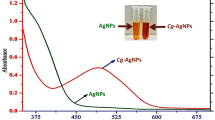
Abstract
Plant extract-mediated synthesis has the advantage of being simple, cost-effective, and environmentally friendly. In this study, we synthesized silver/silver oxide nanoparticles (Ag/Ag2O NPs) using an aqueous Olea Europaea leaf extract as a reducing and stabilizing agent. The Ag/Ag2O NPs were synthesized and the plant extract was processed using only pure water, without the use of any harmful reagents or organic solvents. The synthesized Ag/Ag2O NPs exhibited a spherical morphology with a particle size of 45 nm, and an average crystallite size of 24 nm for the Ag phase and 26 nm for the Ag2O phase. The optical band gap energy of the Ag/Ag2O NPs was calculated to be 2.23 eV. The antimicrobial and antioxidant activities of the Ag/Ag2O NPs were then investigated. The Ag/Ag2O NPs exhibited potent antimicrobial activity against Escherichia coli (E.coli), Staphylococcus aureus (Sa), and Pseudomonas aeruginosa (Pa) at concentrations of 50–200 μg/mL, with the highest activity observed at a concentration of 200 μg/mL, respectively. Moreover, Ag/Ag2O NPs at a concentration of 2.5 mg/kg body weight during a 28-day study in adult male rats (10–11 weeks old) showed remarkable antioxidant activity and no acute toxicity when administered at different doses of Ag/Ag2O NPs (2.5 and 5 mg/kg body weight). The most effective antioxidant exhibited an IC50 value of 7.043 ± 1.352 µg/ml, a FRAP value of 445.34 ± 7.90 mg E FeSO4/g NPs, and a TAC value of 67.72 ± 2.38 mg E GA/g NPs, indicating it’s ferric reducing antioxidant power (FRAP) and total antioxidant capacity (TAC). Accordingly, the developed Ag/Ag2O NPs can be used as chemotherapeutic drug carriers for antioxidant and antibacterial applications.







Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Gupta P, Rai N, Verma A, Saikia D, Singh SP, Kumar R, Singh SK, Kumar D, Gautam V (2022) Green-based approach to synthesize silver nanoparticles using the fungal endophyte penicillium oxalicum and their antimicrobial, antioxidant, and in vitro anticancer potential. ACS Omega 7(50):46653–46673. https://doi.org/10.1021/acsomega.2c05605
Djamila B, Eddine LS, Abderrhmane B, Nassiba A, Barhoum A (2022) In vitro antioxidant activities of copper mixed oxide (CuO/Cu2O) nanoparticles produced from the leaves of Phoenix dactylifera L. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-022-02743-3
Jeevanandam J, Kiew SF, Boakye-Ansah S, Lau SY, Barhoum A, Danquah MK, Rodrigues J (2022) Green approaches for the synthesis of metal and metal oxide nanoparticles using microbial and plant extracts. Nanoscale 14(7):2534–2571. https://doi.org/10.1039/D1NR08144F
Said MM, Rehan M, El-Sheikh SM, Zahran MK, Abdel-Aziz MS, Bechelany M, Barhoum A (2021) Multifunctional hydroxyapatite/silver nanoparticles/cotton gauze for antimicrobial and biomedical applications. Nanomaterials 11(2). https://doi.org/10.3390/nano11020429
Soliman MKY, Salem SS, Abu-Elghait M, Azab MS (2023) Biosynthesis of silver and gold nanoparticles and their efficacy towards antibacterial, antibiofilm, cytotoxicity, and antioxidant activities. Appl Biochem Biotechnol 195(2):1158–1183. https://doi.org/10.1007/s12010-022-04199-7
Simon S, Sibuyi NR, Fadaka AO, Meyer S, Josephs J, Onani MO, Meyer M, Madiehe AM (2022) Biomedical applications of plant extract-synthesized silver nanoparticles. Biomedicines 10(11). https://doi.org/10.3390/biomedicines10112792
