Abstract
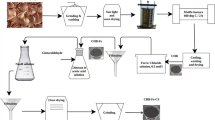
The present study aims to use coconut fruit shell biochar (CFSB), an efficient sorbent for Rhodamine B dye (RhB) elimination from aqueous solution. The availability of functional groups and morphology of CFSB surface were analyzed by applying Fourier transform infrared spectroscopy (FTIR) and scanning electron microscopy (SEM). Batch experiments were executed to test impact of pH, contact time, concentration of dye, biochar amount, agitation speed, temperature on RhB dye elimination from aqueous solution by CFSB. Maximum 96% RhB dye removal was reported at pH 2 by CFSB with RhB dye (250 mg/L concentration). Experimental results were found with best agreement with pseudo second order kinetic model. Equilibrium data were explained with Langmuir, Freundlich and Temkin models but results demonstrated best fit with Langmuir model with a higher correlation coefficient (R2 = 0.9818) and 8.1 mg/g monolayer RhB dye uptake potential. Thermodynamic assessment depicted Gibb's free energy change (ΔG°) had negative values, confirming spontaneity and feasibility of sorption process. Enthalpy (ΔH°) and entropy (ΔS°) changes were 32.039 kJ mol−1 and 92.802 J K−1), respectively. More importantly, even after five adsorption and desorption cycles, CFSB reflected 43% uptake capacity for RhB dye, suggesting that CFSB has a good practical application prospects. Phytotoxicity investigation revealed that CFSB treated solution of RhB dye increased growth of mung bean seedling and biochemical constituents. Therefore, coconut fruit shell biochar can be applied as economic green material for removal of RhB dye from contaminated water and transform industrial effluents into reusable asset.
Graphical Abstract









Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Yusuf TL, Orimolade BO, Masekela D, Mamba B, Mabuba N (2022) The application of photoelectrocatalysis in the degradation of rhodamine B in aqueous solutions: a review. RSC Adv 12:26176–26191. https://doi.org/10.1039/D2RA04236C
Singh S, Parveen N, Gupta H (2018) Adsorptive decontamination of Rhodamine-B from water using banana peel powder: a biosorbent. Environ Technol Innov 12:189–195. https://doi.org/10.1016/j.eti.2018.09.001
Smith CD, Jackson K, Peters H, Herrera Lima S (2020) Lack of safe drinking water for lake chapala basin communities in Mexico inhibits progress toward sustainable development goals 3 and 6. Int J Environ Res Public Health 17:8328. https://doi.org/10.3390/ijerph17228328
Warren-Vega WM, Campos-Rodríguez A, Zárate-Guzmán AI, Romero-Cano LA (2023) A current review of water pollutants in american continent: trends and perspectives in detection, health risks, and treatment technologies. Int J Environ Res Public Health 20(5):4499. https://doi.org/10.3390/ijerph20054499
Bentahar Y, Lomenech C, Mija A, de Jong E, Bonjour E, Jame P, Hurel C (2022) Adsorptive properties of biochar derived from biorefinery industry for basic dye removal. Biomass Conv. Bioref. https://doi.org/10.1007/s13399-022-03398-w
Sarkar S, Echeverria-Vega A, Banerjee A, Bandopadhyay R (2021) Decolourisation and biodegradation of textile di-azo dye congo red by Chryseobacterium geocarposphaerae DD3. Sustainability 13(19):10850. https://doi.org/10.3390/su131910850
Al-Buriahi AK, Al-Gheethi AA, Senthil Kumar P, Radin Mohamed RMS, Yusof H, Alshalif AF, Khalifa NA (2022) Elimination of rhodamine B from textile wastewater using nanoparticle photocatalysts: a review for sustainable approaches. Chemosphere 287(2):132162. https://doi.org/10.1016/j.chemosphere.2021.132162
