Abstract
Due to recent changes in regulation, acetamiprid has become the only neonicotinoid that can be applied without restrictions and in open field cultivations in the EU from 2021. We provide an overview of the current knowledge on the effects of this insecticide on bumblebees and assessed whether available empirical evidence supports the claim that acetamiprid poses negligible risk to these pollinators. We found that there is limited data on the lethal and sublethal effects of this pesticide on bumblebees. While risk assessment results suggest that field-realistic concentrations of acetamiprid have minor acute and chronic toxicity, detrimental sublethal effects, including reduced reproductive output, have been observed when bumblebees were exposed to high doses of this insecticide. We propose that further research on the topic is warranted as the more extensive application of acetamiprid may lead to such high concentrations in the field.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Thirty-five percent of the world crops need pollinators for reproduction (Klein et al. 2007; Reilly et al. 2020). There are a number of environmental factors that have been found to negatively affect pollinator populations worldwide: the most important stressors include the lack of a variety of food sources due to intensive agricultural production (Naug 2009; Donkersley et al. 2014), infection by parasites and diseases (Goulson et al. 2015; Cameron and Sadd 2020), extreme weather due to climate change (Kerr et al. 2015; Martinet et al. 2020), and exposure to various pesticides (Blacquière et al. 2012; Botías et al. 2021). As the current trends in pollinator decline have the potential to jeopardize future sustainability of crop pollination in many agroecosystems (Stanley et al. 2015; Whitehorn et al. 2017; Fijen et al. 2018), it has become essential to explore the pathways through which such environmental factors interfere with pollinator life histories and take actions to mitigate their detrimental effects.
Bumblebees (Apidae: Bombus spp.) are important pollinators of both crops and wildflowers (Hutchinson et al. 2021). These bees have a particular advantage in providing pollination services: they are active in cold weather, and with their large, hairy body, they can carry a greater pollen load than other pollinators (Goulson 2010). By vibrating their bodies during pollination, they can also shake the anthers of the visited flowers/blossoms and increase pollination efficacy, particularly in plants belonging to the Solanaceae and Fabaceae families (Switzer and Combes 2017). Accumulating evidence suggests that modern insecticides, particularly neonicotinoids, can negatively affect the foraging and reproductive performance of bumblebees (Gill et al. 2012; Rundlöf et al. 2015; Stanley et al. 2016; Woodcock et al. 2017; Lu et al. 2020), and may be — at least partly — responsible for the recent decline of wild and managed bumblebee populations, threatening the pollination service these species provide (Goulson et al. 2015; Baron et al. 2017).
Neonicotinoids are systemic insecticide neurotoxins that act as agonists of the nicotinic acetylcholine receptors (nAChRs) of insects (Manjon et al. 2018). These substances have been used effectively against a wide range of agricultural and veterinary pests through both direct contact and consumption since their introduction in the 1990s (Elbert et al. 2008; Simon-Delso et al. 2015; Matsuda et al. 2020). Based on their structure, neonicotinoids can be classified into three major types: nitroguanidines, nitromethylenes, and cyanoamidines (Ghosh and Jung 2017). Cyanoamidines (acetamiprid and thiacloprid) are thought to be less harmful to pollinators than nitro-substituted ones (e.g. clothianidin, imidacloprid and thiamethoxam) (Iwasa et al. 2004; Decourtye and Devillers 2010), because cytochrome P450s in honeybees and bumblebees can rapidly metabolize these substances (Brunet et al. 2005; Manjon et al. 2018).
Due to the growing concerns regarding their toxicity to pollinators, the European Commission has banned the outdoor uses of three neonicotinoids (clothianidin, imidacloprid, thiamethoxam) in 2018 (EC 2018a, b, c), and the approval of thiacloprid also expired by the end of 2020. Acetamiprid, on the other hand, has been identified as having a low risk to bees (EFSA 2016), and its approval has been renewed until 28th February, 2033 (EC 2018d). With that, this substance became the only neonicotinoid that can be used without restrictions and also in open field cultivations in Europe (although some EU countries applied for multiple derogations on major crops since the start of the restrictions). The US Environmental Protection Agency (USEPA) similarly classified acetamiprid as practically non-toxic in acute oral and contact exposures to bumblebees (Douglass et al. 2017). Acetamiprid is the ISO name for (E)-N1-[(6-chloro-3-pyridyl)methyl]-N2-cyano-N1-methylacetamidine (International Union of Pure and Applied Chemistry, IUPAC). Its production began in 1995 in Japan by Nippon Soda (Takahashi et al. 1992). Acetamiprid is distributed (as water-soluble granules) under the trade names of Mospilan, Autentic, Gazelle (containing 20% acetamiprid), and Assail (containing 30% acetamiprid) in the EU, UK, and USA (several other trade names also exist on the global market). It is used to control insect pests, for example, on vegetables, cotton, sunflower, soy, fruits, and tea (Elbert et al. 2008; Jeschke et al. 2011). Based on information available until 2009 (in same cases until 2012), acetamiprid had a low market share among other neonicotinoids (approximately 10.5% of the global sales), and similar or lower percentage of acetamiprid sale was characteristic to countries such as the UK, Sweden, Japan, and also to California, USA (Jeschke et al. 2011; Simon-Delso et al. 2015). However, there is very limited up-to-date information available on the amount of acetamiprid currently sold and used around the world. Moreover, its share is likely to grow in the coming years, especially in many European countries owing to the introduced EU regulations (Camp and Lehmann 2020).
