Abstract
Hidradenitis suppurativa (HS) is a debilitating chronic skin disorder characterized by painful inflammatory nodules, abscesses and sinus tracts involving intertriginous areas and has an adverse impact on patient quality of life. Over the past decade, the therapeutic options of HS have increased significantly to comprise multiple modalities, including topical medication, systemic therapies (mainly antibiotics, retinoids, and biologics), surgical approaches, and lifestyle modifications. Biologics alone or in combination with surgery remain the treatment of choice for moderate to severe disease. However, non-biologic therapies (including retinoids) may be used as monotherapy for mild disease and in combination with biologics and surgical treatment in moderate to severe disease. Retinoids, specifically isotretinoin, acitretin, and alitretinoin, are historically used in the management of HS, supported by anecdotal evidence and with variable treatment response. Although the current American and European guidelines offer different recommendations on the use of retinoids in HS, retinoids remain a valuable ally in HS management. This review provides a comprehensive analysis of the current scientific literature on retinoid therapy (topical and systemic) in HS, highlighting disparities in mechanisms of action, efficacy, and safety to clarify their role in HS treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Retinoids (isotretinoin, acitretin, and alitretinoin) are included in the pharmacopeia of hidradenitis suppurativa (HS). |
Retinoids can be used as monotherapy for mild disease and in combination with biologics and surgical treatment in moderate to severe disease. |
American and European guidelines offer different recommendations on the use of retinoids in HS. |
More robust data about efficacy and safety in patients with HS are needed. |
Introduction
Hidradenitis suppurativa (HS) is an immune-mediated, inflammatory, chronic, and recurrent skin disorder characterized by painful, deep-seated, inflamed lesions including abscesses, nodules, and tunnels, with predilection for intertriginous areas such as axillary, inguinal, and anogenital regions [1]. This condition has a significant negative impact on the well-being of both patients and their families [2]. The exact etiology of HS remains elusive, but recent research suggests a multifactorial causation involving genetic and environmental factors, such as smoking, obesity, and overweight status [3]. Approximately 30–40% of patients with HS have a familial predisposition, and specific gene mutations have been implicated in the disease [4]. HS is characterized by hair follicle obstruction, inflammation, bacterial infection, and immune dysregulation, which culminate in abscess formation and tissue damage [5, 6]. Aberrant glandular function and epidermal alterations also contribute to the pathogenesis [7, 8]. Diagnosis of HS is centered primarily on clinical evaluation, based on the identification of characteristic HS lesions with a predilection for inverse body regions and a tendency to the recurrence [9].
Disease severity is commonly assessed using various scoring systems, including Hurley score, International Hidradenitis Suppurativa 4 score (IHS4), Hidradenitis Suppurativa Severity Index (HSSI), Dermatology Life Quality Index (DLQI), and visual analog scale (VAS) [10].
In addition, high-frequency ultrasound has emerged as a valuable tool in clinical practice for confirming HS diagnoses, assessing disease severity, and monitoring treatment response [11].
The management of HS is challenging because of the limited effectiveness of available therapies and frequent exacerbations [12]. Current medical approaches to HS comprise a range of treatment options, including topical and systemic interventions. Topical treatments mainly include the use of antiseptic washes, topical antibiotics, and 15% resorcinol cream [13, 14]. Systemic therapy for moderate-to-severe HS primarily involves oral and intravenous antibiotics administered alone (e.g., tetracyclines, ertapenem, dalbavancin) or in combination (clindamycin, rifampicin) [15,16,17].
Other viable options include retinoids, dapsone, oral zinc, contraceptive agents, and immunomodulators used alone or in combination [18, 19].
Using long-pulsed neodymium-doped yttrium aluminum garnet or alexandrite lasers, laser hair removal effectively targets hair follicles in HS patients to reduce chronic inflammation and sinus tracts [20].
In the last decades, the introduction of biologic therapy has radically changed the therapeutic armamentarium of several dermatological conditions, particularly psoriasis and HS, by reducing the inflammatory and oxidative burden [21,22,23]. Adalimumab and secukinumab are currently the only biologic agents approved for adolescents and adults with moderate-to-severe HS [24,25,26].
Surgical approach mainly includes incision and drainage for acute flares, de-roofing, narrow margin excision, and wide local excision that can result in a disease-free state [27].
Retinoids have a strong pathophysiological rationale for use in HS by regulating the processes of proliferation, differentiation, and keratinization in epidermal cells [28]. Retinoids are a group of substances derived from vitamin A or having structural and/or functional similarities to vitamin A that interact with different classes of proteins, such as retinoid-binding proteins and retinoid nuclear receptors. This interaction activates specific regulatory segments of DNA, known as retinoic acid response elements, which play a crucial role in controlling cell growth, differentiation and apoptosis [29].
The classification of these compounds into three generations is based on their molecular structures. Isotretinoin and alitretinoin are categorized as first-generation retinoids, whereas acitretin belongs to the second generation [30]. Several studies have investigated the utilization of isotretinoin, alitretinoin, and acitretin in the treatment of HS. According to the North American guidelines, acitretin and isotretinoin are considered second- or third-line treatment options, respectively. Conversely, the European guidelines suggest only the use of acitretin for managing HS [31]. While the existing literature provides more consistent information on isotretinoin and acitretin, there is a limited number of studies regarding the use of alitretinoin in HS. The purpose of this review is to summarize the role of retinoids in the management of HS, highlighting the evidence and disparities in terms of mechanism of action, efficacy, and safety between the different class of retinoids.
