Abstract
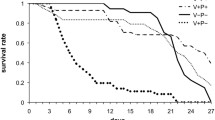
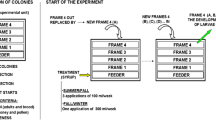
Fatty acids (FAs) are important in metabolism and in the maintenance of Apis mellifera colonies. Our objective was to identify the variation in FA composition of the bodies of A. mellifera worker bees after immunological challenges with sterile injections and Paenibacillus larvae vegetative cells. The expression of the hymenoptaecin and abaecin genes was studied and the FAs were identified and quantified. After a challenge with P. larvae, the immune system was activated and abaecin and hymenoptaecin peptide genes were overexpressed. Also, the content of short-chain FAs such as caprylic, capric, and tridecylic acids increased significantly; on the contrary, oleic acid decreased. To the best of our knowledge, this is the first analysis of changes in the FA profile of honey bee workers after immune stimulation with P. larvae. Health status may be a key factor that alters FA composition. More research about the interaction with other stressors affecting the survival of honey bee colonies is needed.




Similar content being viewed by others
Data availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Abbreviations
- FAs:
-
Fatty acids
- TAGs:
-
Triglycerides
- CH:
-
Cuticular hydrocarbons
- BHT:
-
Butylated hydroxytoluene
- FAMEs:
-
Methyl esters of fatty acids
- GC-FID:
-
Gas chromatography with flame ionization detection
- SFAs:
-
Saturated fatty acids
- MUFAs:
-
Monounsaturated fatty acids
- PUFAs:
-
Polyunsaturated fatty acids
References
Angelo IC, Gôlo PS, Perinotto WMS, Camargo MG, Quinelato S, Sá FA, Pontes EG, Bittencourt VREP (2013) Neutral lipid composition changes in the fat bodies of engorged females Rhipicephalus microplus ticks in response to fungal infections. Parasitol Res 112:501–509. https://doi.org/10.1007/s00436-012-3159-4
Annoscia D, Zanni V, Galbraith D, Quirici A, Grozinger C, Bortolomeazzi R, Nazzi F (2017) Elucidating the mechanisms underlying the beneficial health effects of dietary pollen on honey bees (Apis mellifera) infested by Varroa mite ectoparasites. Sci Rep 7:6258. https://doi.org/10.1038/s41598-017-06488-2
Arien Y, Dag A, Zarchin S, Masci T, Shafir S (2015) Omega-3 deficiency impairs honey bee learning. PNAS 112(51):15761–15766
Arrese EL, Soulages JL (2010) Insect fat body: energy, metabolism, and regulation. Annu Rev Entomol 55(207–225):112408–185356
Bustin SA (2002) Quantification of mRNA using real-time reverse transcription PCR (RT-PCR): trends and problems. J Mol Endocrinol 29:23–39. https://doi.org/10.1677/jme.0.0290023
Castaños CE, Boyce MC, Bates T, Millar AH, Flematti G, Lawler NG, Grassl J (2023) Lipidomic features of honey bee and colony health during limited supplementary feeding. Insect Mol Biol 32(6):658–675. https://doi.org/10.1111/imb.12864
Cheon HM, Shin SW, Bian G, Park JH, Raikhel AS (2006) Regulation of lipid metabolism genes, lipid carrier protein lipophorin, and its receptor during immune challenge in the mosquito Aedes aegypti. J Biol Chem 281(13):8426–8435. https://doi.org/10.1074/jbc.M510957200
Corby-Harris V, Bennett MM, Deeter ME (2021) Fatty acid homeostasis in honey bees (Apis mellifera) fed commercial diet supplements. Apidologie 52:1195–1209. https://doi.org/10.1007/s13592-021-00896-0
Crone MK, Grozinger CM (2021) Pollen protein and lipid content influence resilience to insecticides in honey bees (Apis mellifera). J Exp Biol 224(9):1–11. https://doi.org/10.1242/jeb.242040
Dettloff M, Wittwer D, Weise C, Wiesner A (2001) Lipophorin of lower density is formed during immune responses in the lepidopteran insect Galleria mellonella. Cell Tissue Res 306:449–458. https://doi.org/10.1007/s00441-001-0468-9
Evans JD, Aronstein K, Chen YP, Hetru C, Imler J-L, Jiang H, Kanost M, Thompson GJ, Zou Z, Hultmark D (2006) Immune pathways and defence mechanisms in honey bees Apis mellifera. Abstr Insect Mol Biol 15(5):645–656. https://doi.org/10.1111/j.1365-2583.2006.00682.x
Feldlaufer MF, Knox DA, Lusby WR, Shimanuki H (1993) Antimicrobial activity of fatty acids against Bacillus larvae, the causative agent of American foulbrood disease. Apidologie 24:95–99. https://doi.org/10.1051/apido:19930202
Fernández NJ, Porrini MP, Enrique AP, Damiani N, Liesel BG, Martín JE (2014) A scientific note on the first report of honeybee venom inhibiting Paenibacillus larvae growth. Apidologie 45:719–721. https://doi.org/10.1007/s13592-014-0289-y