Velusamy S, Roy A, Sundaram S, Kumar MT (2021) A review on heavy metal ions and containing dyes removal through graphene oxide-based adsorption strategies for textile wastewater treatment. Chem Rec 21(7):1570–1610. https://doi.org/10.1002/tcr.202000153
Ledakowicz S, Pazdzior K (2021) Recent achievements in dyes removal focused on advanced oxidation processes integrated with biological methods. Molecules 26(4):870. https://doi.org/10.3390/molecules26040870
Partal R, Basturk I, Hocaoglu SM, Baban A, Yilmaz E (2022) Recovery of water and reusable salt solution from reverse osmosis brine in textile industry: a case study. Water Resour Ind 27:100174. https://doi.org/10.1016/j.wri.2022.100174
Hassan MM, Carr CM (2018) A critical review on recent advancements of the removal of reactive dyes from dyehouse effluent by ion-exchange adsorbents. Chemosphere 209:201–219. https://doi.org/10.1016/j.chemosphere.2018.06.043
Al-Gheethi AA, Azhar QM, Kumar PS, Yusuf AA, Al-Buriahi AK, Radin Mohamed RMS, Al-shaibani MM (2022) Sustainable approaches for removing Rhodamine B dye using agricultural waste adsorbents: a review. Chemosphere 287(2):132080. https://doi.org/10.1016/j.chemosphere.2021.132080
Reck IM, Baptista ATA, Paixao RM, Bergamasco R, Vieira MF, Vieira AMS (2020) Application of magnetic coagulant based on fractionated protein of Moringa oleifera Lam. seeds for aqueous solutions treatment containing synthetic dyes. Environ Sci Pollut Res 27 (11): 12192–12201. https://doi.org/10.1007/s11356-020-07638-2.
Tariq M, Muhammad M, Khan J, Raziq A, Uddin MK, Niaz A, Ahmed SS, Rahim A (2020) Removal of Rhodamine B dye from aqueous solutions using photo-fenton processes and novel Ni-Cu@MWCNTs photocatalyst. J Mol Liq 312:113399. https://doi.org/10.1016/j.molliq.2020.113399
Adegoke KA, Adegoke OR, Araoye AO, Ogunmodede J, Agboola OS, Bello OS (2022) Engineered raw, carbonaceous, and modified biomass-based adsorbents for Rhodamine B dye removal from water and wastewater. Bioresour Technol Rep 18:101082. https://doi.org/10.1016/j.biteb.2022.101082
Ghibate R, Senhaji O, Taouil R (2021) Kinetic and thermodynamic approaches on Rhodamine B adsorption onto pomegranate peel. Case Stud Chem Environ Eng 3:100078. https://doi.org/10.1016/j.cscee.2020.100078
Skjolding LM, LvG J, Dyhr KS, Koppl CJ, McKnight US, Bauer-Gottwein P, Mayer P, Bjerg PL, Baun A (2021) Assessing the aquatic toxicity and environmental safety of tracer compounds Rhodamine B and Rhodamine WT. Water Res 197:117109. https://doi.org/10.1016/j.watres.2021.117109
Cheng YY, Tsai TH (2017) Pharmacokinetics and biodistribution of the illegal food colorant Rhodamine B in rats. J Agric Food Chem 65(5):1078–1085. https://doi.org/10.1021/acs.jafc.6b04975
Sharma J, Sharma S, Bhatt U, Soni V (2022) Toxic effects of Rhodamine B on antioxidant system and photosynthesis of Hydrilla Verticillata. J Hazard Mater Lett 3:100069. https://doi.org/10.1016/j.hazl.2022.100069
Sudarshan S, Bharti VS, Harikrishnan S, Shukla SP, Rathi Bhuvaneswari G (2022) Eco-toxicological effect of a commercial dye Rhodamine B on freshwater microalgae Chlorella vulgaris. Arch Microbiol 204(10):658. https://doi.org/10.1007/s00203-022-03254-5
Kooh MRR, Dahri MK, Lim LBL (2016) Jackfruit seed as a sustainable adsorbent for the removal of Rhodamine B dye. J Environ Biotechnol Res 4:7–16