Methods
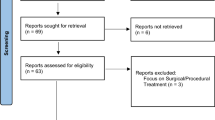
Bibliographic searches for qualitative review were conducted in PubMed up to November 20, 2023, with no date limits, using the terms: (hidradenitis suppurativa OR acne inversa) AND (retinoid) (isotretinoin), (acitretin), (alitretinoin), (tretinoin), (adapalene), (tazarotene), (trifarotene), and (bexarotene). One author (HG) initially screened all titles and abstracts and excluded articles that were clearly ineligible. Reports and cases were excluded if clinical details were lacking. When eligibility was in doubt, another author (EM) was involved. Articles were limited to those in English language. Full texts of the included articles were reviewed, and reference lists were manually searched and were checked for additional sources. A total of 16 articles, consisting of 692 patients, were included in this review. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. Ethical approval was not required.
Isotretinoin
Oral isotretinoin (13-cis retinoic acid) is a derivative of vitamin A employed for the treatment of resistant, nodular acne unresponsive to conventional therapies. It finds application in the treatment of various dermatologic conditions, including moderate acne, rosacea, folliculitis, facial pyoderma, HS, and cutaneous T-cell lymphomas. In addition, it has demonstrated efficacy in non-dermatologic conditions such as neuroblastoma [32]. Daily isotretinoin doses typically range from 0.5 to 1 mg/kg. Notably, isotretinoin is highly teratogenic at any dose. Thus, it is contraindicated during pregnancy, and it is crucial to take measures to prevent pregnancy both during isotretinoin treatment and for 1 month after treatment discontinuation [33].
In sebocytes, the prodrug isotretinoin is transformed into its biologically active form, all-trans retinoic acid (ATRA), which subsequently interacts with retinoid receptors. The primary mechanism underlying the pathophysiology of HS involves the occlusion and subsequent inflammation of hair follicles as well as the dysregulation of both innate and adaptive immune responses [34].
Early HS lesions usually show an altered keratinization of the hair follicle that results in follicular occlusion and consequent follicular dilatation and rupture leading to the release of keratin and bacteria into the adjacent dermal tissue. This process triggers an intense inflammatory response from neutrophils and lymphocytes leading to abscess formation and subsequent pilosebaceous unit destruction [35, 36].
The therapeutic efficacy of isotretinoin can be attributed to its impact on the pathophysiologic processes underlying HS, mainly through its ability to influence keratinocyte differentiation and inflammation. Regulation by isotretinoin of genes related to keratinocyte differentiation and programmed cell death, such TNF-related apoptosis-inducing ligand (TRAIL), may help normalize the differentiation process, potentially reducing follicular occlusion. [34].
Isotretinoin also has anti-inflammatory and immunomodulatory properties that could positively impact the disease course. Several studies have demonstrated that isotretinoin treatment may lead to a decrease in pro-inflammatory cytokines, such as tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), and interleukin-1 alpha (IL-1α), which are key contributors to the inflammatory response characterizing HS lesions [32, 37].
Moreover, it has been observed that isotretinoin can reduce Th17 responses, resulting in decreased interleukin (IL)-17 production, a known cytokine implicated in the pathogenesis of HS [35, 38].
In addition, isotretinoin’s capacity to promote regulatory T cell (Treg) immunosuppressive responses might further contribute to tempering the immune-driven inflammation observed in HS [38, 39].
In the management of acne, the primary role of isotretinoin is to decrease sebum production and minimize the size of sebaceous glands by promoting the apoptosis of sebocytes. This complex process involves interactions with several factors such as TRAIL, neutrophil gelatinase-associated lipocalin (NGAL), and forkhead box transcription factor (FoxO) proteins. Reducing sebum production creates a less favorable environment for bacterial growth, thus reducing inflammation and the risk of secondary infection, which could affect HS lesions [34].
However, this is true for acne, where isotretinoin reduces the ductal population of Cutibacterium acnes, implicated in its pathogenesis, but not for patients with HS, where this bacterium has not been found in either HS lesions or perilesional skin [40, 41].
Although HS mainly affects hair follicles and apocrine sweat glands, a reduction in the size and number of sebaceous glands in regions such as the axilla or groin has been observed in patients with HS [42].
Therefore, the property of isotretinoin to further reduce the size of sebaceous glands could potentially worsen or trigger HS acute flare, as reported in the literature [43,44,45,46,47].
De Vita et al. showed a possible association between the activity of the mammalian target of rapamycin 1 (mTORC1) complex and the response to isotretinoin treatment in HS patients, suggesting that higher mTORC1 activity may be associated with a better response to isotretinoin [48]. The mTORC1 complex is involved in multiple cellular functions, including survival, growth, proliferation, motility, and immune response, and has been implicated in inflammatory diseases such as HS, psoriasis, and acne. It plays a role in promoting T helper 17 (Th17) differentiation and subsequent IL-17 production, which is increased in HS lesions. The effect of isotretinoin on mTORC1 involves an increase in the nuclear level of FoxO1, which inhibits mTORC1 activity. In addition, increased mTORC1 signaling correlates with HS severity, obesity, and insuline resistance, which may lead to a better response to isotretinoin in patients with higher BMI [48, 49]. Furthermore, mTOR gene expression was also reduced after adalimumab (a common anti TNF-α used in HS treatment) therapy in HS lesional skin, suggesting the link between mTOR pathway and TNF-α [50].
It is important to select a phenotype that may respond to isotretinoin treatment, such as patients with Hurley I and II who have acne vulgaris as the primary cutaneous manifestation [46, 51,52,53].
In a retrospective study involving 358 patients with HS, the efficacy of oral isotretinoin was evaluated through patient self-assessment. The results revealed that 6.9% of participants reported a worsening of their HS condition, while 77% indicated no appreciable effect and only 16.1% reported improvement [47].
More promising results were reported in a retrospective study of 25 patients with HS treated with isotretinoin, in which 36% of patients achieved a complete response, 32% had a partial response, and the remaining 32% had no response. A more significant treatment response was noted in patients with a lower BMI, younger age, female gender, milder disease severity (Hurley stages I and II), and a higher incidence of acne [53].