There are several reasons why the predicted increase in the amount of applied acetamiprid in agricultural production deserves the attention of conservation biologists and relevant authorities. Like all neonicotinoids, acetamiprid and its residues can persist in the nectar (Mitchell et al. 2017) and the pollen of treated plants (0.04–104 ng/g; Lentola et al. 2017; Calatayud-Vernich et al. 2018). Its half-life in the soil varies greatly among soil types and strongly depends on soil temporal conditions (Gupta et al. 2008): it is short (1–8 days) in aerobic soils (EPA 2002), but in clay soils, the degradation takes order of magnitude longer (450 days; Goulson 2013). The application of acetamiprid-containing insecticides at the recommended rates has been shown to provide a potential exposure route to foraging bumblebees (Chandler et al. 2020), and its repeated usage in successive years is expected to result in accumulating concentrations in nectar, pollen, and soil (Sanchez-Bayo and Goka 2014; Zioga et al. 2020). Continuous and extensive application of acetamiprid can also contaminate wildflowers near agricultural areas (Botías et al. 2015). Although specific data for acetamiprid is lacking, this substance is likely to exert similar time-dependent effects as other neonicotinoids (Sanchez-Bayo and Tennekes 2020). Due to the mode of action of these chemicals, long exposures of bees to acetamiprid are expected to have much larger effects than acute exposures in terms of lethality. In pollinators such as bumblebees and other wild bee species, which are characterized by long life cycles and small colonies, such time-dependent lethal effects may be much more detrimental than in honeybees, where workers are generally more easily replaced. The other prominent threat that prolonged exposure to this substance imposes on bumblebees is its sublethal effects such as chronic intoxication and compromised foraging. Foraging activity in bumblebees have a direct effect on colony development and growth by impacting gyne (unmated reproductive female) and drone production, and thus, its reduction can negatively impact the number of overwintering queens and their nutritional status (Camp and Lehmann 2020). As a queen’s vitality and foraging behaviour are critical for successful nest initiation in the spring (Goulson 2010), the after effects of compromised foraging in the previous year can diminish the number of nests formed in a season.
In this study, we reviewed our current knowledge on the lethal and sublethal effects of acetamiprid exposure on bumblebees and addressed the question of whether available empirical evidence supports the claim that acetamiprid poses negligible risk to these pollinators. We also identified important knowledge gaps and highlighted potential directions for further research, and provided suggestions for a more efficient risk assessment related to this neonicotinoid substance.
2 Search method
To provide a comprehensive overview, we searched the literature for published research articles using the Scopus, Web of Science, and PubMed databases (date of search: 24 February 2021). We conducted searches using keyword pairs of all combinations between “bombus”, “bumblebee”, “bumble bee”, “apis”, “honeybee”, “honey bee” and “neonicotinoid” “acetamiprid”, “mospilan”, and “assail” keywords (Supplementary Table 1), where keyword pairs were separated with an “AND” operator. Duplicated articles and publications written in languages other than English were excluded. We applied the same process to review papers and book chapters, and references from these works that did not appear during our search for research articles were added to the list of potentially relevant articles. Then, the titles, abstracts, and methods sections were screened, and we retained those articles that were considered relevant (i.e. provided measurement data on the effect of an acetamiprid treatment) from the dataset (Figure 1). We included one additional publication to the list that was not found by the keyword searches because of a consistent typo in a relevant keyword. Figures were created using R 3.6.3. statistical software (R Core Team 2020), and the yEd Graph Editor 3.21 (yFiles software, Tubingen, Germany, http://www.yWorks.com).
PRISMA flow chart showing our search and screening process (Moher et al. 2010).
3 Effect of acetamiprid in bumblebees
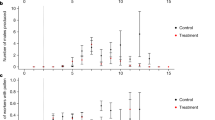
We found 3331 publications in total using the above keyword pair combinations (Supplementary Material 1), out of which we identified 9 and 43 research articles as relevant for bumblebees and honeybees, respectively (Supplementary Material 2). The almost five-fold difference in the number of published papers implies a substantial difference in research effort devoted to honeybees and bumblebees in this topic, probably due to current pesticide risk assessment regulations that require only the use of honeybees as indicator organisms (EFSA 2013). In the studied 16-year period, experimental results related to the effects of acetamiprid on bumblebees were published only occasionally until 2020, while the number of studies on bumblebees in 2020 became similar to those conducted on honeybees (Figure 2).
3.1 Acute contact toxicity
Acute contact toxicity was estimated in three out of nine studies, but the results are difficult to compare due to differences in methodology, model species, and the applied form of acetamiprid. Reid et al. (2020) found that regardless of whether mortality was observed after 48 or 72 h, or whether analytic acetamiprid was used by itself or together with a synergist chemical (piperonyl butoxide; PBO), acetamiprid was practically non-toxic in Bombus terrestris audax (Harris 1790) as the applied treatment did not cause sufficient mortality to generate LD50 (median lethal dose) values even when workers were treated with 100 μg/bee dose of analytical acetamiprid (Figure 3). In Bombus impatiens Cresson, 1863, Chandler et al. (2020) monitored mortality for two days after treating workers with a 14.5-µg/bee dose of acetamiprid-containing Assail (30% acetamiprid content) and found that acetamiprid did not affect mortality either by itself or in mixture with a propiconazole fungicide. Sanchez-Bayo and Goka (2014) used LD50 values obtained from the ECOTOX database to calculate mortality risks under field-realistic exposure conditions and concluded that acetamiprid had a negligible effect on bumblebees in mixture with propiconazole (LD50 = 0.95 μg/bee, risk (%) ≤ 0.07) or fenbuconazole (LD50 = 22.2 μg/bee, risk (%) ≤ 0.01) fungicides.
Median lethal doses (LD50) of acetamiprid for bumblebees and honeybees obtained by oral and contact bioassays. Compound composition is colour-coded as red — acetamiprid only, green — acetamiprid combined with another compound, blue — Mospilan. Different exposure durations are symbol-coded as circles — 24 h, triangles — 48 h, squares — 72 h. Error bars represent 95% confidence intervals (if available). The dashed lines are depicted according to EPA toxicity classification scale for honeybees (highly toxic: LD50 < 2 µg/bee, moderately toxic: LD50 2–10.99 µg/bee, slightly toxic: LD50 11–100 µg/bee, practically non-toxic: LD50 > 100 µg/bee) (USEPA 2014). The identified toxicity studies on bumblebees vary greatly in the testing duration, model species, and/or the form of acetamiprid applied, making the statistical comparison of their findings unfeasible. Therefore, this figure primarily serves illustrational purposes to highlight the substantial difference in the number of studies conducted on honeybees and bumblebees related to the effects of acetamiprid and to demonstrate that the LD50s reported by those few studies may differ in the order of magnitude.