Webb JM, Hansen WH, Desmond A, Fitzhugh OG (1961) Biochemical and toxicologic studies of Rhodamine B and 3,6-Diaminofluoran. Toxicol Appl Pharmacol 3(6):696–706. https://doi.org/10.1016/0041-008X(61)90033-3
Sulistina DR, Ratnawati R, Wiyasa IWA (2014) Rhodamine B increases hypothalamic cell apoptosis and disrupts hormonal balance in rats. Asian Pac J Reprod 3(3):180–183. https://doi.org/10.1016/S2305-0500(14)60023-3
Wu J, Yan X, Li L, Gu J, Zhang T, Tian L, Su X, Lin Z (2021) High-efficiency adsorption of Cr (VI) and RhB by hierarchical porous carbon prepared from coal gangue. Chemosphere 275:130008. https://doi.org/10.1016/j.chemosphere.2021.130008
Zhang J, Tong H, Pei W, Liu W, Shi F, Li Y, Huo Y (2021) Integrated photocatalysis-adsorption-membrane separation in rotating reactor for synergistic removal of RhB. Chemosphere 270:129424. https://doi.org/10.1016/j.chemosphere.2020.129424
Pai S, Kini MS, Selvaraj R (2021) A review on adsorptive removal of dyes from wastewater by hydroxyapatite nanocomposites. Environ Sci Pollut Res 28:11835–11849. https://doi.org/10.1007/s11356-019-07319-9
Koul B, Yakoob M, Shah MP (2022) Agricultural waste management strategies for environmental sustainability. Environ Res 206:112285. https://doi.org/10.1016/j.envres.2021.112285
Bilal M, Ihsanullah I, Shah MUH, Reddy AVB, Aminabhavi TM (2022) Recent advances in the removal of dyes from wastewater using low-cost adsorbents. J Environ Manage 321:115981. https://doi.org/10.1016/j.jenvman.2022.115981
Kapoor RT, Rafatullah M, Tajarudin HA, Siddiqui MR, Alam M (2023) Treatment of Direct Red 28 Dye through Phoenix dactylifera L. fruit seed biochar: equilibrium, kinetics, thermodynamics, and phytotoxicity studies. Sustainability. 15(21):15266. https://doi.org/10.3390/su152115266
Qin C, Wang H, Yuan X, Xiong T, Zhang J, Zhang J (2020) Understanding structure-performance correlation of biochar materials in environmental remediation and electrochemical devices. Chem Eng J 382:122977. https://doi.org/10.1016/j.cej.2019.122977
Eleryan A, Yılmaz M, El-Nemr MA, Ragab S, Helal M, Hassaan MA, El Nemr A (2022) Mandarin Biochar-TETA (MBT) prepared from Citrus reticulata peels for adsorption of Acid Yellow 11 dye from water. Sci Rep 12:17797. https://doi.org/10.1038/s41598-022-22359-x
Sathya K, Jayalakshmi H, Reddy SN, Venkataratnam M, Bandhu D (2023) Effective removal of Congo red dye using adsorbent prepared from bio-waste: isotherm, kinetic, and thermodynamic studies. Biomass Conv Bioref. https://doi.org/10.1007/s13399-023-05213-6
Kim DY, Jung GB (2023) Effects of pyrolysis and ball-milling on the physicochemical and rhodamine b removal characteristics of rice-bran-derived biochar. Appl Sci 13:4288. https://doi.org/10.3390/app13074288
Hou Y, Huang G, Li J, Yang Q, Huang S, Cai J (2019) Hydrothermal conversion of bamboo shoot shell to biochar: preliminary studies of adsorption equilibrium and kinetics for rhodamine B removal. J Anal Appl Pyrol 143:104694. https://doi.org/10.1016/j.jaap.2019.104694
Wang E, Huang L, Yuan X, Zhang X, Guo L, Xue C, Chen X (2021) Nutritional, phytochemical and antioxidant properties of 24 mung bean genotypes. Food Prod Process Nutr 3:28. https://doi.org/10.1186/s43014-021-00073-x
Cebrian G, Condon S, Manas P (2017) Physiology of the inactivation of vegetative bacteria by thermal treatments: mode of action, influence of environmental factors and inactivation kinetics. Foods 6:107. https://doi.org/10.3390/foods6120107