In a retrospective study involving 68 patients with HS who received oral isotretinoin monotherapy for 4–6 months, the effectiveness of their treatment response during follow-up was assessed. Of these patients, 16 (23.5%) achieved a complete response, and 25 (36.8%) achieved a partial response. Notably, among the 16 patients who attained a complete response, this status was maintained for a duration ranging from 6 to 107 months, with an average of 57 months during the follow-up period [54].
Similarly, in a retrospective study Patel et al. analyzed the response rate to isotretinoin in 39 patients with HS. Fourteen patients (35.9%) showed a favorable response, while 25 (64.1%) showed no response. In addition, a significant correlation was observed between a previous occurrence of pilonidal cyst and a favorable response to isotretinoin. In contrast, factors such as sex, age of disease onset, BMI, Hurley stage, and history of acne (cystic or noncystic) did not significantly influence response to treatment [55].
In a retrospective study of 31 patients, the efficacy of combination therapies for the treatment of HS was evaluated. The combination isotretinoin-spironolactone showed positive results for patients with mild and moderate disease (according to IHS4 score) while isotretinoin/adalimumab only in moderate disease. In addition, other combinations that have shown favorable clinical responses in moderate-to-severe disease are isotretinoin/intralesional steroids and isotretinoin/topical benzoyl peroxide [56].
Additionally, two cases of patients with acne vulgaris who developed isotretinoin-induced sacroiliitis and isotretinoin-induced HS have been documented in the literature [57].
There have been several reports of adverse psychiatric reactions to isotretinoin. A retrospective study revealed that nine of ten people affected by bipolar disorder (BD) in treatment with isotretinoin experienced a significant worsening of mood symptoms, and three developed suicidal ideation [58]. Since there is evidence of the association between HS and BD, a different drug must be considered in these patients [59].
In summary, the data in the literature regarding the use and effectiveness of isotretinoin as a therapeutic strategy in patients with HS is inconsistent and lacks strong support.
Acitretin
Acitretin is a second-generation retinoid approved by the Food and Drug Administration for the treatment of severe, pustular, generalized, and localized plaque psoriasis. It is also used off-label for the management of conditions such as Darier’s disease, Grover’s disease, pityriasis rubra pilaris, lamellar ichthyosis, lichen planus, and lupus erythematosus [60, 61].
The initial dose of acitretin, which ranges from 10 to 75 mg daily, must be individually adjusted to achieve an optimal therapeutic response with tolerable side effects [62].
Acitretin is not recommended in women with reproductive potential, and contraception should be maintained for 3 years after discontinuation of treatment because of its long-term teratogenic potential [63].
Acitretin has a broad mechanism of action with anti-proliferative and anti-inflammatory effects. It works by interacting with nuclear retinoic acid receptors (RARs) to regulate gene transcription, resulting in a reduction of abnormal epidermal proliferation and better regulation of keratinocyte differentiation [64].
In addition, acitretin exerts its anti-inflammatory effects by regulating the expression of pro-inflammatory cytokines, including IL-6, migration inhibitory protein-related factor-8 (MRP-8), and interferon-gamma (IFN-γ). These biologic mechanisms support its use in the treatment of psoriasis and provide the rationale for its potentially effective use in HS [60, 64].
Moreover, by targeting the keratinization and inflammatory pathways, it directly addresses the key pathogenic mechanisms of HS, such as follicular obstruction and inflammation. Acitretin appears to be able to reduce IL-36β and IL-36γ expression induced by IL-17A stimulation at both the gene and protein levels [65].
Although IL-17 is a cytokine widely recognized for its significant involvement in HS pathogenesis, recent research has also revealed the contribution of IL-36, in particular IL-36α, -β, and -γ, together with the increased expression of IL-36RA in HS lesions compared to healthy skin [60].
In a study involving 12 patients with HS treated with acitretin, an overall improvement in the entire group was observed, along with a reduction in pain severity measured using the VAS score. Among them, nine patients achieved marked or complete remission after a single cycle of acitretin treatment, while the remaining three patients experienced mild to moderate improvements. The initial improvement was registered within 2 months, with further progress occurring within the first 6 months of therapy. Remarkably, nine patients maintained long-term improvement with no disease flares, ranging from 6 months to over 4 years. Adverse effects were observed in all 12 patients, with cheilitis being the primary side effect [66].
Encouraging results were also reported in a prospective open-label study that involved 17 patients with moderate to severe HS receiving acitretin treatment for a duration of up to 9 months, with clinical assessments conducted every 3 months after the initial month of therapy. Improvement was evident within the first month and persisted in subsequent months. Eight patients (47%) met the response criteria, demonstrating a reduction of ≥ 50% in HSSI score compared to baseline. However, a high drug discontinuation rate of 47% was also noted among patients, primarily due to treatment ineffectiveness and adverse events [67].
In a retrospective study involving 63 patients with moderate to severe HS, the efficacy of acitretin was evaluated. A significant reduction in the IHS4 score was observed at 12 and 24 weeks. All patients transitioned from an IHS4 score of 7.28–5.17 at 12 weeks, but only 35 patients reached an IHS4 value of 4.32 at 24 weeks. However, in a gender-stratified analysis, the reduction in IHS4 scores was not consistent across the two groups. Specifically, male patients, who initially had a higher baseline IHS4 score, showed a significant decrease in their IHS4 scores over time. In contrast, female patients, who started with lower baseline IHS4 scores, did not demonstrate significant improvements over time. Factors such as a higher baseline IHS4 score, a family history of HS, the presence of a follicular phenotype, and a history of follicular plugging conditions like acne conglobate emerged as potential indicators of treatment response. Acitretin has demonstrated a relatively low risk of severe side effects, with hypertriglyceridemia being the most frequent, although it typically did not result in treatment discontinuation [68].
