Abstract
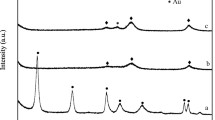
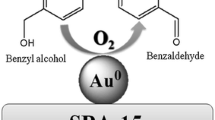
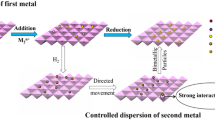
Gold nanoparticles are attractive in catalytic field due to their high activity and selectivity. However, dispersion and sintering resistance have been two of the biggest issues in catalysis for preparation of gold nanoparticles. In this paper, a controlled impregnation method was employed to prepare highly dispersed and thermally stable Au-based catalysts for aerobic oxidation of benzyl alcohol. The size of hydrotalcites (HTs)-supported Au nanoparticles was 2.8 nm at 300 °C of calcination temperature, which was 3.2 nm when calcination temperature was increased to 600 °C. The mechanism of forming metal nanoparticles was proposed. The high dispersion of Au was ascribed to the directing role of Cu and the interaction between Au (and Pt) and Cu. It was proposed that the thermal stability of Au nanoparticles was due to the simultaneous employment of multiple additional effects. The activity of Au nanoparticles was far higher than those prepared by traditional methods. Furthermore, the PtCu elements in Au nanoparticles were homogeneously distributed. In contrast, they were randomly distributed in Au nanoparticles prepared by the traditional method.











Similar content being viewed by others
References
Wei Z, Sun J, Li Y, Datye AK, Wang Y (2012) Bimetallic catalysts for hydrogen generation. Chem Soc Rev 41:7994–8008
Wang S, Zhao Q, Wei H, Wang J, Cho M, Cho HS, Terasaki O, Wan Y (2013) Aggregation-free gold nanoparticles in ordered mesoporous carbons: toward highly active and stable heterogeneous catalysts. J Am Chem Soc 135:11849–11860
Yang B, Burch R, Hardacre C, Headdock G, Hu P (2012) Origin of the increase of activity and selectivity of nickel doped by Au, Ag, and Cu for acetylene hydrogenation. ACS Catal 2:1027–1032
Gu X, Lu Z, Jiang H, Akita T, Xu Q (2011) Synergistic catalysis of metal-organic framework-immobilized Au-Pd nanoparticles in dehydrogenation of formic acid for chemical hydrogen storage. J Am Chem Soc 133:11822–11825
Paalanen P, Weckhuysen BM, Sankar M (2013) Progress in controlling the size, composition and nanostructure of supported gold-palladium nanoparticles for catalytic applications. Catal Sci Technol 3:2869–2880
Li L, Gao Y, Li H, Zhao Y, Pei Y, Chen Z, Zeng X (2013) CO oxidation on TiO2 (110) supported subnanometer gold clusters: size and shape effects. J Am Chem Soc 135:19336–19346
Falsig H, Hvolbæk B, Kristensen IS, Jiang T, Bligaard T, Christensen CH, Nørskov JK (2008) Trends in the catalytic CO oxidation activity of nanoparticles. Angew Chem 120:4913–4917
Cao A, Lu R, Veser G (2010) Stabilizing metal nanoparticles for heterogeneous catalysis. Phys Chem Chem Phys 12:13499–13510
Hansen TW, Delariva AT, Challa SR, Datye AK (2013) Sintering of catalytic nanoparticles: particle migration or Ostwald ripening ? Acc Chem Res 46:1720–1730
Liu H, Zhang L, Wang N, Su D (2014) Palladium nanoparticles embedded in the inner surfaces of carbon nanotubes: synthesis, catalytic activity, and sinter resistance. Angew Chem Int Ed 53:12634–12638
Zhu H, Sigdel A, Zhang S, Su D, Xi Z, Li Q, Sun S (2014) Core/shell Au/MnO nanoparticles prepared through controlled oxidation of AuMn as an electrocatalyst for sensitive H2O2 detection. Angew Chem Int Ed 53:12508–12512
Bracey CL, Ellis PR, Hutchings GJ (2009) Application of copper-gold alloys in catalysis: current status and future perspectives. Chem Soc Rev 38:2231–2243