Giri BS, Gun S, Pandey S, Trivedi A, Kapoor RT, Singh RP, Abdeldayem OM, Rene ER, Yadav S, Chaturvedi P, Sharma N, Singh RS (2020) Reusability of brilliant green dye contaminated wastewater using corncob biochar and Brevibacillus parabrevis: hybrid treatment and kinetic studies. Bioengineered 11:743–758. https://doi.org/10.1080/21655979.2020.1788353
Rivera-Utrilla J, Bautista-Toledo I, Ferro-García MA, Moreno-Castilla C (2001) Activated carbon surface modifications by adsorption of bacteria and their effect on aqueous lead adsorption. J Chem Technol Biotechnol 76:1209–1215
Lagergren SK (1898) About the theory of so-called adsorption of soluble substances. Sven Vetenskapsakad Handingarl 24:1–39
Lopez-Luna J, Ramírez-Montes LE, Martinez-Vargas S, Martínez AI, Mijangos-Ricardez OF, González-Chávez MdCA, Carrillo-González R, Solís-Domínguez FA, Cuevas-Díaz MdC, Vázquez-Hipólito V (2019) Linear and nonlinear kinetic and isotherm adsorption models for arsenic removal by manganese ferrite nanoparticles. SN Appl Sci 1:950. https://doi.org/10.1007/s42452-019-0977-3
Shikuku VO, Zanella R, Kowenje CO, Donato FF, Bandeira NM, Prestes OD (2018) Single and binary adsorption of sulfonamide antibiotics onto iron-modified clay: linear and nonlinear isotherms, kinetics, thermodynamics, and mechanistic studies. Appl Water Sci 8:175. https://doi.org/10.1007/s13201-018-0825-4
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Weber WJ Jr, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div 89(2):31–59. https://doi.org/10.1061/JSEDAI.0000430
Bharathi K, Ramesh S (2013) Removal of dyes using agricultural waste as low-cost adsorbents: a review. Appl Water Sci 3:773–790. https://doi.org/10.1007/s13201-013-0117-y
Ng JCY, Cheung WH, McKay G (2002) Equilibrium studies of the sorption of Cu (II) ions onto chitosan. J Colloid Interface Sci 255:64–74. https://doi.org/10.1006/jcis.2002.8664
Temkin MI, Pyzhev V (1940) Kinetics of ammonia synthesis on promoted iron catalyst. Acta Phys Chim USSR 12:327–356
Kapoor RT, Selvaraju Sivamani S (2021) Exploring the potential of Eucalyptus citriodora biochar against direct red 31 dye and its phytotoxicity assessment. Biomass Convers Biorefin 13:8011–8022. https://doi.org/10.1007/s13399-021-01681-w
ISTA (2008) International rules for seed testing; international seed testing association, ISTA Secretariat. Bassersdorf, Switzerland
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382. https://doi.org/10.1016/0076-6879(87)48036-1
Hedge JE, Hofreiter BT (1962) Carbohydrate chemistry 17. Whistler, R.L. and Be Miller, J. N., Eds., Academic Press, New York. 17–22
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275 (PMID: 14907713)
Shaheen SM, Niazi NK, Hassan NEE, Bibi I, Wang H, Tsang DCW, Ok YS, Bolan N, Rinklebe J (2019) Wood-based biochar for the removal of potentially toxic elements in water and wastewater: a critical review. Int Mater Rev 64(4):216–247. https://doi.org/10.1080/09506608.2018.1473096
Oliveira LMTM, Oliveira LFAM, Sonsin AF, Duarte JLS, Soletti JI, Fonseca EJS, Ribeiro LMO, Meili L (2020) Ultrafast diesel oil spill removal by fibers from silk-cotton tree: characterization and sorption potential evaluation. J Clean Prod 263:121448. https://doi.org/10.1016/J.JCLEPRO.2020.121448
Tran HN, Chao HP, You SJ (2018) Activated carbons from golden shower upon different chemical activation methods: synthesis and characterizations. Adsorpt Sci Technol 36:95–113. https://doi.org/10.1177/0263617416684837
