Abstract
Background
Glioblastoma multiforma (GBM) is the most malignant intrinsic tumor of the central nervous system (CNS), with high morbidity of 3.19/100,000 per year and a poor 5-year survival rate (< 5%) worldwide. Numerous studies have indicated that GBM shows remarkable radioresistance and aggressive recurrence. However, the mechanisms to endow GBM cells with radioresistance are complex and unclear.
Methods
Cell growth curve and colony formation assays were used to analyze the radioresistance of GBM. Immunoprecipitation and immunoblotting experiments were carried out to analyze protein expression and interaction.
Results
In the present study, we found that LITAF, lipopolysaccharide (LPS)-induced tumor necrosis factor (TNF)-α factor, is up-regulated both in mRNA and protein in GBM tumors. Meanwhile, we observed that high LITAF expression contributes to radioresistance of GBM cell lines (including U87, U251, DK, and AM38 cells), indicated by knockout or knockdown of LITAF in cells sensitizing them to radiation treatment both in vitro and in vivo. Furthermore, we demonstrated that kavain, an active constituent of Piper methysticum Forst., effectively ablates GSC-like cells’ (such as CD133 + U87, U251, DK, and AM38 populations) radioresistance in a LITAF-dependent manner.
Conclusion
In mechanism, our results indicated that 1) the elevation of LITAF in GBM cells activates the NF-κB pathway to promote mesenchymal transition, and 2) kavain disturbs STAT6B/LITAF protein interaction and then expels LITAF from the nucleus. Therefore, we consider that kavain may be a potential candidate to develop an irradiation therapy adjuvant for GBM.






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon request.
References
R. Batash et al., Glioblastoma Multiforme, Diagnosis and Treatment Recent Literature Review. Curr. Med. Chem. 24(27), 3002–3009 (2017)
D.A. Reardon et al., Immunomodulation for glioblastoma. Curr. Opin. Neurol. 30(3), 361–369 (2017)
Melhem, J.M., et al., Updates in IDH-Wildtype Glioblastoma. Neurotherapeutics. 31,1–19 (2022). https://doi.org/10.1007/s13311-022-01251-6
O.G. Taylor, J.S. Brzozowski, K.A. Skelding, Glioblastoma Multiforme: An Overview of Emerging Therapeutic Targets. Front. Oncol. 9, 963 (2019)
C. Alifieris, D.T. Trafalis, Glioblastoma multiforme: Pathogenesis and treatment. Pharmacol. Ther. 152, 63–82 (2015)
A. Malmstrom et al., Temozolomide versus standard 6-week radiotherapy versus hypofractionated radiotherapy in patients older than 60 years with glioblastoma: the Nordic randomised, phase 3 trial. Lancet Oncol. 13(9), 916–926 (2012)
L. Cheng et al., L1CAM regulates DNA damage checkpoint response of glioblastoma stem cells through NBS1. EMBO J. 30(5), 800–813 (2011)
M.E. Hardee et al., Resistance of glioblastoma-initiating cells to radiation mediated by the tumor microenvironment can be abolished by inhibiting transforming growth factor-beta. Cancer Res. 72(16), 4119–4129 (2012)
S.P. Caragher, S. Sachdev, A. Ahmed, Radiotherapy and Glioma Stem Cells: Searching for Chinks in Cellular Armor. Curr. Stem Cell Rep. 3(4), 348–357 (2017)
J.A. Fritzell et al., Role of DNA mismatch repair in the cytotoxicity of ionizing radiation. Cancer Res. 57(22), 5143–5147 (1997)
S. Bao et al., Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 444(7120), 756–760 (2006)
F. De Bacco et al., MET inhibition overcomes radiation resistance of glioblastoma stem-like cells. EMBO Mol. Med. 8(5), 550–568 (2016)