Folch J, Less M, Sloane Stanley GH (1957) A simple method for isolation and purification of total lipids from animal tissue. J Biol Chem 226:497–509. https://doi.org/10.1016/S0021-9258(18)64849-5
Ghosh S, Herren P, Meyer-Rochow VB, Jung C (2021) Nutritional composition of honey bee drones of two subspecies relative to their pupal developmental stages. InSects 12:759. https://doi.org/10.3390/insects12080759
Ghosh S, Jung C, Meyer-Rochow VB (2016) Nutritional value and chemical composition of larvae, pupae, and adults of worker honey bee, Apis mellifera ligustica as a sustainable food source. J Asia Pac Entomol 19:487–495. https://doi.org/10.1016/j.aspen.2016.03.008
Giri S, Rule DC, Dillon ME (2018) Fatty acid composition in native bees: associations with thermal and feeding ecology. Comp. Biochem Physiol Part A Mol Integr Physiol 218:70–79. https://doi.org/10.1016/j.cbpa.2018.01.013
Gołębiowski M, Urbanek A, Pietrzak A, Naczk AM, Bojke A, Tkaczuk C, Stepnowski P (2020) Effects of the entomopathogenic fungus Metarhizium flavoviride on the fat body lipid composition of Zophobas morio larvae (Coleoptera: Tenebrionidae). Sci Nat. 107:7. https://doi.org/10.1007/s00114-019-1662-5
Hernández López J, Riessberger-Gallé U, Crailsheim K, Schuehly W (2017) Cuticular hydrocarbon cues of immune-challenged workers elicit immune activation in honeybee queens. Mol Ecol 26:3062–3073. https://doi.org/10.1111/mec.14086
Howard RW, Blomquist GJ (2005) Ecological, behavioral, and biochemical aspects of insect hydrocarbons. Annu Rev Entomol 50:371–393. https://doi.org/10.1146/annurev.ento.50.071803.130359
Kaczmarek A, Boguś M (2021) The metabolism and role of free fatty acids in key physiological processes in insects of medical, veterinary and forensic importance. PeerJ 9:e12563. https://doi.org/10.7717/peerj.12563
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lourenço AP, Martins JR, Bitondi MMG, Simões ZLP (2009) Trade-off between immune stimulation and expression of storage protein genes. Arch Insect Biochem Physiol 71:70–87. https://doi.org/10.1002/arch.20301
Maggi M, Antúnez K, Invernizzi C et al (2016) Honeybee health in South America. Apidologie 47:835–854. https://doi.org/10.1007/s13592-016-0445-7
Martin N, Hulbert AJ, Brenner GC, Brown SHJ, Mitchell TW, Else PL (2019) Honey bee caste lipidomics in relation to life-history stage and the long life of the queen. J Exp Biol 16:222. https://doi.org/10.1242/jeb.207043
McAfee A, Chapman A, Iovinella I, Gallagher-Kurtzke Y, Collins TF, Higo H, Madilao LL, Pelosi P, Foster LJ (2018) A death pheromone, oleic acid, triggers hygienic behavior in honey bees (Apis mellifera L.). Sci Rep 8:5719. https://doi.org/10.1038/s41598-018-24054-2
Mullen L, Goldsworthy G (2003) Changes in lipophorins are related to the activation of phenoloxidase in the haemolymph of Locusta migratoria in response to injection of immunogens. Insect Biochem Mol Biol 33:661–670. https://doi.org/10.1016/S0965-1748(03)00045-6
Nelson EV, Milstead J, Kulinčević JM (1971) Fatty acids in virus-infected worker honey bees (Apis mellifera). J Invertebr Pathol 17(3):366–368. https://doi.org/10.1016/0022-2011(71)90010-3
Plettner E, Slessor KN, Winston ML (1998) Biosynthesis of mandibular acids in honey bees (Apis mellifera): de novo synthesis, route of fatty acid hydroxylation and caste selective β-oxidation. Insect Biochem Mol Biol 28(1):31–42. https://doi.org/10.1016/S0965-1748(97)00079-9
Porrini LP, Quintana S, Brasesco C, Porrini MP, Garrido PM, Eguaras MJ, Müller F, Fernandez Iriarte P (2020) Southern limit of Africanized honey bees in Argentina inferred by mtDNA and wing geometric morphometric analysis. J Apic Res 59:648–657. https://doi.org/10.1080/00218839.2019.1681116
Potts SG, Imperatriz-Fonseca V, Ngo HT, Aizen MA, Biesmeijer JC, Breeze TD, Vanbergen AJ (2016) Safeguarding pollinators and their values to human well-being. Nature 540(7632):220–229. https://doi.org/10.1038/nature20588
Randolt K, Gimple O, Geissendörfer J, Reinders J, Prusko C, Mueller MJ, Albert S, Tautz J, Beier H (2008) Immune-related proteins induced in the hemolymph after aseptic and septic injury differ in honey bee worker larvae and adults. Arch Insect Biochem Physiol 69:155–167. https://doi.org/10.1002/arch.20269