Oyekanmi AA, Ahmad A, Hossain K, Rafatullah M (2019) Statistical optimization for adsorption of Rhodamine B dye from aqueous solutions. J Mol Liq 281:48–58. https://doi.org/10.1016/J.MOLLIQ.2019.02.057
Gunduz G, Oral MA, Akyuz M, Aydemir D, Yaman B, Asik N, Bulbul AS, Allahverdiyev S (2016) Physical, morphological properties and raman spectroscopy of chestnut blight diseased castanea sativa mill. Wood, cerne 22:43–58. https://doi.org/10.1590/01047760201622012101
Vasconcelos KC, Alencar SG, Ferro AB, Oliveira L FAM, Fonseca EJS, Bernardo VB, Zanta C LPS, Duarte J L S, Oliveira L MTM (2023) Novel kapok pods (Ceiba pentandra (l.) Gaerth) adsorbent and its reusability by transformation on hydrochar with an outstanding adsorptive capacity for Rhodamine B removal. Sep Purif Technol 326: 124787. https://doi.org/10.1016/j.seppur.2023.124787
Matias CA, de Oliveira LJGG, Geremias R, Stolberg J, Matias CA, de Oliveira LJGG, Geremias R, Stolberg J (2020) Biosorption of rhodamine b from aqueous solution using araucaria angustifolia sterile bracts. Rev Int Contam Ambient 36: 97–104. https://doi.org/10.20937/RICA.2020.36.53282
Song M, Jin B, Xiao R, Yang L, Wu Y, Zhong Z, Huang Y (2013) The comparison of two activation techniques to prepare activated carbon from corn cob. Biomass Bioenerg 48:250–256. https://doi.org/10.1016/j.biombioe.2012.11.007
Yin M, Pan Y, Pan C (2019) Adsorption properties of graphite oxide for Rhodamine B. Micro Nano Lett 14:1121–1212. https://doi.org/10.1049/mnl.2018.5820open_in_newISSN1750-0443
Lopez-Sanchez P, Martinez-Sanz M, Bonilla MR, Wang D, Gilbert EP, Stokes JR, Gidley MJ (2017) Cellulose-pectin composite hydrogels: intermolecular interactions and material properties depend on order of assembly. Carbohydr Polym 162:71–81. https://doi.org/10.1016/j.carbpol.2017.01.049
Wisniewska M, Chibowski S, Wawrzkiewicz M, Onyszko M, Bogatyrov VCI (2022) Basic Red 46 removal from sewage by carbon and silica based composite: equilibrium, kinetic and electrokinetic studies. Molecules 27:1043. https://doi.org/10.3390/molecules27031043
Kamarehie B, Jafari A, Ghaderpoori M, Amin Karami M, Mousavi K, Ghaderpoury A (2019) Catalytic ozonation process using PAC/γ-Fe2O3 to alizarin red S degradation from aqueous solutions: a batch study. Chem Eng Commun 206(7):898–908. https://doi.org/10.1080/00986445.2018.1537266
Daouda A, Honorine AT, Bertrand NG, Richard D, Domga (2019) Adsorption of Rhodamine B onto orange peel powder. Am J Chem 9:142-149. https://doi.org/10.5923/j.chemistry.20190905.02
Hii SL, Yong SY, Wong CL (2009) Removal of Rhodamine B from aqueous solution by sorption on Turbinaria conoides. J Appl Phycol 21:625–631. https://doi.org/10.1007/s10811-009-9448-3
Gul S, Gul H, Gul M, Khattak R, Rukh G, Khan MS, Aouissi HA (2022) Enhanced adsorption of rhodamine b on biomass of cypress/false cypress (Chamaecyparis lawsoniana) fruit: optimization and kinetic study. Water 14:2987. https://doi.org/10.3390/w14192987
Ren Z, Chen F, Wang B, Song Z, Zhou Z, Ren D (2020) Magnetic biochar from alkali-activated rice straw for removal of rhodamine B from aqueous solution. Environ Eng Res 25(4):536–544. https://doi.org/10.4491/eer.2019.232
Mousavi SA, Kamarehie B, Almasi A, Darvishmotevalli M, Salari M, Moradnia M, Azimi F, Ghaderpoori M, Neyazi Z, Karami MA (2021) Removal of Rhodamine B from aqueous solution by stalk corn activated carbon: adsorption and kinetic study. Biomass Convers Biorefin 9:7927–7936. https://doi.org/10.1007/s13399-021-01628-1