K. Tamura et al., Expansion of CD133-positive glioma cells in recurrent de novo glioblastomas after radiotherapy and chemotherapy. J. Neurosurg. 119(5), 1145–1155 (2013)
S.K. Singh et al., Identification of human brain tumour initiating cells. Nature 432(7015), 396–401 (2004)
A. Wyllie, Apoptosis. Clues in the p53 murder mystery. Nature 389(6648), 237–8 (1997)
F. Myokai et al., A novel lipopolysaccharide-induced transcription factor regulating tumor necrosis factor alpha gene expression: molecular cloning, sequencing, characterization, and chromosomal assignment. Proc. Natl. Acad. Sci. U. S. A. 96(8), 4518–4523 (1999)
X. Tang, M.J. Fenton, S. Amar, Identification and functional characterization of a novel binding site on TNF-alpha promoter. Proc. Natl. Acad. Sci. USA 100(7), 4096–4101 (2003)
X. Tang et al., LPS induces the interaction of a transcription factor, LPS-induced TNF-alpha factor, and STAT6(B) with effects on multiple cytokines. Proc. Natl. Acad. Sci. USA 102(14), 5132–5137 (2005)
K.P.L. Bhat et al., Mesenchymal differentiation mediated by NF-κB promotes radiation resistance in glioblastoma. Cancer Cell 24(3), 331–346 (2013)
D.W. Lee et al., The NF-kappaB RelB protein is an oncogenic driver of mesenchymal glioma. PLoS ONE 8(2), e57489 (2013)
J. Ji et al., TRIM22 activates NF-κB signaling in glioblastoma by accelerating the degradation of IκBα. Cell Death Differ. 28(1), 367–381 (2021)
J. Lee, E. Hoxha, H.R. Song, A novel NFIA-NFκB feed-forward loop contributes to glioblastoma cell survival. Neuro. Oncol. 19(4), 524–534 (2017)
D.N. Louis et al., The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta. Neuropathol. 131(6), 803–820 (2016)
K.P.L. Bhat et al., Mesenchymal differentiation mediated by NF-kappaB promotes radiation resistance in glioblastoma. Cancer Cell 24(3), 331–346 (2013)
M.S. Carro et al., The transcriptional network for mesenchymal transformation of brain tumours. Nature 463(7279), 318–325 (2010)
X. Tang, S. Amar, Kavain Involvement in LPS-Induced Signaling Pathways. J. Cell Biochem. 117(10), 2272–2280 (2016)
X. Tang, S. Amar, Kavain Inhibition of LPS-Induced TNF-alpha via ERK/LITAF. Toxicol. Res. (Camb.) 5(1), 188–196 (2016)
P. Wang et al., Enzymes and Pathways of Kavain Bioactivation and Biotransformation. Chem. Res. Toxicol. 32(7), 1335–1342 (2019)
J. Zou et al., Lipopolysaccharide-induced tumor necrosis factor-alpha factor enhances inflammation and is associated with cancer (Review). Mol. Med. Rep. 12(5), 6399–6404 (2015)
A.J. Shirk et al., SIMPLE interacts with NEDD4 and TSG101: evidence for a role in lysosomal sorting and implications for Charcot-Marie-Tooth disease. J. Neurosci. Res. 82(1), 43–50 (2005)
S.M. Lee, L.S. Chin, L. Li, Charcot-Marie-Tooth disease-linked protein SIMPLE functions with the ESCRT machinery in endosomal trafficking. J. Cell. Biol. 199(5), 799–816 (2012)
M.H. Pittler, E. Ernst, Efficacy of kava extract for treating anxiety: systematic review and meta-analysis. J. Clin. Psychopharmacol. 20(1), 84–89 (2000)
C.M. White, The Pharmacology, Pharmacokinetics, Efficacy, and Adverse Events Associated With Kava. J. Clin. Pharmacol. 58(11), 1396–1405 (2018)
M.P. Pollastri et al., Identification and characterization of kava-derived compounds mediating TNF-alpha suppression. Chem. Biol. Drug Des. 74(2), 121–128 (2009)
Funding
This study was supported by Cyrus Tang Foundation and Shanghai Municipal Commission of Health and Family Planning(20194Y0370).