Less favorable results emerged from a retrospective study involving 14 patients with HS, mainly with moderate to severe HS, who were poorly responsive to standard systemic treatments. Acitretin was administered as monotherapy in six patients (43%) and as an adjuvant therapy to other systemic medications (infliximab, moxifloxacin, cyclosporine, trimethoprim and sulfamethoxazole, rifampin, clindamycin, clarithromycin, ciprofloxacin, cephalexin, prednisone, spironolactone) in eight patients (57%). The results showed that acitretin monotherapy did not lead to clinical improvements among the patients. Conversely, in the group receiving acitretin as an adjuvant therapy, most (87.5%) experienced clinical improvements, with 50% achieving a good response and 37.5% showing a partial response. It is important to note that the patients in this study received a lower dose of acitretin (≤ 25 mg/day) for a shorter period compared to previous studies [69].
The use of acitretin as an adjunct therapy was also explored in a retrospective study that focused on patients with HS. These patients followed a treatment regimen that combined macrolides (azithromycin 500 mg daily for 3 days for 4 weeks) with acitretin (25–35 mg) during the acute phase. This was followed by subsequent monotherapy with acitretin for at least 9 months. The results were promising, with most patients (14 out of 15) reporting a positive outcome, including a decrease in IHS4 scores, improved DLQI, and reduced pain VAS scores after 9 months of treatment. One patient experienced partial improvement [70].
The most effective strategy to minimize recurrences in chronic and extensive cases of the HS disease involves the excision of the affected area, encompassing the surrounding apocrine glandular zone and extending 2 cm beyond the affected area. In accordance with this, Puri et al. conducted a study involving 30 patients divided into two groups. In Group I, a 12-week regimen of oral acitretin monotherapy was administered, while Group II underwent a combined therapy of oral acitretin and wide surgical excision. Notably, at the 6-month follow-up, the recurrence rate was significantly lower in Group II (20%) compared to Group I (40%), confirming acitretin as a good option for combined treatment [71].
In the literature, two cases of successful acitretin use in HS have been described. The first case involves an 11-month-old Chinese child with early-onset HS due to a PSENEN gene mutation. Initial treatments with topical and systemic antibiotics and topical retinoids were ineffective, but acitretin therapy led to significant improvement with minimal adverse effects [72]. In the second case, a patient affected by both HS and Darier’s disease successfully managed both dermatologic conditions with acitretin [73].
Acitretin therapy is known for increasing triglyceride serum level. Furthermore, acitretin could affect glucose metabolism and insulin resistance depending on the dose and duration of therapy. It is important to monitor serum glucose and lipids before and during therapy of patients with comorbidities [74].
Alitretinoin
Alitretinoin (the 9-cis isomer of retinoic acid) is a derivative of vitamin A that has found applications both topically and systemically in the treatment of severe acne vulgaris, psoriasis, specific types of cancer such as Kaposi’s sarcoma, and chronic hand eczema that does not respond to traditional topical steroid therapies. The standard dosage of alitretinoin ranges from 10 to 30 mg/day [75].
Alitretinoin acts as a pan-agonist at retinoid receptors, binding to all six known retinoid receptors, with a pronounced preference for RARs over retinoid x receptors (RXRs). Research has demonstrated its capacity to inhibit cellular proliferation and prompt apoptosis. Moreover, alitretinoin exerts anti-inflammatory effects by suppressing the generation of nitric oxide induced by cytokines or lipopolysaccharides, along with the inhibition of other proinflammatory molecules like TNF-α, IL-1β, and IL-12p40 [76].
In the literature, there is limited evidence regarding the use of alitretinoin in HS patients. Verdolini et al. conducted a study on 14 patients of childbearing age with HS that did not respond to previous conventional treatments. These patients were subsequently treated with alitretinoin at a dose of 10 mg/day for 24 weeks. Surprisingly, a significant improvement was observed in both Sartorius scores and DLQI in 78.5% of cases [77].
Alitretinoin is a teratogenic drug and shares a pharmacologic mechanism similar to acitretin. However, alitretinoin has a significantly shorter half-life, lasting only 4 weeks, compared to acitretin’s extended half-life, which can extend up to 3 years. This characteristic makes alitretinoin a considerably more appealing choice for women of childbearing age who wish to avoid unplanned pregnancies [78].
Ultimately, alitretinoin was successfully administered to a patient with keratitis-ichthyosis-deafness (KID) syndrome, dissecting cellulitis of the scalp, and HS that had shown resistance to conventional treatments and to other oral retinoids, including both acitretin and isotretinoin. Remarkably, alitretinoin led to improvements in both dissecting cellulitis of the scalp and HS [79].
Topical Retinoids
Topical retinoids consist of six distinct classes of compounds, each with unique properties and applications. These classes include tretinoin, adapalene, tazarotene, trifarotene, alitretinoin, and bexarotene. Over the past few decades, the use of topical retinoids has expanded to encompass a wide range of dermatological conditions. Approved indications include acne vulgaris, psoriasis, photoaging, cutaneous T-cell lymphoma, and Kaposi’s sarcoma. In addition, these agents are used off-label in the management of other conditions such as keratosis pilaris, actinic keratosis, melasma, and solar lentigines [80].
As systemic retinoids, topical retinoids enhance the turnover of follicular epithelial cells, facilitating the elimination of mature comedones and suppressing the formation of microcomedones. Moreover, they regulate the activity of retinoid-responsive genes, including those associated with cell proliferation and inflammation [81].
The use of topical retinoids during pregnancy is generally discouraged. Specifically, tretinoin and adapalene are categorized as class C, while tazarotene falls under class X. Nevertheless, a systematic meta-analysis involving 654 pregnant women in their first trimester who were exposed to topical retinoids showed no significant increase in the risk of major congenital malformations, spontaneous abortions, stillbirths, elective terminations, low birthweight, or prematurity in this group. Although this provides some reassurance, the study does not definitively support the use of topical retinoids during pregnancy because of statistical limitations, emphasizing caution in their use during this period [82].
Research on topical retinoid exposure during the second and third trimesters has not indicated a significant risk; however, caution is warranted when treating extensive body surface areas [83].
