://zenodo.org/record/3575048#.Xut8uefgodV.
References
Alexander MP (1969) Differential staining of aborted and nonaborted pollen. Stain Technol 44:117–122. https://doi.org/10.3109/10520296909063335
Burke JM, Tang S, Knapp SJ, Rieseberg LH (2002) Genetic analysis of sunflower domestication. Genetics 161:1257–1267
Crow JF (1998) 90 years ago: the beginning of hybrid maize. Genetics 148:923–928
DeHaan LR, van Tassel DL, Anderson JA, Asselin SR, Barnes R, Baute GJ, Cattani DJ, Culman SW, Dorn KM, Hulke BS, Kantar M, Larson S, Marks MD, Miller AJ, Poland J, Ravetta DA, Rude E, Ryan MR, Wyse D, Zhang X (2016) A pipeline strategy for grain crop domestication. Crop Sci 56:917–930. https://doi.org/10.2135/cropsci2015.06.0356
Eckert CG, Kalisz S, Geber MA, Sargent R, Elle E, Cheptou PO, Goodwillie C, Johnston MO, Kelly JK, Moeller DA, Porcher E, Ree RH, Vallejo-Marín M, Winn AA (2010) Plant mating systems in a changing world. Trends Ecol Evol 25:35–43. https://doi.org/10.1016/j.tree.2009.06.013
Glover JD, Reganold JP, Bell LW, Borevitz JO, Brummer EC, Buckler ES et al (2010) Increased food and ecosystem security via perennial grains. Science 328:1638–1639. https://doi.org/10.1126/science.1188761
Gonthier L, Blassiau C, Mörchen M, Cadalen T, Poiret M, Hendriks T, Quillet MC (2013) High-density genetic maps for loci involved in nuclear male sterility (NMS1) and sporophytic self-incompatibility (S-locus) in chicory (Cichorium intybus L., Asteraceae). Theor Appl Genet 126:2103–2121. https://doi.org/10.1007/s00122-013-2122-9
Hallauer AR (2007) History, contribution, and future of quantitative genetics in plant breeding: lessons from maize. Crop Sci 47:4–19. https://doi.org/10.2135/cropsci2007.04.0002IPBS
Heiser CB, and Smith DM (1969) The North American sunflowers (Helianthus). Durham N.C.: published for the Club by the Seeman Printery Available at: https://www.jstor.org/stable/43390641. Accessed 22 May 2020
Hübner S, Bercovich N, Todesco M, Mandel JR, Odenheimer J, Ziegler E, Lee JS, Baute GJ, Owens GL, Grassa CJ, Ebert DP, Ostevik KL, Moyers BT, Yakimowski S, Masalia RR, Gao L, Ćalić I, Bowers JE, Kane NC, Swanevelder DZH, Kubach T, Muños S, Langlade NB, Burke JM, Rieseberg LH (2019) Sunflower pan-genome analysis shows that hybridization altered gene content and disease resistance. Nature Plants 5:54–62. https://doi.org/10.1038/s41477-018-0329-0
Jessee LH, Yatskievych G (2011) A new putative hybrid in Silphium (Asteraceae: Heliantheae). Phytoneuron 62:1–7
Kao TH, McCubbin AG (1996) How flowering plants discriminate between self and non-self pollen to prevent inbreeding. Proc Natl Acad Sci 93:12059–12065. https://doi.org/10.1073/pnas.93.22.12059
Keshava Murthy MN, Nanja Reddy YA, Virupakshappa K (1994) Development of suitable germination medium for trinucleate, pollen grains: an illustration. J Oilseeds Res 11:304–307
Kowalski R, Wiercinski J (2004) Evaluation of chemical composition of some Silphium L. Species Seeds As. Polish J Food Nutr Sci 13:349–354
Muñoz-Sanz JV, Zuriaga E, Cruz-Garcia F, McClure B, Romero C (2020) Self-(in)compatibility systems: target traits for crop-production, plant breeding, and biotechnology. Front Plant Sci 11
Nazzi F, Pennacchio F (2014) Disentangling multiple interactions in the hive ecosystem. Trends Parasitol 30:556–561. https://doi.org/10.1016/j.pt.2014.09.006
Reinert S, van Tassel DL, Schlautman B, Kane NC, Hulke BS (2019) Assessment of the biogeographical variation of seed size and seed oil traits in wild Silphium integrifolium Michx . genotypes. Plant Genetic Resources: Characterization and Utilization 17:1–10. https://doi.org/10.1017/S1479262119000248
Settle WJ, and Fisher TR (1972) A biosystematic study of Silphium Integrifolium Michaux (Compositae). The Ohio Journal of Science 72, 254–265. Available at: https://kb.osu.edu/handle/1811/5717. Accessed 22 May 2020
Silva NF, Goring DR (2001) Mechanisms of self-incompatibility in flowering plants. Cell Mol Life Sci 58:1988–2007. https://doi.org/10.1007/PL00000832.