3.2 Acute oral toxicity
Acute oral toxicity tests were performed in four studies, but the same issues apply here related to the comparability of their results as in the acute contact toxicity tests. Wu et al. (2010) found that after 48 h of oral exposure to a 1:5000 v/v dose of acetamiprid-containing Mospilan (20% acetamiprid content [erroneously indicated as containing 3% acetamiprid in the original paper]) resulted in significantly higher mortality in three Asian bumblebee species (Bombus hypocrita Pérez 1905, Bombus ignitus Smith 1869, Bombus patagiatus Nylander 1848) compared to the control, with the LD50 values of Mospilan being 0.0023 μg/bee, 0.0028 μg/bee, and 0.0021 μg/bee for B. ignitus, B. hyprocrita, and B. patagiatus workers, respectively (Figure 3). In Baines et al. (2017), B. impatiens workers were exposed to eight doses of analytical acetamiprid (from 0.039 pg/μL to 50 µg/µL) for 24 h, and mortality and neurological impairments were recorded at 24, 48, 72, 96, and 336 h. Comparisons of mortality between specific doses and the control group were not reported in the study, but the LD50 value associated with the oral application of acetamiprid was estimated to be 300 µg/bee (this value was equivalent to the 2.5 µg/µL treatment with a 120-μL consumption rate, i.e. the highest amount consumed in the bioassays) (Figure 3). However, the authors also described early-onset (minutes to 24 h after acute exposure) neurological symptoms such as slow to no movements in the four highest doses (from 6.25 ng/μL to 50 µg/µL) and delayed-onset (5 to 7 days after acute exposure) symptoms such as slow movements and abnormal stance in the moderate and low dose treatment groups. Animals showing these symptoms remained alive during the 14-day assessment period, but did not recover and ceased their routine daily activity including feeding (Baines et al. 2017). These observations support the expectation that acetamiprid has a relatively slow mode of action and may exert substantial time-dependent effects that cannot be captured by traditional acute endpoints. In B. terrestris audax workers, oral application of acetamiprid was reported to be slightly toxic (according to the toxicity classification used by Felton et al. 1986 and USEPA 2014) with LD50 values being 13.13 µg/bee (95% CI: 9.27–18.63) after 48 h and 12.88 µg/bee (95% CI: 9.18–18.03) after 72 h, respectively (Reid et al. 2020). When used together with PBO, LD50 values decreased slightly (48 h after exposure: 9.03 µg/bee (95% CI: 5.44–13.07); 72 h after exposure: 8.45 µg/bee (95% CI: 5.47–11.69)), and thus, their synergist effect was classified as moderately toxic (Figure 3). Sanchez-Bayo and Goka (2014) used the LD50 values of 22.2 µg/bee for acetamiprid when applied in itself and 0.21 µg/bee when used together with propiconazole, and estimated that this neonicotinoid substance posed a minor risk to bumblebees in oral application as well (both risks (%) ≤ 0.02).
3.3 Chronic contact toxicity
We found only one study that estimated the contact toxicity of chronic exposure to acetamiprid in bumblebees. Wu et al. (2010) found that the mortality of the treated workers 16 days after the initial contact with a 1:5000 v/v dose of Mospilan was significantly higher than those of the control group in all three species (with the percentage mortality being close to 100% in B. patagiatus and B. ignitus), with the survival functions indicating a slow-acting effect.
3.4 Chronic oral toxicity
Three out of the nine studies reported the results of chronic oral toxicity tests. In B. impatiens workers, analytical acetamiprid was found to be non-toxic even at the highest dose applied (50 µg/µl) after 14 days of exposure (Baines et al. 2017). The applied doses were nevertheless associated with a high variability of neuromuscular dysfunctions (similar to those observed in the acute oral test), which can be regarded as important non-lethal symptoms of intoxication and manifestations of the substance’s time-dependent effect. In addition, two studies reported data on mortality associated with chronic exposure to different doses of acetamiprid in B. impatiens microcolonies. Camp et al. (2020a) found that even the highest concentration of analytical acetamiprid in pollen (4520 μg/kg) did not increase worker mortality during a 6-week-long observation period, and worker behaviour was unaffected by all levels of exposure. Similarly, even the highest dose of acetamiprid in syrup (11300 μg/L) had no adverse effect on worker survival over 7 weeks (on the contrary, the highest concentration group had a significantly higher survival than the control group), while altered behaviour (i.e. changes in activity) occurred in the two highest exposure concentrations (1300 μg/L and 11300 μg/L, respectively) (Camp et al. 2020b).
3.5 Sublethal effects
The potential sublethal effects of acetamiprid on bumblebees were investigated in five out of the nine studies. Chandler et al. (2020) reported that the addition of Assail to the syrup over 2 weeks had no significant negative impact on the consumption rate, colony weight, or gyne production in B. impatiens queenright colonies compared to the control. However, they found that there were significantly more small-sized workers in colonies exposed to Assail (either in itself or in mixture with a propiconazole fungicide) than in the control colonies and higher wax moth infestations and densities in colonies that were exposed to the mixture of the two pesticides. In B. impatiens microcolonies, adverse negative effects of acetamiprid delivered in pollen or syrup were observed in nest size and complexity, food consumption, and drone production, but only at the two highest concentrations (1130 and 11300 μg/L) that exceed the currently expected environmental concentrations (Camp et al. 2020a, b). Van Oystaeyen et al. (2020) studied the sublethal effects of Gazelle (20% acetamiprid content) exposure in Bombus terrestris (Linnaeus 1758) microcolonies and queenright colonies under short-term and long-term laboratory, and long-term field conditions. The measured output variables included the number and weight of workers and reproductives (i.e. summed number of gynes and drones), flight activity, pollen income, total colony size, and colony weight gain. The findings indicated no significant negative effects of acetamiprid in either setting, only a trend for the reduced number of reproductives in queenright colonies in the field. Nevertheless, the authors argued that as the applied dose in their study can be considered conservative, further research is needed to investigate the effects of acetamiprid in higher, yet field-realistic concentrations. In a 3-year outdoor small-plot experiment, the treatment of different sunflower cultivars with Mospilan did not influence significantly the pooled visitation rate of two bumblebee species (B. terrestris and Bombus lapidarius (Linnaeus 1758)) either (Stejskalova et al. 2018).