Data on the use of topical retinoids in HS are limited. In the literature, only one case report describes the successful use of topical retinoids in HS. The case involves a 12-year-old girl with HS who was treated with tretinoin cream. This topical treatment was administered in association with other topical therapies, including chlorhexidine wash and clindamycin solution, as well as systemic treatment with oral doxycycline [84].
A second case report discusses the use of tretinoin cream in a patient with dissecting cellulitis of the scalp. In this case, topical application of tretinoin cream was successful in controlling disease progression, preventing scarring alopecia, and preventing nodule formation [85].
Anecdotal cases about the use of adapalene in monotherapy (1 patient) and in combination with benzoyl peroxide (2 patients) can be found in the literature, but there is no information regarding its efficacy [46, 56] (Table 1).
Discussion and Conclusions
The management of HS represents a significant therapeutic challenge, and ongoing studies are currently underway to identify effective treatments. One of the explored approaches involves the use of retinoids, particularly isotretinoin, acitretin, and alitretinoin. However, the scientific and medical community is divided on the effectiveness of these treatments.
An interesting aspect is the difference between North American and European guidelines regarding the use of isotretinoin and acitretin. While North American guidelines recommend the use of isotretinoin only in specific situations, such as severe acne concurrent with severe HS, European guidelines suggest the use of acitretin in early and chronic stages of HS, while discouraging the use of isotretinoin due to disappointing results in the scientific literature [6, 12]. This underscores the need for further studies aimed to clarify the effectiveness of these treatments.
A recent study evaluated the pharmacologic survival of oral retinoids in patients with HS. This study revealed that, although isotretinoin and acitretin were associated with partial improvement of HS lesions, inefficacy remains the primary cause of treatment discontinuation. Some patients, particularly those with the folliculitis scar phenotype, experienced a more prolonged drug survival duration with acitretin therapy compared with those with the typical phenotype. These results indicate that the effectiveness of retinoids can vary significantly among patients and may be influenced by specific clinical characteristics [51].
An important aspect to consider is that while acitretin appears to demonstrate favorable results in HS management, isotretinoin might contribute to exacerbating the disease because of its action in reducing sebaceous glands, which are already diminished in size and number in HS. This difference in the mechanisms of action of retinoids raises important questions about the applicability of these treatments. In fact, in similar disease, such as acne fulminans, where isotretinoin can cause a clinical flare, the off-label use of anti-TNF-α was recommended [87]. However, both isotretinoin and alitretinoin could represent valuable supportive treatment options in combination with other approaches for HS, such as antibiotics, TNF-α inhibitors, spironolactone, and extensive surgical excision. As for alitretinoin, the body of evidence currently available is limited, with only one study assessing its efficacy in women of childbearing age. Consequently, further research is needed before considering alitretinoin a valid treatment option for HS, particularly in this specific patient group. Currently, in the literature, there are no data related to retinoids not mentioned in this review. Furthermore, there are no ongoing clinical studies on the use of retinoids other than those mentioned [88].
In conclusion, the choice of treatment for HS should be based on a comprehensive assessment of the individual patient’s characteristics and the severity of the disease. Further clinical studies and research are required to establish clear guidelines on the use of retinoids in HS and determine which patients may derive the most benefit from these treatments. Additionally, the long-term safety and efficacy of these treatments require more in-depth evaluations to ensure optimal care for HS patients.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Molinelli E, Gioacchini H, Sapigni C, Diotallevi F, Brisigotti V, Rizzetto G, et al. New insight into the molecular pathomechanism and immunomodulatory treatments of hidradenitis suppurativa. Int J Mol Sci. 2023;24:8428.
Chernyshov PV, Finlay AY, Tomas-Aragones L, Poot F, Sampogna F, Marron SE, et al. Quality of life in hidradenitis suppurativa: an update. Int J Environ Res Public Health. 2021;18:6131.
Bianchi L, Caposiena Caro RD, Ganzetti G, Molinelli E, Dini V, Oranges T, et al. Sex-related differences of clinical features in hidradenitis suppurativa: analysis of an Italian-based cohort. Clin Exp Dermatol. 2019;44:e177–80.
Napolitano M, Megna M, Timoshchuk E, Patruno C, Balato N, Fabbrocini G, et al. Hidradenitis suppurativa: from pathogenesis to diagnosis and treatment. Clin Cosmet Investig Dermatol. 2017;10:105–15.
Prens E, Deckers I. Pathophysiology of hidradenitis suppurativa: an update. J Am Acad Dermatol. 2015;73:S8-11.
Zouboulis CC, Desai N, Emtestam L, Hunger RE, Ioannides D, Juhász I, et al. European S1 guideline for the treatment of hidradenitis suppurativa/acne inversa. J Eur Acad Dermatol Venereol. 2015;29:619–44.
Kathju S, Lasko L-A, Stoodley P. Considering hidradenitis suppurativa as a bacterial biofilm disease. FEMS Immunol Med Microbiol. 2012;65:385–9.
Bechara FG, Sand M, Skrygan M, Kreuter A, Altmeyer P, Gambichler T. Acne inversa: evaluating antimicrobial peptides and proteins. Ann Dermatol. 2012;24:393.
Zouboulis CC, del Marmol V, Mrowietz U, Prens EP, Tzellos T, Jemec GBE. Hidradenitis suppurativa/acne inversa: criteria for diagnosis, severity assessment, classification and disease evaluation. Dermatology. 2015;231:184–90.
Napolitano M, Calzavara-Pinton PG, Zanca A, Bianchi L, Caposiena Caro RD, Offidani AM, et al. Comparison of clinical and ultrasound scores in patients with hidradenitis suppurativa: results from an Italian ultrasound working group. J Eur Acad Dermatol Venereol. 2019. https://doi.org/10.1111/jdv.15235.