Shah J, Rasul Jan M, Haq A, Khan Y (2013) Removal of rhodamine B from aqueous solutions and wastewater by walnut shells : kinetics, equilibrium and thermodynamics studies. Front Chem Sci Eng 7:428–436. https://doi.org/10.1007/s11705-013-1358-x
Khan M, Siddiqui MR, Otero M, Alshareef SA, Rafatullah M (2020) Removal of rhodamine B from water using a solvent impregnated polymeric dowex 5wx8 resin: statistical optimization and batch adsorption studies. Polymers 12(2):500. https://doi.org/10.3390/polym12020500
Wu J, Yang J, Huang G, Xu C, Lin B (2020) Hydrothermal carbonization synthesis of cassava slag biochar with excellent adsorption performance for Rhodamine B. J Clean Prod 251:119717. https://doi.org/10.1016/j.jclepro.2019.119717
Mondal PAK, MK, (2022) Green synthesis of Fe3O4-onion peel biochar nanocomposites for adsorption of Cr(VI), methylene blue and congo red dye from aqueous solutions. J Mol Liq 349(1):118161. https://doi.org/10.1016/j.molliq.2021.118161
Enaime G, Baçaoui A, Yaacoubi A, Lübken M (2020) Biochar for wastewater treatment-conversion technologies and applications. Appl Sci 10:3492. https://doi.org/10.3390/app10103492
El Haddad M, Mamouni R, Saffaj N, Lazar S (2016) Evaluation of performance of animal bone meal as a new low cost adsorbent for the removal of a cationic dye Rhodamine B from aqueous solutions. J Saudi Chem Soc 20:S53–S59. https://doi.org/10.1016/j.jscs.2012.08.005
Kaya N, Yıldız Z, Ceylan S (2018) Preparation and characterisation of biochar from hazelnut shell and its adsorption properties for methylene blue dye. Politek Derg 21:765–776. https://doi.org/10.2339/politeknik.386963
Goswami L, Kushwaha A, Kafle SR, Kim BS (2022) Surface modification of biochar for dye removal from wastewater. Catalysts 12:817. https://doi.org/10.3390/catal12080817
Saigl Z M, Ahmed A M (2021) Separation of Rhodamine B dye from aqueous media using natural pomegranate peels. Indones J Chem 21(1): 212–224. https://doi.org/10.22146/ijc.58592
Neupane S, Ramesh ST, Gandhimathi R, Nidheesh PV (2015) Pineapple leaf (Ananas comosus) powder as a biosorbent for the removal of crystal violet from aqueous solution. Desalination Water Treat 54:2041–2054. https://doi.org/10.1080/19443994.2014.903867
Ambaye TG, Vaccari M, van Hullebusch ED, Amrane A, Rtimi S (2021) Mechanisms and adsorption capacities of biochar for the removal of organic and inorganic pollutants from industrial wastewater. Int J Environ Sci Technol 18:3273–3294. https://doi.org/10.1007/s13762-020-03060-w
Roy TK, Mondal NK (2017) Biosorption of congo red from aqueous solution onto burned root of Eichhornia crassipes biomass. Appl Water Sci 7:1841–1854. https://doi.org/10.1007/s13201-015-0358-z
Hu Z, Chen H, Ji F, Yuan S (2010) Removal of Congo Red from aqueous solution by cattail root. J Hazard Mater 173:292–297. https://doi.org/10.1016/j.jhazmat.2009.08.082
Fu J, Chen Z, Wang M, Liu S, Zhang J, Zhang J, Han R, Xu Q (2015) Adsorption of methylene blue by a high-efficiency adsorbent (polydopamine microspheres): kinetics, isotherm, thermodynamics and mechanism analysis. Chem Eng J 259:53–61. https://doi.org/10.1016/j.cej.2014.07.101
Ozturk G, Silah H (2020) Adsorptive removal of Remazol Brilliant Blue R from water by using a macroporous polystyrene resin: isotherm and kinetic studies. Environ Process 7:479–492. https://doi.org/10.1007/s40710-020-00429-4
Tolkou AK, Trikalioti S, Makrogianni O, Katsoyiannis IA, Kyzas GZ (2023) Magnesium modified activated carbons derived from coconut shells for the removal of fluoride from water. Sustain Chem Pharm 31:100898. https://doi.org/10.1016/j.scp.2022.100898