Requier F, Odoux JF, Tamic T, Moreau N, Henry M, Decourtye A, Bretagnolle V (2015) Honey bee diet in intensive farmland habitats reveals an unexpectedly high flower richness and a major role of weeds. Ecol Appl 28(4):881–890. https://doi.org/10.1890/14-1011.1
Richard F-J, Aubert A, Grozinger C (2008) Modulation of social interactions by immune stimulation in honey bee, Apis mellifera, workers. BMC Biol 6:50. https://doi.org/10.1186/1741-7007-6-50
Richard F-J, Holt HL, Grozinger CM (2012) Effects of immunostimulation on social behavior, chemical communication and genome-wide gene expression in honey bee workers (Apis mellifera). BMC Genomics 13:558. https://doi.org/10.1186/1471-2164-13-558
Roberts MI (1968) Fatty acids in honey bees (Apis mellifera) infected with the protozoan Nosema apis. J Invertebr Pathol 11:234–236. https://doi.org/10.1016/0022-2011(68)90154-7
Robinson FA, Nation JL (1970) Long-chain fatty acids in honeybees in relation to sex, caste, and food during development. J Apic Res 9(3):121–127. https://doi.org/10.1080/00218839.1970.11100258
Shahbandeh M (2023) Number of beehives worldwide 2010–2021. Statista. https://www.statista.com/statistics/818286/number-of-beehives-worldwide/. Accessed 15 Mar 2024
Slessor KN, Winston ML, Le Conte Y (2005) Pheromone communication in the honeybee (Apis mellifera L.). J Chem Ecol 31:2731–2745. https://doi.org/10.1007/s10886-005-7623-9
Team RC (2016) R: a language and environment for statistical computing
Toprak U (2020) The role of peptide hormones in insect lipid metabolism. Front Physiol 11:434. https://doi.org/10.3389/fphys.2020.00434
Toth AL, Kantarovich S, Meisel AF, Robinson GE (2005) Nutritional status influences socially regulated foraging ontogeny in honey bees. J Exp Biol 208:4641–4649. https://doi.org/10.1242/jeb.01956
Van der Meer-Janssen YPM, Van Galen J, Batenburg JJ, Helms JB (2010) Lipids in host–pathogen interactions: pathogens exploit the complexity of the host cell lipidome. Prog Lipid Res 49:1–26. https://doi.org/10.1016/j.plipres.2009.07.003
Wright GA, Nicolson SW, Shafir S (2018) Nutritional physiology and ecology of honey bees. Annu Rev Entomol 63(1):327–344. https://doi.org/10.1146/annurev-ento-020117-043423
Yang X, Cox-Foster DL (2005) Impact of an ectoparasite on the immunity and pathology of an invertebrate: evidence for host immunosuppression and viral amplification. Proc Natl Acad Sci 102(21):7470–7475. https://doi.org/10.1073/pnas.0501860102
Zalewski K, Zaobidna E, Żóltowska K (2016) Fatty acid composition of the parasitic mite Varroa destructor and its host the worker prepupae of Apis mellifera. Physiol Entomol 41(1):31–37. https://doi.org/10.1111/phen.12121
Zdybicka-Barabas A, Cytryńska M (2013) Apolipophorins and insects immune response. Invertebr Surviv J 10(1):58–68
Acknowledgements
We appreciate the help provided by Dr. Karina Miglioranza (IIMyC-UNMdP) and Carlos Mendez (Fares Taie lab) in the extracting and determining of fatty acids.
Funding
This work was supported by Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) with a scholarship to Lic Enzo Dominguez, Universidad Nacional de Mar del Plata with subsidy UNMdP-EXA project No. 973/20 to Dr. Martín Eguaras, and Agencia Nacional de Promoción Científica y Técnica with subsidy ANPCyT-PICT project No. 2018-1758 to Dra. Natalia Damiani.
Author information
Authors and Affiliations
Contributions
ED developed the study idea, managed the honey bee colonies, collected data and samples, analyzed the samples in the laboratory, statistically analyzed the data, and wrote the manuscript. PMG managed the honey bee colonies, collected data and samples, and analyzed the samples in the laboratory. SQ, MPM, MC, and SKM analyzed the samples in the laboratory. LBG and ND direct the investigation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This is an observational study, and no ethical approval is required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Manuscript editor: David Tarpy
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Domínguez, E., Giardini, P.M., Quintana, S. et al. Fatty acid profile of Apis mellifera workers in the face of an immune challenge. Apidologie 55, 15 (2024). https://doi.org/10.1007/s13592-024-01059-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13592-024-01059-7