Lacarrubba F, Dini V, Napolitano M, Venturini M, Caposiena Caro DR, Molinelli E, et al. Ultrasonography in the pathway to an optimal standard of care of hidradenitis suppurativa: the Italian Ultrasound Working Group experience. J Eur Acad Dermatol Venereol. 2019;33:10–4.
Alikhan A, Sayed C, Alavi A, Alhusayen R, Brassard A, Burkhart C, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations. J Am Acad Dermatol. 2019;81:91–101.
Molinelli E, Brisigotti V, Simonetti O, Sapigni C, D’Agostino GM, Rizzetto G, et al. Efficacy and safety of topical resorcinol 15% versus topical clindamycin 1% in the management of mild-to-moderate hidradenitis suppurativa: a retrospective study. Dermatol Ther. 2022. https://doi.org/10.1111/dth.15439.
Molinelli E, Brisigotti V, Simonetti O, Campanati A, Sapigni C, D’Agostino GM, et al. Efficacy and safety of topical resorcinol 15% as long-term treatment of mild-to-moderate hidradenitis suppurativa: a valid alternative to clindamycin in the panorama of antibiotic resistance. Br J Dermatol. 2020;183:1117–9.
Saunte DML, Jemec GBE. Hidradenitis suppurativa. JAMA. 2017;318:2019.
Molinelli E, De Simoni E, Candelora M, Sapigni C, Brisigotti V, Rizzetto G, et al. Systemic antibiotic therapy in hidradenitis suppurativa: a review on treatment landscape and current issues. Antibiotics. 2023;12:978.
Molinelli E, Sapigni C, D’Agostino GM, Brisigotti V, Rizzetto G, Bobyr I, et al. The effect of dalbavancin in moderate to severe hidradenitis suppurativa. Antibiotics. 2022;11:1573.
Molinelli E, Brisigotti V, Campanati A, Sapigni C, Giacchetti A, Cota C, et al. Efficacy of oral zinc and nicotinamide as maintenance therapy for mild/moderate hidradenitis suppurativa: a controlled retrospective clinical study. J Am Acad Dermatol. 2020;83:665–7.
Molinelli E, Paolinelli M, Campanati A, Brisigotti V, Offidani A. Metabolic, pharmacokinetic, and toxicological issues surrounding dapsone. Expert Opin Drug Metab Toxicol. 2019;15:367–79.
Molinelli E, Sapigni C, Simonetti O, Brisigotti V, Giuliodori K, Offidani A. Alexandrite laser as an adjuvant therapy in the management of mild to moderate hidradenitis suppurativa: a controlled prospective clinical study. J Am Acad Dermatol. 2022;87:674–5.
Molinelli E, Campanati A, Ganzetti G, Offidani A. Biologic therapy in immune mediated inflammatory disease: basic science and clinical concepts. Curr Drug Saf. 2016;11:35–43.
Simonetti O, Lucarini G, Campanati A, Goteri G, Zizzi A, Marconi B, et al. VEGF, survivin and NOS overexpression in psoriatic skin: critical role of nitric oxide synthases. J Dermatol Sci. 2009;54:205–8.
Simonetti O, Ferretti G, Salvi A, Offidani AM, Bossi G. Plasma lipid changes in psoriatic children. Dermatology. 1992;185:96–100.
Snyder CL, Gibson RS, Porter ML, Kimball AB. Secukinumab in the treatment of hidradenitis suppurativa. Immunotherapy. 2023;15:1449–57.
Goldburg SR, Strober BE, Payette MJ. Hidradenitis suppurativa. J Am Acad Dermatol. 2020;82:1061–82.
Molinelli E, Sapigni C, Campanati A, Brisigotti V, Offidani A. Metabolic, pharmacokinetic, and toxicological issues of biologic therapies currently used in the treatment of hidradenitis suppurativa. Expert Opin Drug Metab Toxicol. 2020;16:1019–37.
Wollina U, Brzezinski P, Koch A, Philipp-Dormston WG. Immunomodulatory drugs alone and adjuvant to surgery for hidradenitis suppurativa/acne inversa—a narrative review. Dermatol Ther. 2020. https://doi.org/10.1111/dth.13877.
Bastien J, Rochette-Egly C. Nuclear retinoid receptors and the transcription of retinoid-target genes. Gene. 2004;328:1–16.
Khalil S, Bardawil T, Stephan C, Darwiche N, Abbas O, Kibbi AG, et al. Retinoids: a journey from the molecular structures and mechanisms of action to clinical uses in dermatology and adverse effects. J Dermatol Treat. 2017;28:684–96.
Mukherjee S, Date A, Patravale V, Korting HC, Roeder A, Weindl G. Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety. Clin Interv Aging. 2006;1:327–48.
Hendricks AJ, Hsiao JL, Lowes MA, Shi VY. A comparison of international management guidelines for hidradenitis suppurativa. Dermatology. 2021;237:81–96.
Pile HD, Sadiq NM. Isotretinoin. In: StatPearls. Treasure Island: StatPearls Publishing; 2023.
Wiegand U-W, Chou RC. Pharmacokinetics of oral isotretinoin. J Am Acad Dermatol. 1998;39:S8-12.
Melnik B. Apoptosis may explain the pharmacological mode of action and adverse effects of isotretinoin, including teratogenicity. Acta Dermatol Venereol. 2017;97:173–81.
Scala E, Cacciapuoti S, Garzorz-Stark N, Megna M, Marasca C, Seiringer P, et al. Hidradenitis suppurativa: where we are and where we are going. Cells. 2021;10:2094.
Frew JW. Hidradenitis suppurativa is an autoinflammatory keratinization disease: a review of the clinical, histologic, and molecular evidence. JAAD Int. 2020;1:62–72.
Bergler-Czop B, Brzezińska-Wcisło L. Pro-inflammatory cytokines in patients with various kinds of acne treated with isotretinoin. Adv Dermatol Allergol. 2014;1:21–8.