Author information
Authors and Affiliations
Contributions
Investigation and validation: Jianzhong Yu, Jun Shi, Hui Zeng, Lin Ge,
Data curation: Jianzhong Yu, Jun Shi, Hui Zeng, Lin Ge,
Writing—original draft preparation: Jianzhong Yuand Jun Shi.
Writing—review and editing: Hao Li and Xiongwei Wang.
Supervision: Hao Li and Xiongwei Wang.
Funding acquisition: Hao Li and Jun Shi.
Corresponding authors
Ethics declarations
Ethical approval and Consent to participate
All the protocols involved in this study have been approved by the ethics committee of the Children`s Hospital of Fudan University. All families of involved participants have signed the written informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Availability of supporting data
The data sets supporting the results of this article are included within the article and its additional files.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
13402_2022_743_MOESM1_ESM.pdf
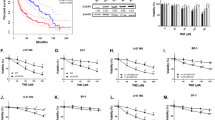
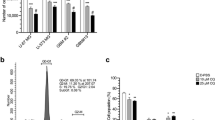
Supplementary file1 Fig.S1 LITAF expression is elevated in tumors of IDH-wildtype GBM patients. A. LITAF-based overall survival analysis of gliomas. Data were collected from the CGGA database. The medium of LITAF expression level was used as a cutoff value. B. The Violin plot showed the association between IDH-mutation/1p9q-codeletion status and LITAF expression. Data from the CGGA database were used (n=325), **P<0.01 and ***P<0.001 by one-way ANOVA. IDH-mutation, IDH1/2 mutation; MGMTp_methylation, MGMT promoter methylation. C. UMAP plot showed the association between LITAF expression and the indicated clinical features in detail. Data were collected from the CGGA database (n=325). MGMTp_methylation, MGMT promoter methylation; rGBM, recurrent glioblastoma; A, astrocytoma; O, oligodendroglioma; OA, oligo-astrocytoma; AOA, anaplastic oligo-astrocytoma; AA: anaplastic astrocytoma; rGBM: recurrent glioblastoma; rAA, recurrent anaplastic astrocytoma; rA, recurrent astrocytoma; AO, anaplastic oligodendroglioma; rAO, recurrent anaplastic oligodendroglioma; rO, recurrent oligodendroglioma; rAOA, recurrent anaplastic oligo-astrocytoma. The dashed circle indicates IDH-wildtype GBM samples. Fig.S2 LITAF expression is elevated in tumors of IDH-wildtype GBM patients. A-B. UMAP plot showed the association between LITAF expression and the indicated clinical features in detail. RNA_array (N=475) and RNA_seq (n=702) datasheets from the TCGA database were used. The dashed circle indicates IDH-wildtype GBM samples. C-D. The Violin plot showed the association between LITAF expression and the indicated clinical features. The data same as A-B from the TCGA database were used. *P<0.05 and ***P<0.001 by one-way ANOVA. Fig.S3 LITAF is up-regulated in glioma stem cells. A. Western blot showed the protein level of LITFA glioma cell lines. N/T, pooled protein samples of normal (N)/tumor (T) tissues. B. Count number distribution of CD133+ cells in the indicated glioma cell lines. ~5000 glioma cells (U87, U251, DK and AM38) were subjected to CD133-labeled FACS for sorting the CD133+ GSC-like cells. C. Cell growth curve determined by MTT assay. The parental tumor cells and CD133+ GSC-like cells obtained in B were used for cell growth determination at the indicated time points. n=3, *P<0.05, **P<0.01 and ***P<0.001 by two-way ANOVA. D. Wound-healing assay determined the migration ability of parental tumor cells and CD133+ GSC-like cells. Cells in B were seeded on a 24-well plate and scratched using 10-μl tips after 12hrs of seeding. n=3; ns, no significance, *P<0.05 by student’s t-test. E. The cell cycle phase was determined by PI staining. CD133+ U87/U251 GSC-like cells in B were used. 2N indicates the G1 phase; 