4 Discussion
Currently, we have limited data about the lethal effects of acute and chronic acetamiprid exposure on bumblebees. Although we found slightly more publications on this topic than a recent review (Camp and Lehmann 2020), acetamiprid is still rarely the focus of ecotoxicological studies on Bombus species (also compared to those conducted on honeybees). Toxicity studies vary greatly in the testing duration, model species, and/or the form of acetamiprid applied that makes it difficult to directly compare their findings and draw general conclusions. Besides, the presentation of the observational data and the results of related statistical tests comparing the effects of different treatment doses to control is often lacking. While LD50 (with or without associated uncertainty) values are usually reported, their numerical value can be greatly influenced by factors such as the number, age or diet of animals tested, food deprivation prior to dosing, housing, or other experimental conditions; therefore, they should not be regarded as biological constants (Zbinden and Flury-Roversi 1981). Nevertheless, findings so far generally implied that present field-realistic concentrations of this neonicotinoid have a minor acute and chronic effect on bumblebee mortality irrespective of the form of application (i.e. oral or contact). On the other hand, chronic oral exposure to doses (2–3000 µg/L) that correspond to residue concentrations in flowers 4–6 days after the application of acetamiprid-containing pesticides (Stejskalova et al. 2018; Chandler et al. 2020) has been reported to cause altered behaviour in bumblebees (Camp et al. 2020b).
There are also only a few published studies on the sublethal effects of acetamiprid that indicates a large knowledge gap and highlight several areas for potential research. For instance, previous studies suggest that bumblebees can get addicted to neonicotinoid contaminated food (Kessler et al. 2015; Arce et al. 2018, but see Muth et al. 2020), but similar investigations have not been carried out on acetamiprid so far, so it is still unknown whether or not acetamiprid-treated food patches can become preferred by foraging workers. This is particularly important, because preference for contaminated food sources may influence navigation (Fischer et al. 2014; Stanley et al. 2016), foraging activity (Gill et al. 2012; Stanley et al. 2016), and level of exposure in individuals and also enhance sublethal effects in both individuals and colonies (Gill et al. 2012; Bryden et al. 2013). Previous experiments conducted on bumblebee microcolonies indicated that acetamiprid can have substantial sublethal effects, including colony size, food consumption, and drone production, when present in high concentrations (Camp et al. 2020a, b). A thorough investigation on queenright colonies in field conditions also implied that the reproductive output of colonies may become reduced at concentrations that are higher than those applied in the study (Van Oystaeyen et al. 2020). At the moment, these concentrations are not field-realistic, but can be present in flowers for days after pesticide application (Stejskalova et al. 2018; Chandler et al. 2020). If the use of acetamiprid-containing pesticides in agricultural production increases in the coming years as expected (also due to increasing pest resistance in the future; Bass et al. 2015), however, bumblebees may become exposed to such high concentrations regularly and/or for an extended period of time. Such high concentrations may be especially detrimental to mated queens prior to colony initiation during the spring (Baron et al. 2017) and lead to reduced foraging efficiency (Gill et al. 2012; Stanley et al. 2016) and compromised reproductive success within the established colony (Rundlöf et al. 2015; Woodcock et al. 2017). Since the degradation of acetamiprid is reported to be influenced by soil type as well (Gupta et al. 2008; Goulson 2013), the accumulation of acetamiprid residues in the soil may also contribute to prolonged exposures to this substance in some agroecosystems. The effect of analytical acetamiprid may differ from that of the commercially available products as well. For instance, the LD50 values for the Mospilan formulation reported by Wu et al. (2010) following chronic oral exposure were four orders of magnitude lower than for the technical acetamiprid estimated by other studies. It should also be noted, however, that this striking effect was reported only by a single study (where the product name and active substance content was erroneously indicated) and has not been confirmed since then. This issue certainly deserves more attention and experiments investigating the effects of both analytical acetamiprid and Mospilan formulation within the same experimental setup should be conducted to verify if additive substances in the Mospilan formulation are indeed responsible for such enhanced toxicity.
An important direction for future research is the investigation of the potential synergistic effects between acetamiprid and other pesticides at present and predicted field-realistic concentrations. Different pesticides are commonly applied simultaneously in field conditions, and the effect of other neonicotinoids is known to be highly dependent on what other pesticides are used with them (Wood and Goulson 2017). Although there is no convincing evidence to date that the toxicity of acetamiprid surpasses the benchmark values for toxicity according to USEPA (2014) when used together with other pesticides (Sanchez-Bayo and Goka 2014; Chandler et al. 2020; Reid et al. 2020), the number of potential synergies far exceeds the number of studies that scrutinized the combined lethal and sublethal effects of acetamiprid and other pesticides on bumblebees so far. The potential difference in sensitivity to acetamiprid between different Bombus species should also be investigated more in detail. The difference in sensitivity to other pesticides, including neonicotinoids, are known from previous works (Arena and Sgolastra 2014; Sgolastra et al. 2017), but a comprehensive study in which the various effects of acetamiprid is examined on different Bombus species within the same experimental setup is lacking. For instance, research on the sublethal effects of acetamiprid has mostly focused on two common bumblebee species, B. terrestris and B. impatiens. However, other Bombus species, including brood parasitic cuckoo bumblebees (subgenus Psithyrus) or cold-tolerant mountain species, are likely to react differently to acetamiprid than the above two species due to their difference in size, physiology, and/or life history parameters that potentially affect the level of exposure to insecticides or the metabolism of this particular substance. Furthermore, Crall et al. (2019) also reported that colony size is another important parameter that determines the sublethal consequences of pesticide exposure in B. impatiens colonies as larger colonies were found to be less sensitive to the neonicotinoid imidacloprid than smaller ones. Because of that, studies should also be conducted simultaneously on more than one Bombus species and with different colony sizes, so we could assess more realistically the risk that acetamiprid poses on natural bumblebee assemblages (Alkassab and Kirchner 2017).