Dispenza MC, Wolpert EB, Gilliland KL, Dai JP, Cong Z, Nelson AM, et al. Systemic isotretinoin therapy normalizes exaggerated TLR-2-mediated innate immune responses in acne patients. J Investig Dermatol. 2012;132:2198–205.
Hessam S, Gambichler T, Höxtermann S, Skrygan M, Sand M, Garcovich S, et al. Frequency of circulating subpopulations of T-regulatory cells in patients with hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2020;34:834–8.
Nolan ZT, Banerjee K, Cong Z, Gettle SL, Longenecker AL, Kawasawa YI, et al. Treatment response to isotretinoin correlates with specific shifts in Cutibacterium acnes strain composition within the follicular microbiome. Exp Dermatol. 2023;32:955–64.
Ring HC, Thorsen J, Saunte DM, Lilje B, Bay L, Riis PT, et al. The follicular skin microbiome in patients with hidradenitis suppurativa and healthy controls. JAMA Dermatol. 2017;153:897.
Kamp S, Fiehn AM, Stenderup K, Rosada C, Pakkenberg B, Kemp K, et al. Hidradenitis suppurativa: a disease of the absent sebaceous gland? Sebaceous gland number and volume are significantly reduced in uninvolved hair follicles from patients with hidradenitis suppurativa. Br J Dermatol. 2011;164:1017–22.
Poli F, Revuz J. «Poussées inflammatoires d’acné » sous isotrétinoïne révélant une hidradénite suppurée: 4 cas. Ann Dermatol Venereol. 2019;146:4–8.
Gallagher CG, Kirthi SK, Cotter CC, Revuz JR, Tobin AMT. Could isotretinoin flare hidradenitis suppurativa? A case series. Clin Exp Dermatol. 2019;44:777–80.
Caposiena Caro RD, Bianchi L. Can retinoids flare hidradenitis suppurativa? A further case series. Clin Exp Dermatol. 2019;44:e153–4.
Jørgensen A-HR, Thomsen SF, Ring HC. Isotretinoin and hidradenitis suppurativa. Clin Exp Dermatol. 2019;44:e155–6.
Soria A, Canoui-Poitrine F, Wolkenstein P, Poli F, Gabison G, Pouget F, et al. Absence of efficacy of oral isotretinoin in hidradenitis suppurativa: a retrospective study based on patients’ outcome assessment. Dermatology. 2009;218:134–5.
De Vita V, Melnik BC. The magnitude of mTORC1 signalling may predict the response to isotretinoin treatment in patients with hidradenitis suppurativa. Dermatology. 2017;233:399–400.
Marasca C, Balato A, Annunziata MC, Cacciapuoti S, Fabbrocini G. Insulin resistance, mTOR and hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2019. https://doi.org/10.1111/jdv.15271.
Balato A, Caiazzo G, Annunziata MC, Marasca C, Scala E, Cacciapuoti S, et al. Anti-TNF-α therapy modulates mTORC1 signalling in hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2019. https://doi.org/10.1111/jdv.15160.
Bouwman K, Aarts P, Dudink K, Hao J, Alizadeh BZ, Prens LM, et al. Drug survival of oral retinoids in hidradenitis suppurativa: a real-life cohort study. Am J Clin Dermatol. 2022;23:905–14.
Libow LFFDA. Arthropathy associated with cystic acne, hidradenitis suppurativa, and perifolliculitis capitis abscedens et suffodiens: treatment with isotretinoin. Cutis. 1999;64(2):87–90.
Huang CM, Kirchhof MG. A new perspective on isotretinoin treatment of hidradenitis suppurativa: a retrospective chart review of patient outcomes. Dermatology. 2017;233:120–5.
Boer J, van Gemert MJP. Long-term results of isotretinoin in the treatment of 68 patients with hidradenitis suppurativa. J Am Acad Dermatol. 1999;40:73–6.
Patel N, McKenzie SA, Harview CL, Truong AK, Shi VY, Chen L, et al. Isotretinoin in the treatment of hidradenitis suppurativa: a retrospective study. J Dermatol Treat. 2021;32:473–5.
McPhie ML, Bridgman AC, Kirchhof MG. Combination therapies for hidradenitis suppurativa: a retrospective chart review of 31 patients. J Cutan Med Surg. 2019;23:270–6.
Coskun BN, Yagiz B, Pehlivan Y, Dalkilic E. Isotretinoin-induced sacroiliitis in patients with hidradenitis suppurativa: a case-based review. Rheumatol Int. 2019;39:2159–65.
Schaffer LC, Schaffer CB, Hunter S, Miller A. Psychiatric reactions to isotretinoin in patients with bipolar disorder. J Affect Disord. 2010;122:306–8.
Tzur Bitan D, Berzin D, Cohen A. Hidradenitis suppurativa and bipolar disorders: a population-based study. Dermatology. 2020;236:298–304.
Zito PM, Mazzoni T. Acitretin. In: StatPearls. Treasure Island: StatPearls Publishing; 2023.
Ganzetti G, Campanati A, Santarelli A, Pozzi V, Molinelli E, Minnetti I, et al. Periodontal disease: an oral manifestation of psoriasis or an occasional finding? Drug Dev Res. 2014. https://doi.org/10.1002/ddr.21194.
Pilkington T, Brogden RN. Acitretin. Drugs. 1992;43:597–627.
Ortiz NEG, Nijhawan RI, Weinberg JM. Acitretin. Dermatol Ther. 2013;26:390–9.
Guenther LC, Kunynetz R, Lynde CW, Sibbald RG, Toole J, Vender R, et al. Acitretin use in dermatology. J Cutan Med Surg. 2017;21:2S-12S.
Tu J, Yin Z, Guo J, He F, Long F, Yin Z. Acitretin inhibits IL-17A-induced IL-36 expression in keratinocytes by down-regulating IκBζ. Int Immunopharmacol. 2020;79: 106045.