4N indicates the G2/M phase. n=3, *P<0.05 by student's t-test. F-G. Colony formation assay determined tumorigenicity of parental tumor cells and CD133+ GSC-like cells in vitro. The same cells in B were used. Colony formation assays were carried out in 2D (D) and 3D (E) cultures. n=6, **P<0.01 and ***P<0.001 by student's t-test. Fig.S4 LITAF contributes to tumorigenesis of glioma stem cells. A. Cell growth curve determined by MTT assay. The parental tumor cells and CD133+ DK/AM38 GSC-like cells were used for cell growth determination at the indicated time points. n=3, *P<0.05 and **P<0.01 by two-way ANOVA. B. The cell cycle phase was determined by PI staining. CD133+ DK/AM38 GSC-like cells in A were used. 2N indicates the G1 phase; 4N indicates the G2/M phase. n=3, *P<0.05 and **P<0.01 by student's t-test. C. Wound-healing assay determined the migration ability of parental and CD133+ DK/AM38 GSC-like cells. Cells in A were seeded on a 24-well plate and scratched using 10-μl tips after 12hrs of seeding. n=3, *P<0.05 and **P<0.01 by student’s t-test. D-E. Colony formation assay determined tumorigenicity of parental and CD133+ DK/AM38 GSC-like cells in vitro. The same cells in A were used. Colony formation assays were carried out in 2D (D) and 3D (E) cultures. n=6; ns, no significance, **P<0.01 and ***P<0.001 by student's t-test. Fig.S5 LITAF expression involves cytokines produced by the NF-κB pathway. A. The expression of STAT3 and TAZ in GBM and normal controls. Expression data of STAT3 and TAZ were downloaded from the public database of Gene Expression Profiling Interactive Analysis (GEPIA). B. qPCR determined the mRNA level of STAT3 and TAZ in specimens of glioma and normal controls. NC, adjacent normal controls; T, gliomas (both primary and recurrent gliomas). n=10. **P<0.01 and ***P<0.001 by student's t-test. C. Western blot showed the protein level of STAT3 and TAZ in glioma specimens. Samples are the same as D. N, adjacent normal controls; T, gliomas (both primary and recurrent gliomas). n=10, *P<0.05 and **P<0.01 by student's t-test. D. IF determined the subcellular localization of LITAF in A431 and U251 cells. Data were downloaded from the public database of the Human Protein Atlas. Scale bar, 10 μm. E. The expression correlation of LITAF and mesenchymal markers. A Scatter plot was generated using the GEPIA correlation analysis tool. GBM (n=163) and normal brain tissues (n=207, from a database of Genotype-Tissue Expression Project). R, Pearson coefficient. Fig.S6 LITAF elevation contributes to radioresistance of GBM cells. A. The cell cycle phase was determined by PI staining. U87 cell lines received IR treatment with a dose of 4Gy, and 24 hrs later, PI staining was performed. 2N indicates the G1 phase; 4N indicates the G2/M phase. The percentage of cells in the G2/M phase was analyzed (Right). n=3, *P<0.05 and ***P<0.001 by student's t-test. B. Cell growth curve determined by MTT assay. U87 cell lines received IR treatment with a dose of 4 Gy, and the cell growth was determined at the indicated time points. IR, irradiation treatment. n=3; ns, no significance and ***P<0.001 by two-way ANOVA. C. WB showed the indicated protein levels in U87 cells. Cells were treated as B. n=3, **P<0.01 and ***P<0.001 by the student's t-test. D. EdU staining determined the proliferation of indicated cells. Cells were treated as B. EdU-positive cells were counted (Right). n=3, **P<0.01 and ***P<0.001 by student's t-test. E. Soft agar assay determined tumorigenicity of indicated cells in vitro. Colony formation (Left) and size (Right) were analyzed. n=6, **P<0.01 and ***P<0.001 by student's t-test. F. Tumor growth curve of U87 xenograft. n=3; *P<0.05, **P<0.01 and ***P<0.001 by two-way ANOVA followed by