5 Conclusions
The environmental risk assessment of plant protection products, including neonicotinoids, use only honeybees as indicator organisms and in accordance with that, the duration of the standard chronic oral toxicity test is defined for adult worker honeybees (10 days; EFSA 2013). Toxicity classification is the same for honeybees and bumblebees in the UK and the European Union (Lewis and Tzilivakis 2019), and no such classification exists for bumblebees in the USA (USEPA 2012). However, honeybees may be poor models for assessing the effects of pesticides on other pollinators such as bumblebees due to differences in body size, physiology, and social tendencies that may influence the level of exposure and subsequent intoxication (Goulson 2010; Arena and Sgolastra 2014; Stoner 2016; Franklin & Raine 2019; Gradish et al. 2019). To address this issue, the European Food Safety Authority (EFSA) is working on the revision of the “Guidance Document on the Risk Assessment of Plant Protection Products on Bees” to include risk assessments for bumblebees and solitary bees as well (More et al. 2021; EFSA 2021). We argue that this is an important step forward as currently available experimental results do not provide enough information to accurately evaluate the risk of acetamiprid exposure on bumblebees; especially its sub-lethal effects and the potential population-level consequences are unknown due to insufficient testing. Including measurements of colony fitness and foraging efficiency into risk assessment studies could provide a more complete view of the threats this substance poses to pollinators. The current protocols may lead to an overestimation of survival in both acute and chronic toxicity tests in bumblebees, so adding non-lethal endpoints to and/or extending the duration of these tests to capture all adverse effects of acetamiprid would be highly advisable (as also recommended for honeybees by Simon-Delso et al. 2018). Such extension is particularly important for correctly estimating the time-dependent effects of some compounds, particularly neonicotinoids and some systemic fungicides, under field conditions where bees are exposed to residues in pollen and nectar over their entire life cycle. Investigations on queenright colonies can provide more realistic information including population-level effects of acetamiprid than microcolonies, thus using the former should also be preferred whenever possible. As the concentrations of acetamiprid and its metabolites are likely to increase in agroecosystems across Europe in the coming years, we propose that more research is warranted if we are to better understand the long-term impacts of this insecticide on bumblebees and other non-target organisms.
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Alkassab, A.T., Kirchner, W.H. (2017) Sublethal exposure to neonicotinoids and related side effects on insect pollinators: honeybees, bumblebees and solitary bees. J. Plant. Dis. Prot. 124, 1–30. https://doi.org/10.1007/s41348-016-0041-0
Arce, A.N., Ramos-Rodrigues, A., Yu, J., Colgan, T.J., Wurm, Y., Gill, R.J. (2018) Foraging bumblebees acquire a preference for neonicotinoid-treated food with prolonged exposure. Proc. Royal. Soc. B. 285, 20180655. https://doi.org/10.1098/rspb.2018.0655
Arena, M., Sgolastra, F. (2014) A meta-analysis comparing the sensitivity of bees to pesticides. Ecotoxicology. 23, 324–334. https://doi.org/10.1007/s10646-014-1190-1
Baines, D., Wilton, E., Pawluk, A., de Gorter, M., Chomistek, N. (2017) Neonicotinoids act like endocrine disrupting chemicals in newly-emerged bees and winter bees. Sci. Rep. 7, 10979. https://doi.org/10.1038/s41598-017-10489-6
Baron, G.L., Jansen, V.A.A., Brown, M.J.F., Raine, N.E. (2017) Pesticide reduces bumblebee colony initiation and increases probability of population extinction. Nat. Ecol. Evol. 1, 1308–1316. https://doi.org/10.1038/s41559-017-0260-1
Bass, C., Denholm, I., Williamson, M.S., Nauen, R. (2015) The global status of insect resistance to neonicotinoid insecticides. Pestic. Biochem. Physiol. 121, 78–87. https://doi.org/10.1016/j.pestbp.2015.04.004
Blacquière, T., Smagghe, G., van Gestel, C.A.M., Mommaerts, V. (2012) Neonicotinoids in bees: a review on concentrations, side-effects and risk assessment. Ecotoxicology. 21, 973–992. https://doi.org/10.1007/s10646-012-0863-x
Botías, C., David, A., Horwood, J., Abdul-Sada, A., Nicholls, E., Hill, E., Goulson, D. (2015) Neonicotinoid residues in wildflowers, a potential route of chronic exposure for bees. Environ. Sci. Technol. 49, 12731–12740. https://doi.org/10.1021/acs.est.5b03459
Botías, C., Jones, J.C., Pamminger, T., Bartomeus, I., Hughes, W.O.H., Goulson, D. (2021) Multiple stressors interact to impair the performance of bumblebee Bombus terrestris colonies. J. Anim. Ecol. 90, 415–431. https://doi.org/10.1111/1365-2656.13375
Brunet, J.-L., Badiou, A., Belzunces, L.P. (2005) In vivo metabolic fate of [14C]-acetamiprid in six biological compartments of the honeybee, Apis mellifera L. Pest. Manag. Sci. 61, 742–748. https://doi.org/10.1002/ps.1046
Bryden, J., Gill, R.J., Mitton, R.A.A., Raine, N.E., Jansen, V.A.A. (2013) Chronic sublethal stress causes bee colony failure. Ecol. Lett. 16, 1463–1469. https://doi.org/10.1111/ele.12188
Calatayud-Vernich, P., Calatayud, F., Simó, E., Picó, Y. (2018) Pesticide residues in honeybees, pollen and beeswax: assessing beehive exposure. Environ. Pollut. 241, 106–114. https://doi.org/10.1016/j.envpol.2018.05.062
Cameron, S.A., Sadd, B.M. (2020) Global trends in bumble bee health. Annu. Rev. Entomol. 65, 209–232. https://doi.org/10.1146/annurev-ento-011118-111847
Camp, A.A., Batres, M.A., Williams, W.C., Koethe, R.W., Stoner, K.A., Lehmann, D.M. (2020a) Effects of the neonicotinoid acetamiprid in pollen on Bombus impatiens microcolony development. Environ. Toxicol. Chem. 39, 2560–2569. https://doi.org/10.1002/etc.4886