Li X, Fang SSS, Teo J, Foo YL, Borgna A, Lin M, Zhong Z (2012) Activation and deactivation of Au-Cu/SBA-15 catalyst for preferential oxidation of CO in H2-rich gas. ACS Catal 2:360–369
Bauer JC, Mullins D, Li M, Wu Z, Payzant EA, Overbury SH, Dai S (2011) Synthesis of silica supported AuCu nanoparticle catalysts and the effects of pretreatment conditions for the CO oxidation reaction. Phys Chem Chem Phys 13:2571–2581
Zhong R, Sun K, Hong Y, Xu B (2014) Impacts of organic stabilizers on catalysis of Au nanoparticles from colloidal preparation. ACS Catal 4:3982–3993
Wang H, Wang C, Yan H, Yi H, Lu J (2015) Precisely-controlled synthesis of Au@Pd core-shell bimetallic catalyst via atomic layer deposition for selective oxidation of benzyl alcohol. J Catal 324:59–68
Barbier J, Ertl in G, Knözinger H, Weitkamp J (1999) Preparation of solid catalysts. Wiley-VCH Verlag GmbH, Weinheim
Rebelli J, Rodriguez AA, Ma S, Williams CT, Monnier JR (2011) Preparation and characterization of silica-supported, group IB-Pd bimetallic catalysts prepared by electroless deposition methods. Catal Today 160:170–178
Barbier J, Marécot P, Del Angel G, Bosch P, Boitiaux JP, Didillon B, Dominguez JM, Schifter I, Espmosa G (1994) Preparation of platinum-gold bimetallic catalysts by redox reactions. Appl Catal A 116:179–186
Rodriguez AA, Williams CT, Monnier JR (2014) Selective liquid-phase oxidation of glycerol over Au-Pd/C bimetallic catalysts prepared by electroless deposition. Appl Catal A 475:161–168
Wang H, Liu D, Xu C (2016) Directed synthesis of well dispersed and highly active AuCu and AuNi nanoparticle catalysts. Catal Sci Technol 6:7137–7150
Wang Y, He J, Liu C, Chong W, Chen H (2015) Thermodynamics versus kinetics in nanosynthesis. Angew Chem Int Ed 54:2022–2051
Chen C, Nan C, Wang D, Su Q, Duan H, Liu X, Zhang L, Chu D, Song W, Peng Q, Li Y (2011) Mesoporous multicomponent nanocomposite colloidal spheres: ideal high-temperature stable model catalysts. Angew Chem Int Ed 50:3725–3729
Chen J, Zhang R, Han L, Tu B, Zhao D (2013) One-pot synthesis of thermally stable gold@mesoporous silica core-shell nanospheres with catalytic activity. Nano Res 6:871–879
Cao A, Veser G (2010) Exceptional high-temperature stability through distillation-like self-stabilization in bimetallic nanoparticles. Nat Mater 9:75–81
Tang H, Wei J, Liu F, Qiao B, Pan X, Li L, Liu J, Wang J, Zhang T (2015) Strong metal-support interactions between gold nanoparticles and nonoxides. J Am Chem Soc 138:56–59
Li W, Kovarik L, Mei D, Liu J, Wang Y, Peden CHF (2013) Stable platinum nanoparticles on specific MgAl2O4 spinel facets at high temperatures in oxidizing atmospheres. Nat Commun 4:2481
Im J, Choi M (2016) Physicochemical stabilization of Pt against sintering for a dehydrogenation catalyst with high activity, selectivity, and durability. ACS Catal 6:2819–2826
Tang H, Liu F, Wei J, Qiao B, Zhao K, Su Y, Jin C, Li L, Liu J, Wang J, Zhang T (2016) Ultrastable hydroxyapatite/titanium- dioxide-supported gold nanocatalyst with strong metal-support interaction for carbon monoxide oxidation. Angew Chem Int Ed 55:1–7
Wang H, Liu N, Xu C, Dong W, Liu C (2017) Directed dispersion of Au based catalysts at H2 reduction process for aerobic oxidation of benzyl alcohol. Catal Lett 147:547–565
Zope BN, Hibbitts DD, Neurock M, Davis RJ (2010) Reactivity of the gold/water interface during selective oxidation catalysis. Science 330:74–78
Pina CD, Falletta E, Rossi M (2012) Update on selective oxidation using gold. Chem Soc Rev 41:350–369
