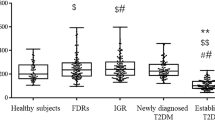
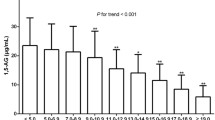
Abstract
Objective
Serum 1, 5-AG is a glycaemic marker reflecting control and fluctuations of short-term glucose. To reveal the relationship between 1, 5-AG with β-cell function, we investigated a certain number of Chinese adults with different glucose metabolisms.
Methods
In clinical cross-sectional study, 2184 subjects with no prior diabetes history are included and underwent an OGTT. HOMA-IR and HOMA-β were calculated. Correlations between 1, 5-AG and HOMA-IR or HOMA-β were observed, correcting for interference factors, and independent factors for HOMA-β and HOMA-IR were analysed. Subjects were divided into three groups according to OGTT results, and 1, 5-AG levels differed significantly between them.
Results
A significant positive correlation existed between 1, 5-AG and HOMA-β only in the diabetes mellitus group (r = 0.265, p < 0.001). Above phenomenon remained after adjusting for indicators, however, disappeared after considering serum uric acid. Both 1, 5-AG and HbA1c were independent factors for HOMA-β (1, 5-AG: β = 0.772, p = 0.023; HbA1c: β = -7.52, p = 0.003). Conclusion: 1, 5-AG remained a factor for HOMA-β only for those with NUA. 1, 5-AG reflects different glucose metabolism statuses and is an auxiliary observation reflecting the secretory function of β cells in NUA patients.

Similar content being viewed by others
Data availability
All the data supporting the results are shown in the paper and can be applicable from the corresponding author.
Abbreviations
- 1, 5-AG:
-
Serum 1, 5-anhydroglucitol
- OGTT:
-
Oral glucose tolerance test
- HOMA-IR:
-
Homeostasis model assessments for insulin resistance
- HOMA-β:
-
Homeostasis model assessments for β cells
- NUA:
-
Normal serum uric
- T2DM:
-
Type 2 diabetes mellitus
- OGIRT:
-
Oral glucose insulin release test
- HbA1c:
-
Glycosylated hemoglobin
- GA:
-
Glycated albumin
- FPG:
-
Fasting blood glucose
- 2hPG:
-
2-Hour postprandial plasma glucose
- TC:
-
Total cholesterol
- TG:
-
Triglycerides
- HDL-c:
-
High-density lipoprotein cholesterol
- LDL-c:
-
Low-density lipoprotein cholesterol
- BUN:
-
Blood urea nitrogen
- SCr:
-
Serum creatinine
- SUA:
-
Serum uric acid
- eGFR:
-
Estimated glomerular filtration rate
- NGT:
-
Normal glucose tolerance
- IGR:
-
Impaired glucose regulation
- HUA:
-
High uric acid
References
Sun H, Saeedi P, Karuranga S, et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2022;183:109119.
Abdul-Ghani MA, Tripathy D, DeFronzo RA. Contributions of beta-cell dysfunction and insulin resistance to the pathogenesis of impaired glucose tolerance and impaired fasting glucose. Diabetes Care. 2006;29:1130–9.
Hong J, Gu WQ, Zhang YF, et al. The interplay of insulin resistance and beta-cell dysfunction involves the development of type 2 diabetes in Chinese obeses. Endocrine. 2007;31:93–9.
Pitkanen E. Serum 1, 5-Anhydroglucitol in Normal Subjects and in Patients with Insulin-Dependent Diabetes-Mellitus. Scand J Clin Lab Inv. 1982;42:445–8.
Ying L, Ma X, Yin J, et al. The metabolism and transport of 1, 5-anhydroglucitol in cells. Acta Diabetol. 2018;55:279–86.
Ma C, Sheng J, Liu Z, Guo M. Excretion rates of 1, 5-anhydro-D-glucitol, uric acid and microalbuminuria as glycemic control indexes in patients with type 2 diabetes. Sci Rep. 2017;7:44291.
Kim WJ, Park CY. 1, 5-Anhydroglucitol in diabetes mellitus. Endocrine. 2013;43:33–40.
Seok H, Huh JH, Kim HM, et al. 1, 5-anhydroglucitol as a useful marker for assessing short-term glycemic excursions in type 1 diabetes. Diabetes Metab J. 2015;39:164–70.