Camp, A.A., Lehmann, D.M. (2020) Impacts of neonicotinoids on the bumble bees Bombus terrestris and Bombus impatiens examined through the lens of an adverse outcome pathway framework. Environ. Toxicol. Chem. 40, 309–322. https://doi.org/10.1002/etc.4939
Camp, A.A., Williams, W.C., Eitzer, B.D., Koethe, R.W., Lehmann, D.M. (2020b) Effects of the neonicotinoid acetamiprid in syrup on Bombus impatiens (Hymenoptera: Apidae) microcolony development. PLoS ONE. 15, e0241111. https://doi.org/10.1371/journal.pone.0241111
Chandler, A.J., Drummond, F.A., Collins, J.A., Lund, J., Alnajjar, G. (2020) Exposure of the common eastern bumblebee, Bombus impatiens (Cresson), to sub-lethal doses of acetamiprid and propiconazole in wild blueberry. J. Agric. Urban. Entomol. 36, 1–23. https://doi.org/10.3954/1523-5475-36.1.1
Crall, J.D., de Bivort, B.L., Dey, B., Versypt, A.N.F. (2019) Social buffering of pesticides in bumblebees: agent-based modeling of the effects of colony size and neonicotinoid exposure on behavior within nests. Front. Ecol. Evol. 7, 51. https://doi.org/10.3389/fevo.2019.00051
Decourtye, A., Devillers, J. (2010) Ecotoxicity of neonicotinoid insecticides to bees. In: Thany, S.H. (ed) Insect Nicotinic Acetylcholine Receptors. Advances in Experimental Medicine and Biology, vol 683. Springer, New York, NY, pp 85–95. https://doi.org/10.1007/978-1-4419-6445-8_8
Donkersley, P., Rhodes, G., Pickup, R.W., Jones, K.C., Wilson, K. (2014) Honeybee nutrition is linked to landscape composition. Ecol. Evol. 4, 4195–4206. https://doi.org/10.1002/ece3.1293
Douglass, C., White, B.K., Sutton, C., Steeger, T., Holmes, J. (2017) Preliminary environmental fate and ecological risk assessment in support of the registration review of acetamiprid. https://www.regulations.gov/document/EPA-HQ-OPP-2012-0329-0026. Accessed 26 February 2021
EFSA. (2016) Peer review of the pesticide risk assessment of the active substance acetamiprid. EFSA. J. 14, e04610. https://doi.org/10.2903/j.efsa.2016.4610
EFSA. (2013) Guidance on the risk assessment of plant protection products on bees (Apis mellifera, Bombus spp. and solitary bees). EFSA. J. 11, 3295. https://doi.org/10.2903/j.efsa.2013.3295
EFSA. (2021) Public consultation on the draft EFSA Scientific Committee Opinion on a systems-based approach to the environmental risk assessment of multiple stressors in honey bees. In: European Food Safety Authority. https://www.efsa.europa.eu/en/consultations/call/public-consultation-draft-efsa-scientific-committee-opinion-1. Accessed 26 February 2021
EC. (2018a) European Commission Implementing Regulation (EU) 2018/783. Off. J. Eur. Union. 132, 31–34
EC. (2018b) European Commission Implementing Regulation (EU) 2018/784. Off. J. Eur. Union. 132, 35–39
EC. (2018c) European Commission Implementing Regulation (EU) 2018/785. Off. J. Eur. Union. 132, 40–44
EC. (2018d) European Commission Implementing Regulation (EU) 2018/113. Off. J. Eur. Union. 20, 7–10
Elbert, A., Haas, M., Springer, B., Thielert, W., Nauen, R. (2008) Applied aspects of neonicotinoid uses in crop protection. Pest. Manag. Sci. 64, 1099–1105. https://doi.org/10.1002/ps.1616
EPA. (2002) Name of chemical: acetamiprid. Reason for issuance: conditional registration. https://www3.epa.gov/pesticides/chem_search/reg_actions/registration/fs_PC-099050_15-Mar-02.pdf. Accessed 26 February 2021
Felton, J.C., Oomen, P.A., Stevenson, J.H. (1986) Toxicity and hazard of pesticides to honeybees: harmonization of test methods. Bee. World. 67, 114–124. https://doi.org/10.1080/0005772X.1986.11098883
Fijen, T.P., Scheper, J.A., Boom, T.M., Janssen, N., Raemakers, I., Kleijn, D. (2018) Insect pollination is at least as important for marketable crop yield as plant quality in a seed crop. Ecol. Lett. 21, 1704–1713. https://doi.org/10.1111/ele.13150
Fischer, J., Müller, T., Spatz, A.-K., Greggers, U., Grünewald, B., Menzel, R. (2014) Neonicotinoids interfere with specific components of navigation in honeybees. PLoS ONE. 9, e91364. https://doi.org/10.1371/journal.pone.0091364
Franklin, E.L., Raine, N.E. (2019) Moving beyond honeybee-centric pesticide risk assessments to protect all pollinators. Nat. Ecol. Evol. 3, 1373–1375. https://doi.org/10.1038/s41559-019-0987-y
Ghosh, S., Jung, C. (2017) A short review on neonicotinoids: use in crop protection and issues on honeybee and hive products. J. Apic. Sci. 32, 333–344. https://doi.org/10.17519/apiculture.2017.11.32.4.333
Gill, R.J., Ramos-Rodriguez, O., Raine, N.E. (2012) Combined pesticide exposure severely affects individual- and colony-level traits in bees. Nature. 491, 105–108. https://doi.org/10.1038/nature11585
Goulson, D. (2010) Bumblebees: behaviour, ecology, and conservation, 2nd edn. Oxford University Press, Oxford, New York
Goulson, D. (2013) An overview of the environmental risks posed by neonicotinoid insecticides. J. Appl. Ecol. 50, 977–987. https://doi.org/10.1111/1365-2664.12111
Goulson, D., Nicholls, E., Botias, C., Rotheray, E.L. (2015) Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science. 347, 1255957. https://doi.org/10.1126/science.1255957
Gradish, A.E., van der Steen, J.J.M., Scott-Dupree, C.D., Cabrera, A.R., Cutler, G.C., et al. (2019) Comparison of pesticide exposure in honey bees (Hymenoptera: Apidae) and bumble bees (Hymenoptera: Apidae): implications for risk assessments. Environ. Entomol. 48, 12–21. https://doi.org/10.1093/ee/nvy168
Gupta, S., Gajbhiye, V.T., Gupta, R.K. (2008) Effect of light on the degradation of two neonicotinoids viz acetamiprid and thiacloprid in soil. Bull. Environ. Contam. Toxicol. 81, 185–189. https://doi.org/10.1007/s00128-008-9405-x
Hutchinson, L.A., Oliver, T.H., Breeze, T.D., Bailes, E.J., Brünjes, L., et al. (2021) Using ecological and field survey data to establish a national list of the wild bee pollinators of crops. Agric. Ecosyst. Environ. 315, 107447. https://doi.org/10.1016/j.agee.2021.107447.