Xu C, Du Y, Li C, Yang J, Yang G (2015) Insight into effect of acid/base nature of supports on selectivity of glycerol oxidation over supported Au-Pt bimetallic catalysts. Appl Catal B 164:334–343
Xu Z, Zhang J, Adebajo MO, Zhang H, Zhou C (2011) Catalytic applications of layered double hydroxides and derivatives. Appl Clay Sci 53:139–150
Xu C, Sun J, Zhao B, Liu Q (2010) On the study of KF/Zn (Al) O catalyst for biodiesel production from vegetable oil. Appl Catal B 99:111–117
Xu J, Yue H, Liu S, Wang H, Du Y, Xu C, Dong W, Liu C (2016) Cu-Ag/hydrotalcite catalysts for dehydrogenative cross-coupling of primary and secondary benzylic alcohols. RSC Adv 6:24164–24174
Zhang J, Jin Y, Li C, Shen Y, Han L, Hua Z, Di X, Liu Z (2009) Creation of three-dimensionally ordered macroporous Au/CeO2 catalysts with controlled pore sizes and their enhanced catalytic performance for formaldehyde oxidation. Appl Catal B 91:11–20
Dixit M, Mishra M, Joshi PA, Shah DO (2013) Physico-chemical and catalytic properties of Mg-Al hydrotalcite and Mg-Al mixed oxide supported copper catalysts. J Ind Eng Chem 19:458–468
Dragoi B, Ungureanu A, Chirieac A, Ciotonea C, Rudolf C, Royer S, Dumitriua E (2015) Structural and catalytic properties of mono- and bimetallic nickel-copper nanoparticles derived from MgNi (Cu)Al-LDHs under reductive conditions. Appl Catal A 504:92–102
Meshesha BT, Barrabés N, Föttinger K, Chimentão RJ, Llorca J, Medina F, Rupprechter G, Sueiras JE (2012) Gas-phase hydrodechlorination of trichloroethylene over Pd/NiMgAl mixed oxide catalysts. Appl Catal B 117:236–245
Obalová L, Karásková K, Jirátová K, Kovanda F (2009) Effect of potassium in calcined Co-Mn-Al layered double hydroxide on the catalytic decomposition of N2O. Appl Catal B 90: 132-140
Zhang Y, Gu Y, Lin S, Wei J, Wang Z, Wang C, Du Y, Ye W (2011) One-step synthesis of PtPdAu ternary alloy nanoparticles on graphene with superior methanol electrooxidation activity. Electrochim Acta 56:8746–8751
Xiang X, He W, Xie L, Li F (2013) A mild solution chemistry method to synthesize hydrotalcite-supported platinum nanocrystals for selective hydrogenation of cinnamaldehyde in neat water. Catal Sci Technol 3:2819–2827
Applestone D, Yoon S, Manthiram A (2012) Cu2Sb-Al2O3-C nanocomposite alloy anodes with exceptional cycle life for lithium ion batteries. J Mater Chem 22:3242–3248
Du X, Luo S, Du H, Tang M, Huang X, Shen PK (2016) Monodisperse and self-assembled Pt-Cu nanoparticles as an efficient electrocatalyst for the methanol oxidation reaction. J Mater Chem A 4:1579–1585
Zhang Z, Hui J, Guo Z, Yu Q, Xu B, Zhang X, Liu Z, Xu C, Gao J, Wang X (2012) Solvothermal synthesis of Pt-Pd alloys with selective shapes and their enhanced electrocatalytic activities. Nano 4:2633–2639
Liu Y, He Y, Zhou D, Feng J, Li D (2016) Catalytic performance of Pd-promoted Cu hydrotalcite-derived catalysts in partial hydrogenation of acetylene: effect of Pd-Cu alloy formation. Catal Sci Technol 6:3027–3037
Acknowledgments
This work was supported by projects funded by the Major Research Plan of the National Natural Science Foundation of China (Program No. 91545130).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 4271 kb)
Rights and permissions
About this article
Cite this article
Liu, N., Chen, G., Dong, W. et al. Preparation of Au nanoparticles with high dispersion and thermal stability by a controlled impregnation method for alcohol oxidation. Gold Bull 50, 163–175 (2017). https://doi.org/10.1007/s13404-017-0206-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13404-017-0206-z