Kulozik F, Hasslacher C. 1, 5-Anhydroglucitol as a Marker for Short-Term Glycaemic Excursions. Diabetes Stoffwech H. 2015;24:89–94.
Mook-Kanamori DO, El-Din SMM, Takiddin AH, et al. 1, 5-Anhydroglucitol in Saliva Is a Noninvasive Marker of Short-Term Glycemic Control. J Clin Endocr Metab. 2017;102:3867–3867.
Kim MJ, Jung HS, Hwang-Bo Y, et al. Evaluation of 1, 5-anhydroglucitol as a marker for glycemic variability in patients with type 2 diabetes mellitus. Acta Diabetol. 2013;50:505–10.
Januszewski AS, Karschimkus C, Davis KE, et al. Plasma 1, 5 anhydroglucitol levels, a measure of short-term glycaemia: assay assessment and lower levels in diabetic vs. non-diabetic subjects. Diabetes Res Clin Pract. 2012;95:17–9.
Won JC, Park CY, Park HS, et al. 1, 5-Anhydroglucitol reflects postprandial hyperglycemia and a decreased insulinogenic index, even in subjects with prediabetes and well-controlled type 2 diabetes. Diabetes Res Clin Pract. 2009;84:51–7.
Ma X, Hao Y, Hu X, et al. 1, 5-anhydroglucitol is associated with early-phase insulin secretion in chinese patients with newly diagnosed type 2 diabetes mellitus. Diabetes Technol Ther. 2015;17:320–6.
Ma YC, Zuo L, Chen JH, et al. Modified glomerular filtration rate estimating equation for Chinese patients with chronic kidney disease. J Am Soc Nephrol. 2006;17:2937–44.
Matthews DR, Hosker JP, Rudenski AS, et al. Homeostasis model assessment, pp. insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–9.
Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabetic Med. 1998;15:539–53.
Yamanouchi T, Inoue T, Ichiyanagi K, et al. 1, 5-Anhydroglucitol stimulates insulin release in insulinoma cell lines. Biochim Biophys Acta. 2003;1623:82–7.
Nakamura S, Tanabe K, Yoshinaga K, et al. Effects of 1, 5-anhydroglucitol on postprandial blood glucose and insulin levels and hydrogen excretion in rats and healthy humans. Br J Nutr. 2017;118:81–91.
Chizynski K, Rozycka M. Hyperuricemia. Pol Merkur Lekarski. 2005;19:693–6.
DeFronzo RA, Abdul-Ghani MA. Preservation of beta-Cell Function: The Key to Diabetes Prevention. J Clin Endocr Metab. 2011;96:2354–66.
Kahn SE. The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of Type 2 diabetes. Diabetologia. 2003;46:3–19.
Perley MJ, Kipnis DM. Plasma Insulin Responses to Oral and Intravenous Glucose - Studies in Normal and Diabetic Subjects. J Clin Invest. 1967;46:1954–8.
Brunzell JD, Robertson RP, Lerner RL, et al. Relationships between Fasting Plasma Glucose Levels and Insulin-Secretion during Intravenous Glucose-Tolerance Tests. J Clin Endocr Metab. 1976;42:222–9.
Ehrmann DA, Sturis J, Byrne MM, et al. Insulin Secretory Defects in Polycystic-Ovary-Syndrome - Relationship to Insulin Sensitivity and Family History of Non-Insulin-Dependent Diabetes-Mellitus. J Clin Invest. 1995;96:520–7.
Ward WK, Johnston C, Beard JC, et al. Insulin Resistance and Impaired Insulin-Secretion in Subjects with Histories of Gestational Diabetes-Mellitus. Diabetes. 1985;34:861–9.
Ryan EA, Imes S, Liu DT, et al. Defects in Insulin-Secretion and Action in Women with a History of Gestational Diabetes. Diabetes. 1995;44:506–12.
Matsuda M, DeFronzo RA. Insulin sensitivity indices obtained from oral glucose tolerance testing - Comparison with the euglycemic insulin clamp. Diabetes Care. 1999;22:1462–70.
Koga M, Murai J, Saito H, Kasayama S. Glycated Albumin and Glycated Hemoglobin Are Influenced Differently by Endogenous Insulin Secretion in Patients With Type 2 Diabetes. Diabetes Care. 2010;33:270–7.