Iwasa, T., Motoyama, N., Ambrose, J.T., Roe, R.M. (2004) Mechanism for the differential toxicity of neonicotinoid insecticides in the honey bee, Apis mellifera. Crop. Prot. 23, 371–378. https://doi.org/10.1016/j.cropro.2003.08.018
Jeschke, P., Nauen, R., Schindler, M., Elbert, A. (2011) Overview of the status and global strategy for neonicotinoids. J. Agric. Food. Chem. 59, 2897–2908. https://doi.org/10.1021/jf101303g
Kerr, J.T., Pindar, A., Galpern, P., Packer, L., Potts, S.G., et al. (2015) Climate change impacts on bumblebees converge across continents. Science. 349, 177–180. https://doi.org/10.1126/science.aaa7031
Kessler, S., Tiedeken, E.J., Simcock, K.L., Derveau, S., Mitchell, J., Softley, S., Radcliffe, A., Stout, J.C., Wright, G.A. (2015) Bees prefer foods containing neonicotinoid pesticides. Nature. 521, 74–76. https://doi.org/10.1038/nature14414
Klein, A.-M., Vaissière, B.E., Cane, J.H., Steffan-Dewenter, I., Cunningham, S.A., Kremen, C., Tscharntke, T. (2007) Importance of pollinators in changing landscapes for world crops. Proc. Royal. Soc. B. 274, 303–313. https://doi.org/10.1098/rspb.2006.3721
Lentola, A., David, A., Abdul-Sada, A., Tapparo, A., Goulson, D., Hill, E.M. (2017) Ornamental plants on sale to the public are a significant source of pesticide residues with implications for the health of pollinating insects. Environ. Pollut. 228, 297–304. https://doi.org/10.1016/j.envpol.2017.03.084
Lewis, K.A., Tzilivakis, J. (2019) Wild bee toxicity data for pesticide risk assessments. Data. 4, 98. https://doi.org/10.3390/data4030098
Lu, C., Hung, Y.-T., Cheng, Q. (2020) A review of sub-lethal neonicotinoid insecticides exposure and effects on pollinators. Curr. Pollut. Rep. 6, 137–151. https://doi.org/10.1007/s40726-020-00142-8
Manjon, C., Troczka, B.J., Zaworra, M., Beadle, K., Randall, E., et al. (2018) Unravelling the molecular determinants of bee sensitivity to neonicotinoid insecticides. Curr. Biol. 28, 1137–1143. https://doi.org/10.1016/j.cub.2018.02.045
Martinet, B., Dellicour, S., Ghisbain, G., Przybyla, K., Zambra, E., Lecocq, T., Boustani, M., Baghirov, R., Michez, D., Rasmont, P. (2020) Global effects of extreme temperatures on wild bumblebees. Conserv. Biol. 35, 1507–1518. https://doi.org/10.1111/cobi.13685
Matsuda, K., Ihara, M., Sattelle, D.B. (2020) Neonicotinoid insecticides: molecular targets, resistance, and toxicity. Annu. Rev. Pharmacol. Toxicol. 60, 241–255. https://doi.org/10.1146/annurev-pharmtox-010818-021747
Mitchell, E.A.D., Mulhauser, B., Mulot, M., Mutabazi, A., Glauser, G., Aebi, A. (2017) A worldwide survey of neonicotinoids in honey. Science. 358, 109–111. https://doi.org/10.1126/science.aan3684
Moher, D., Liberati, A., Tetzlaff, J., Altman, D.G., the PRISMA Group* (2010) Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 8, 336–341. https://doi.org/10.1016/j.ijsu.2010.02.007
More, S.J., Auteri, D., Rortais, A., Pagani, S. (2021) EFSA is working to protect bees and shape the future of environmental risk assessment. EFSA. J. 19, e190101. https://doi.org/10.2903/j.efsa.2021.e190101
Muth, F., Gaxiola, R.L., Leonard, A.S. (2020) No evidence for neonicotinoid preferences in the bumblebee Bombus impatiens. R. Soc. Open. Sci. 7, 191883. https://doi.org/10.1098/rsos.191883
Naug, D. (2009) Nutritional stress due to habitat loss may explain recent honeybee colony collapses. Biol. Conserv. 142:2369–2372. https://doi.org/10.1016/j.biocon.2009.04.007
R Core Team. (2020) R: A language and environment for statistical computing. R version 3.6.3. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Reid, R.J., Troczka, B.J., Kor, L., Randall, E., Williamson, M.S., Field, L.M., Nauen, R., Bass, C., Davies, T.G.E. (2020) Assessing the acute toxicity of insecticides to the buff-tailed bumblebee (Bombus terrestris audax). Pestic. Biochem. Physiol. 166, 104562. https://doi.org/10.1016/j.pestbp.2020.104562
Reilly, J.R., Artz, D.R., Biddinger, D., Bobiwash, K., Boyle, N.K., et al. (2020) Crop production in the USA is frequently limited by a lack of pollinators. Proc. Royal. Soc. B. 287, 20200922. https://doi.org/10.1098/rspb.2020.0922
Rundlöf, M., Andersson, G.K.S., Bommarco, R., Fries, I., Hederström, V., et al. (2015) Seed coating with a neonicotinoid insecticide negatively affects wild bees. Nature. 521, 77–80. https://doi.org/10.1038/nature14420
Sanchez-Bayo, F., Goka, K. (2014) Pesticide residues and bees – a risk assessment. PLoS. ONE. 9, e94482. https://doi.org/10.1371/journal.pone.0094482
Sanchez-Bayo, F., Tennekes, H.A. (2020) Time-cumulative toxicity of neonicotinoids: experimental evidence and implications for environmental risk assessments. J. Environ. Res. Public. Health. 17, 1629. https://doi.org/10.3390/ijerph17051629
Sgolastra, F., Medrzycki, P., Bortolotti, L., Renzi, M.T., Tosi, S., Bogo, G., Teper, D., Porrini, C., Molowny-Horas, R., Bosch, J. (2017) Synergistic mortality between a neonicotinoid insecticide and an ergosterol-biosynthesis-inhibiting fungicide in three bee species. Pest. Manag. Sci. 73, 1236–1243. https://doi.org/10.1002/ps.4449
Simon-Delso, N., Amaral-Rogers, V., Belzunces, L.P., Bonmatin, J.M., Chagnon, M., et al. (2015) Systemic insecticides (neonicotinoids and fipronil): trends, uses, mode of action and metabolites. Environ. Sci. Pollut. Res. 22, 5–34. https://doi.org/10.1007/s11356-014-3470-y
Simon-Delso, N., San Martin, G., Bruneau, E., Hautier, L. (2018) Time-to-death approach to reveal chronic and cumulative toxicity of a fungicide for honeybees not revealed with the standard ten-day test. Sci. Rep. 8, 7241. https://doi.org/10.1038/s41598-018-24746-9