USDA-NRCS (2019) Classification for Kingdom Plantae down to Genus Silphium L. https://plants.usda.gov/java/ClassificationServlet?source=profile&symbol=SILPH&display=31. Accessed 13 July 2020
van Tassel D, DeHaan L (2013) Wild plants to the rescue. Am Sci 101:218–225. https://doi.org/10.1511/2013.102.218
van Tassel DL, Asselin SR, Cox SA, Sideli G, and Cattani DJ (2014) Evaluating perennial candidates for domestication: lessons from wild sunflower relatives. Perennial Crops for Food Security; Proceedings of the FAO Expert Workshop, 112–140
van Tassel DL, Albrecht KA, Bever JD, Boe AA, Brandvain Y, Crews TE, Gansberger M, Gerstberger P, González-Paleo L, Hulke BS, Kane NC, Johnson PJ, Pestsova EG, Picasso Risso VD, Prasifka JR, Ravetta DA, Schlautman B, Sheaffer CC, Smith KP, Speranza PR, Turner MK, Vilela AE, von Gehren P, Wever C (2017) Accelerating Silphium domestication: an opportunity to develop new crop ideotypes and breeding strategies informed by multiple disciplines. Crop Sci 57:1274–1284. https://doi.org/10.2135/cropsci2016.10.0834
Whitehead MR, Lanfear R, Mitchell RJ, Karron JD (2018) Plant mating systems often vary widely among populations. Front Ecol Evol 6:1–9. https://doi.org/10.3389/fevo.2018.00038
Zilverberg CJ, Teoh K, Boe A, Johnson WC, Owens V (2016) Strategic use of native species on environmental gradients increases diversity and biomass relative to switchgrass monocultures. Agric Ecosyst Environ 215:110–121. https://doi.org/10.1016/j.agee.2015.09.006
Acknowledgments
The authors would like to acknowledge Dustin Bailey for his assistance in performing cross- and self-pollinations at the Prosper location and Karen Beaubein for her assistance in performing pollinations at the St. Paul location.
Author information
Authors and Affiliations
Contributions
Conceptualization, SR, BSH, DVT, and JHP; methodology, SR, BSH, DVT, and JHP; investigation, SR, JHP, and BCS; formal analysis, SR, JHP, BCS, CSP, NCK, and BSH; resources, DVT; data curation, SR; writing (original draft preparation) SR, JHP, and BSH; writing (review and editing) NCK, DVT, BSH, SR, JHP, BCS, and CSP; visualization, CSP and JHP; project administration, BSH; and funding acquisition, NCK, BSH, and DVT.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Reinert, S., Price, J.H., Smart, B.C. et al. Mating compatibility and fertility studies in an herbaceous perennial Aster undergoing de novo domestication to enhance agroecosystems. Agron. Sustain. Dev. 40, 27 (2020). https://doi.org/10.1007/s13593-020-00632-5
Accepted:
Published:
DOI: https://doi.org/10.1007/s13593-020-00632-5