posthoc Bonferroni test. Red labels indicated the IR time points.. G. Final tumor weight of U87 xenograft. n=3, *P<0.05 and ***P<0.001 by student's t-test. Fig.S7 LITAF elevation contributes to radioresistance of GBM cells. A. Cell growth curve determined by MTT assay. DK cell lines received IR treatment with a dose of 4 Gy, and the cell growth was determined at the indicated time points. IR, irradiation treatment. n=3; ns, no significance and ***P<0.001 by two-way ANOVA. B. The cell cycle phase was determined by PI staining. Indicated cell lines received IR treatment with a dose of 4Gy, and 24 hrs later, PI staining was performed. .2N indicates the G1 phase; 4N indicates the G2/M phase. The percentage of cells in the G2/M phase was analyzed (Right). n=3, *P<0.05 and **P<0.01 by student's t-test. C. The cell cycle phase was determined by PI staining. Indicated cell lines received treatment as in A. oeLITAF, overexpressed LITAF. The percentage of cells in the G2/M phase was analyzed (Right). n=3, *P<0.05 and ***P<0.001 by student's t-test. D. WB showed the indicated protein levels in DK cells. Cells were treated as D. n=3, *P<0.05 and ***P<0.001 by the student's t-test. E. EdU staining determined the proliferation of indicated cells. Cells were treated as D. EdU-positive cells were counted (Right). n=3, **P<0.01 and ***P<0.001 by student's t-test. F. Soft agar assay determined the tumorigenicity of DK cells in vitro. Colony formation (Left) and size (Right) were analyzed. n=6, **P<0.01 and ***P<0.001 by student's t-test. G. Tumor growth curve (Left) and final tumor weight (Right) of the indicated DK xenograft. n=3; **P<0.01 and ***P<0.001 by two-way ANOVA followed by posthoc Bonferroni test. Red labels indicated the IR time points. Fig.S8 LITAF elevation contributes to radioresistance of GSC-like cells. A-D. Colony formation assay determined tumorigenicity of parental and CD133+ U87/U251(A, C) and DK/AM38 (B , D) GSC-like cells in vitro. The same cells in A were used. Colony formation assays were carried out in 2D (A, B) and 3D (C, D) cultures. The indicated cell lines received IR treatment with a dose of 4 Gy. IR, irradiation treatment. n=6; ns, no significance, *P<0.05, **P<0.01 and ***P<0.001 by student's t-test. Fig.S9 Kavain weakens the radiation resistance of GSC-like cells. A. WB showed the LITAF protein levels in different U87 cell lines. Three independent targets (based on CRISPR/Cas9) were transfected into U87 cells respectively. #, single clone; T1/2/3, target 1/2/3. B. Sanger sequencing confirmed LITAF-/- single clones in A. Arrow indicates the mutant site in the LITAF gene. C-D. Colony formation assay determined tumorigenicity of parental and CD133+ U87/U251 and DK/AM38 GSC-like cells in vitro. The same cells in A were used. Colony formation assays were carried out in 2D (C) and 3D (D) cultures. The indicated cell lines were pre-treated with kavain (50 μg/ml) for 8 hr, and then, received IR treatment with a dose of 4 Gy. IR, irradiation treatment. n=6; *P<0.05, **P<0.01 and ***P<0.001 by student's t-test. Fig.S10 Kavain suppresses NF-κB signaling by targeting LITAF. A. Protein interaction between ectopic LITAF and STAT6B was determined by Co-IP. CD133+ U251 GSC-like cells were treated with kavain (50 μg/ml) for 8 hrs and then were subjected to Co-IP. B. WB showed the protein level of indicated genes in U87 cell lines. Cells received treatment as Fig. 6C.(PDF 18845 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, J., Shi, J., Yuan, F. et al. Kavain ablates the radio-resistance of IDH-wildtype glioblastoma by targeting LITAF/NF-κB pathway. Cell Oncol. 46, 179–193 (2023). https://doi.org/10.1007/s13402-022-00743-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-022-00743-z