Stanley, D.A., Garratt, M.P.D., Wickens, J.B., Wickens, V.J., Potts, S.G., Raine, N.E. (2015) Neonicotinoid pesticide exposure impairs crop pollination services provided by bumblebees. Nature. 528, 548–550. https://doi.org/10.1038/nature16167
Stanley, D.A., Russell, A.L., Morrison, S.J., Rogers, C., Raine, N.E. (2016) Investigating the impacts of field-realistic exposure to a neonicotinoid pesticide on bumblebee foraging, homing ability and colony growth. J. Appl. Ecol. 53, 1440–1449. https://doi.org/10.1111/1365-2664.12689
Stejskalová, M., Konradyová, V., Suchanová, M., Kazda, J. (2018) Is pollinator visitation of Helianthus annuus (sunflower) influenced by cultivar or pesticide treatment? Crop. Prot. 114, 83–89. https://doi.org/10.1016/j.cropro.2018.08.018
Stoner, K.A. (2016) Current pesticide risk assessment protocols do not adequately address differences between honey bees (Apis mellifera) and bumble bees (Bombus spp.). Front. Environ. Sci. 4, 79. https://doi.org/10.3389/fenvs.2016.00079
Switzer, C.M., Combes, S.A. (2017) Bumblebee sonication behavior changes with plant species and environmental conditions. Apidologie. 48, 223–233 https://doi.org/10.1007/s13592-016-0467-1
Takahashi, H., Mitsui, J., Takakusa, N., Matsuda, M., Yoneda, H., Suzuki, J., Ishimitsu, K., Kishimoto, T. (1992) NI-25 a new type of systemic and broad spectrum insecticide. In: Proceedings, Brighton Crop Protection Conference, Pests and Diseases, 1992 Brighton, November 23–26, 1992. (pp. 89–96) British Crop Protection Council
USEPA. (2012) Ecological effects test guidelines OCSPP 850.3030: honeybee toxicity of residues on foliage. U.S. Environmental Protection Agency, Prevention, Pesticides and Toxic Substances. https://www.regulations.gov/document/EPA-HQ-OPPT-2009-0154-0017. Accessed 27 February 2021
USEPA. (2014) Guidance for assessing pesticide risks to bees. Office of Chemical Safety and Pollution Prevention Office of Pesticide Programs Environmental Fate and Effects Division, Environmental Protection Agency, Washington DC; Environmental Assessment Directorate, Pest Management Regulatory Agency, Health Canada, Ottawa, CN; California Department of Pesticide Regulation. https://www.epa.gov/sites/production/files/2014-06/documents/pollinator_risk_assessment_guidance_06_19_14.pdf. Accessed 27 February 2021
Van Oystaeyen, A., Klatt, B.K., Petit, C., Lenaerts, N., Wäckers, F. (2020) Short-term lab assessments and microcolonies are insufficient for the risk assessment of insecticides for bees. Chemosphere. 273, 128518. https://doi.org/10.1016/j.chemosphere.2020.128518
Whitehorn, P.R., Wallace, C., Vallejo-Marin, M. (2017) Neonicotinoid pesticide limits improvement in buzz pollination by bumblebees. Sci. Rep. 7, 15562. https://doi.org/10.1038/s41598-017-14660-x
Wood, T.J., Goulson, D. (2017) The environmental risks of neonicotinoid pesticides: a review of the evidence post 2013. Environ. Sci. Pollut. Res. 24, 17285–17325. https://doi.org/10.1007/s11356-017-9240-x
Woodcock, B.A., Bullock, J.M., Shore, R.F., Heard, M.S., Pereira, M.G., et al. (2017) Country-specific effects of neonicotinoid pesticides on honey bees and wild bees. Science. 356, 1393–1395. https://doi.org/10.1126/science.aaa1190
Wu, J., Li, J.-L., Peng, W.-J., Hu, F.-L. (2010) Sensitivities of three bumblebee species to four pesticides applied commonly in greenhouses in China. Insect. Sci. 17, 67–72. https://doi.org/10.1111/j.1744-7917.2009.01286.x
Zbinden, G., Flury-Roversi, M. (1981) Significance of the LD50-test for the toxicological evaluation of chemical substances. Arch. Toxicol. 47, 77–99. https://doi.org/10.1007/BF00332351
Zioga, E., Kelly, R., White, B., Stout, J.C. (2020) Plant protection product residues in plant pollen and nectar: a review of current knowledge. Environ. Res. 189, 109873. https://doi.org/10.1016/j.envres.2020.109873
Acknowledgements
Zsófia Varga-Szilay would like to thank Gabor Pozsgai for his advice and comments on the first draft of the manuscript. Zsófia Varga-Szilay was supported by the ÚNKP-20-3 New National Excellence Program of the Ministry for Innovation and Technology from the source of the National Research, Development and Innovation Fund. Zoltán Tóth was financially supported by the Prémium postdoctoral research programme of the Hungarian Academy of Sciences (MTA, PREMIUM-2018-198).
Funding
Open access funding provided by ELKH Centre for Agricultural Research. Zsófia Varga-Szilay was supported by the ÚNKP-20-3 New National Excellence Program of the Ministry for Innovation and Technology from the source of the National Research, Development and Innovation Fund. Zoltán Tóth was financially supported by the Prémium postdoctoral research programme of the Hungarian Academy of Sciences (MTA, PREMIUM-2018-198).
Author information
Authors and Affiliations
Contributions
ZVS and ZT conceived the idea of review; ZVS and ZT participated in the analysis and interpretation of the data; ZVS and ZT wrote the paper. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
This is a review study, so no ethical approval was required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Manuscript editor: Monique Gauthier
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Varga-Szilay, Z., Tóth, Z. Is acetamiprid really not that harmful to bumblebees (Apidae: Bombus spp.)?. Apidologie 53, 2 (2022). https://doi.org/10.1007/s13592-022-00909-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13592-022-00909-6