Boer J, Nazary M. Long-term results of acitretin therapy for hidradenitis suppurativa. Is acne inversa also a misnomer? Br J Dermatol. 2011;164:170–5.
Matusiak Ł, Bieniek A, Szepietowski JC. Acitretin treatment for hidradenitis suppurativa: a prospective series of 17 patients. Br J Dermatol. 2014;171:170–4.
Sánchez-Díaz M, Díaz-Calvillo P, Rodríguez-Pozo JÁ, Arias-Santiago S, Molina-Leyva A. Effectiveness and safety of acitretin for the treatment of hidradenitis suppurativa, predictors of clinical response: a cohort study. Dermatology. 2023;239:52–9.
Tan MG, Shear NH, Walsh S, Alhusayen R. Acitretin. J Cutan Med Surg. 2017;21:48–53.
Molinelli E, Sapigni C, Simonetti O, D’Agostino GM, Brisigotti V, Rizzetto G, et al. Acitretin plus macrolides and acitretin monotherapy in the management of hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2023. https://doi.org/10.1111/jdv.18706.
Puri N, Talwar A. A study on the management of hidradenitis suppurativa with retinoids and surgical excision. Indian J Dermatol. 2011;56:650.
Chen A-W, Chen Z, Bai X-M, Luo X-Y, Wang H. Successful treatment of early-onset hidradenitis suppurativa with acitretin in an infant with a novel mutation in PSENEN gene. Indian J Dermatol Venereol Leprol. 2022;88:445.
D’Ambra I, Caccavale S, Ronchi A, Franco R, Argenziano G. A case of hidradenitis suppurativa and Darier disease in a patient treated with acitretin: how to kill two birds with one stone. Italian J Dermatol Venereol. 2022. https://doi.org/10.23736/S2784-8671.20.06568-2.
Sachdev SS, Jamil A, Gunabalasingam P, Safdar NA. The effects of acitretin on insulin resistance, glucose metabolism, and lipid levels in patients with psoriasis. Indian J Dermatol. 2022;67(4):349–54.
Diepgen T, Pfarr E, Zimmermann T. Efficacy and tolerability of alitretinoin for chronic hand eczema under daily practice conditions: results of the TOCCATA open study comprising 680 patients. Acta Derm Venereol. 2012;92:251–5.
Cheng C, Michaels J, Scheinfeld N. Alitretinoin: a comprehensive review. Expert Opin Investig Drugs. 2008;17:437–43.
Verdolini R, Simonacci F, Menon S, Pavlou P, Mannello B. Alitretinoin: a useful agent in the treatment of hidradenitis suppurativa, especially in women of child-bearing age. G Ital Dermatol Venereol. 2015;150(2):155–62 (Epub 2014 Jun 13).
Bubna A. Alitretinoin in dermatology—an update. Indian J Dermatol. 2015;60:520.
Prasad S, Bygum A. Successful treatment with alitretinoin of dissecting cellulitis of the scalp in keratitis-ichthyosis-deafness syndrome. Acta Derm Venereol. 2013;93:473–4.
Motamedi M, Chehade A, Sanghera R, Grewal P. A clinician’s guide to topical retinoids. J Cutan Med Surg. 2022;26:71–8.
Thielitz A, Abdel-Naser MB, Fluhr JW, Zouboulis CC, Gollnick H. Topical retinoids in acne—an evidence-based overview. JDDG Journal der Deutschen Dermatologischen Gesellschaft. 2008;6:1023–31.
Kaplan YC, Ozsarfati J, Etwel F, Nickel C, Nulman I, Koren G. Pregnancy outcomes following first-trimester exposure to topical retinoids: a systematic review and meta-analysis. Br J Dermatol. 2015;173:1132–41.
Leachman SA, Reed BR. The use of dermatologic drugs in pregnancy and lactation. Dermatol Clin. 2006;24:167–97.
Scheinfeld N. Hidradenitis suppurativa in prepubescent and pubescent children. Clin Dermatol. 2015;33:316–9.
Karpouzis A, Giatromanolaki A, Sivridis E, Kouskoukis C. Perifolliculitis capitis abscedens et suffodiens successfully controlled with topical isotretinoin. Eur J Dermatol. 2003;13(2):192–5.
Das K, Daveluy S, Kroumpouzos G, Agarwal K, Podder I, Farnbach K, et al. Efficacy and toxicity of classical immunosuppressants, retinoids and biologics in hidradenitis suppurativa. J Clin Med. 2022;11:670.
Marasca C, Fabbrocini G, Abategiovanni L, Camela E, Nocerino M, Di Guida A, et al. Adalimumab in the management of isotretinoin-induced acne fulminans: report of a case. Skin Appendage Disord. 2021;7:115–9.
Clinicaltrials.gov and Pubmed.ncbi.nlm.nih.gov.
Medical Writing and Editorial Assistance
The authors did not use any medical writing or editorial assistance for this article.
Funding
No funding or sponsorship was received for this study or publication of this article.
Author information
Authors and Affiliations
Contributions
Elisa Molinelli, Helena Gioacchini, Andrea Marani: conceptualization, supervision, data curation, writing-review and editing. Daisy Gambini, Giulio Rizzetto, Edoardo De Simoni: data curation, writing-review, editing and methodology. Annamaria Offidani, Oriana Simonetti: methodology, supervision, formal analysis and editing.
Corresponding author
Ethics declarations
Conflict of Interest
Elisa Molinelli, Helena Gioacchini, Andrea Marani, Daisy Gambini, Giulio Rizzetto, Edoardo De Simoni, Annamaria Offidani, Oriana Simonetti have nothing to disclose.
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Molinelli, E., Gioacchini, H., Marani, A. et al. Topical and Systemic Retinoids in the Management of Hidradenitis Suppurativa: A Comprehensive Literature Review. Dermatol Ther (Heidelb) 14, 1079–1091 (2024). https://doi.org/10.1007/s13555-024-01169-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-024-01169-1