Kim D, Kim KJ, Huh JH, et al. The ratio of glycated albumin to glycated haemoglobin correlates with insulin secretory function. Clin Endocrinol (Oxf). 2012;77:679–83.
Pitkanen E. Occurrence of 1, 5-Anhydroglucitol in Human Cerebrospinal-Fluid. Clin Chim Acta. 1973;48:159–66.
Yamanouchi T, Tachibana Y, Akanuma H, et al. Origin and disposal of 1, 5-anhydroglucitol, a major polyol in the human body. Am J Physiol. 1992;263:268–73.
Wang Y, Yuan Y, Zhang Y, et al. Serum 1, 5-anhydroglucitol level as a screening tool for diabetes mellitus in a community-based population at high risk of diabetes. Acta Diabetol. 2017;54:425–31.
Al-Masri AA, Eter EE, Tayel S, et al. Differential associations of circulating peroxiredoxins levels with indicators of glycemic control in type 2 diabetes mellitus. Eur Rev Med Pharmacol Sci. 2014;18(5):710–6.
Christensen BL, Williams M. Assessing postprandial glucose using 1, 5-anhydroglucitol, pp. An integrative literature review. J Am Acad Nurse Pract. 2009;21:542–8.
Schindhelm RK, Diamant M, Bilo HJ, Slingerland RJ. Association of 1, 5-anhydroglucitol and 2-h postprandial blood glucose in type 2 diabetic patients. Diabetes Care. 2009;32:207–207.
González C, Alonso A, Grueso NA, et al. Effect of treatment with different doses of 17-beta-estradiol on the insulin receptor. Life Sci. 2019;70(14):1621–30.
Puig JG, Mateos FA, Miranda ME, et al. Purine Metabolism in Women with Primary Gout. Am J Med. 1994;97:332–8.
Hak AE, Choi HK. Menopause, postmenopausal hormone use and serum uric acid levels in US women - The Third National Health and Nutrition Examination Survey. Arthritis Res Ther. 2008;10:1–11.
Sumino H, Ichikawa S, Kanda T, et al. Reduction of serum uric acid by hormone replacement therapy in postmenopausal women with hyperuricaemia. Lancet. 1999;354:650–650.
Koga M, Murai J, Saito H, et al. Close relationship between serum concentrations of 1, 5-anhydroglucitol and uric acid in non-diabetic male subjects implies common renal transport system. Clin Chim Acta. 2009;410:70–3.
Ouchi M, Oba K, Aoyama J, et al. Serum uric acid in relation to serum 1, 5-anhydroglucitol levels in patients with and without type 2 diabetes mellitus. Clin Biochem. 2013;46:1436–41.
Hu YM, Liu J, Li HQ, et al. The association between elevated serum uric acid levels and islet β-cell function indexes in newly diagnosed type 2 diabetes mellitus: a cross-sectional study. PeerJ. 2018;6(7):4515–8.
Zhong X, Zhang D, Yang L. The relationship between serum uric acid within the normal range and beta-cell function in Chinese patients with type 2 diabetes: differences by body mass index and gender. PeerJ. 2019;7:6666.
Tang W, Fu Q, Zhang Q, et al. The association between serum uric acid and residual beta -cell function in type 2 diabetes. J Diabetes Res. 2014;2014: 709691.
Ling Y, Li XM, Gu Q. Correlation of serum uric acid and islet beta cell functions in female type 2 diabetics. Nat Med J China. 2012;92:541–5.
Acknowledgements
Not applicable.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was approved in 2017 by the Clinical Ethics Committee of Zhongda Hospital, an affiliate of Southeast University (No.2017ZDSYLL006-P01), and all subjects signed informed consent forms before registration. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. This study was approved by the Clinical Ethics Committee of our hospital and all subjects signed informed consent forms before registration.
Conflicts of interest
The authors declare that there are no conflicts of interest regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yuan, Y., Tan, Y., Wang, Y. et al. The correlation between serum 1, 5-anhydroglucitol and β-cell function in Chinese adults with different glucose metabolism statuses. Int J Diabetes Dev Ctries (2023). https://doi.org/10.1007/s13410-023-01295-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13410-023-01295-0