Mei Y, Xu J, Zhang Y, Li B, Fan S, Xu H (2021) Effect of Fe-N modification on the properties of biochars and their adsorption behavior on tetracycline removal from aqueous solution. Bioresour Technol 325:124732. https://doi.org/10.1016/j.biortech.2021.124732
Guo S, Zou Z, Chen Y, Long X, Liu M, Li X, Tan J, Chen R (2023) Synergistic effect of hydrogen bonding and π-π interaction for enhanced adsorption of rhodamine B from water using corn straw biochar. Environ Pollut 320:121060. https://doi.org/10.1016/j.envpol.2023.121060
Shen K, Gondal MA (2017) Removal of hazardous rhodamine dye from water by adsorption onto exhausted coffee ground. J Saudi Chem Soc 21:S120–S127. https://doi.org/10.1016/j.jscs.2013.11.005
Zare EN, Motahari A, Sillanpaa M (2018) Nanoadsorbents based on conducting polymer nanocomposites with main focus on polyaniline and its derivatives for removal of heavy metal ions/dyes: a review. Environ Res 162:173–195. https://doi.org/10.1016/j.envres.2017.12.025
Cheng N, Wang B, Wu P, Lee X, XingY CM, Gao B (2021) Adsorption of emerging contaminants from water and wastewater by modified biochar: a review. Environ Pollut 273:116448. https://doi.org/10.1016/j.envpol.2021.116448
Du XC, Zhu JH, Quan ZJ, Wang XC (2021) Adsorption of rhodamine B by organic porous materials rich in nitrogen, oxygen, and sulfur heteroatoms. New J Chem 45:3448–3453. https://doi.org/10.1039/D0NJ05750A
Jha P, Modi N, Jobby R, Desai N (2015) Differential expression of antioxidant enzymes during degradation of azo dye reactive black 8 in hairy roots of Physalis minima L. Int J Phytoremediation 17:305–312. https://doi.org/10.1080/15226514.2013.876963
Khan TA, Ali I, Singh V V, Sharma S (2009) Utilization of fly ash as low-cost adsorbent for the removal of methylene blue, malachite green and rhodamine B dyes from textile wastewater. J Environ Prot Sci 3: 11–22. Corpus ID: 54812329
He H, Chai K, Wu T, Qiu Z, Wang S, Hong J (2022) Adsorption of Rhodamine B from simulated waste water onto kaolin-bentonite composites. Materials 15:4058. https://doi.org/10.3390/ma15124058
Zamouche M, Hamdaoui O (2012) Sorption of rhodamine B by cedar cone : effect of pH and ionic strength. Energy Procedia 18:1228–1239. https://doi.org/10.1016/j.egypro.2012.05.138
Adekola FA, Ayodele SB, Inyinbor AA (2019) Efficient Rhodamine B removal using acid and alkaline-activated Musa paradisiaca biochar. Pol J Environ Stud 28 (5): 3063–3070. https://doi.org/10.15244/pjoes/94386
Acknowledgements
The authors are grateful to the Researchers Supporting Project Number (RSP2024R326), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Conceptualization, RTK, MR, and MRS, Methodology, RTK, MR and MRS, Supervision, MR and MA; Writing-original Draft preparation, RTK, MR and MRS; Writing-Review & Editing, MR and MA; Funding acquisition MRS. All authors have read and agreed to the submitted version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no conflict of interest.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kapoor, R.T., Rafatullah, M., Siddiqui, M.R. et al. Adsorptive potential of coconut fruit shell biochar as low-cost adsorbent for sequestration of rhodamine B dye: kinetics, thermodynamics and phytotoxicity studies. Biomass Conv. Bioref. (2024). https://doi.org/10.1007/s13399-024-05688-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-024-05688-x