Jaswal T, Gupta J (2023) A review on the toxicity of silver nanoparticles on human health. Mater Today: Proc 81:859–863. https://doi.org/10.1016/j.matpr.2021.04.266
Ashique S, Upadhyay A, Hussain A, Bag S, Chaterjee D, Rihan M, Mishra N, Bhatt S, Puri V, Sharma A, Prasher P, Singh SK, Chellappan DK, Gupta G, Dua K (2022) Green biogenic silver nanoparticles, therapeutic uses, recent advances, risk assessment, challenges, and future perspectives. J Drug Delivery Sci Technol 77:103876. https://doi.org/10.1016/j.jddst.2022.103876
Lee SH, Jun B-H (2019) Silver nanoparticles: synthesis and application for nanomedicine. Int J Mol Sci 20(4):865
Tariq M, Mohammad KN, Ahmed B, Siddiqui MA, Lee J (2022) Biological synthesis of silver nanoparticles and prospects in plant disease management. Molecules 27(15). https://doi.org/10.3390/molecules27154754
Li R, Chen Z, Ren N, Wang Y, Wang Y, Yu F (2019) Biosynthesis of silver oxide nanoparticles and their photocatalytic and antimicrobial activity evaluation for wound healing applications in nursing care. J Photochem Photobiol B 199:111593. https://doi.org/10.1016/j.jphotobiol.2019.111593
Danish MSS, Estrella-Pajulas LL, Alemaida IM, Grilli ML, Mikhaylov A, Senjyu T (2022) Green synthesis of silver oxide nanoparticles for photocatalytic environmental remediation and biomedical applications. Metals 12(5):769
Dara PK, Mahadevan R, Digita PA, Visnuvinayagam S, Kumar LRG, Mathew S, Ravishankar CN, Anandan R (2020) Synthesis and biochemical characterization of silver nanoparticles grafted chitosan (Chi-Ag-NPs): in vitro studies on antioxidant and antibacterial applications. SN Appl Sci 2(4):665. https://doi.org/10.1007/s42452-020-2261-y
Akter M, Sikder MT, Rahman MM, Ullah A, Hossain KFB, Banik S, Hosokawa T, Saito T, Kurasaki M (2018) A systematic review on silver nanoparticles-induced cytotoxicity: physicochemical properties and perspectives. J Adv Res 9:1–16. https://doi.org/10.1016/j.jare.2017.10.008
Elyamny S, Eltarahony M, Abu-Serie M, Nabil MM, Kashyout AE-HB (2021) One-pot fabrication of Ag @Ag2O core–shell nanostructures for biosafe antimicrobial and antibiofilm applications. Sci Rep 11(1):22543. https://doi.org/10.1038/s41598-021-01687-4
Ozdal M, Gurkok S (2022) Recent advances in nanoparticles as antibacterial agent. ADMET DMPK 10(2):115–129. https://doi.org/10.5599/admet.1172
Yin IX, Zhang J, Zhao IS, Mei ML, Li Q, Chu CH (2020) The antibacterial mechanism of silver nanoparticles and its application in dentistry. Int J Nanomed 15:2555–2562. https://doi.org/10.2147/ijn.s246764
Kumar B, Smita K, Cumbal L, Debut A (2016) Ficus carica (Fig) fruit mediated green synthesis of silver nanoparticles and its antioxidant activity: a comparison of thermal and ultrasonication approach. BioNanoScience 6(1):15–21. https://doi.org/10.1007/s12668-016-0193-1
Singh JP, Kaur A, Singh N, Nim L, Shevkani K, Kaur H, Arora DS (2016) In vitro antioxidant and antimicrobial properties of jambolan (Syzygium cumini) fruit polyphenols. LWT Food Sci Technol 65:1025–1030. https://doi.org/10.1016/j.lwt.2015.09.038
Asadi S, Ahmadiani A, Esmaeili MA, Sonboli A, Ansari N, Khodagholi F (2010) In vitro antioxidant activities and an investigation of neuroprotection by six Salvia species from Iran: a comparative study. Food Chem Toxicol 48(5):1341–1349. https://doi.org/10.1016/j.fct.2010.02.035
Bauer AW, Kirby WM, Sherris JC, Turck M (1966) Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol 45(4):493–496
Della Rosa RJ, Stannard JN (1964) Acute toxicity as a function of route of administration. Radiat Res Suppl 5:205–215. https://doi.org/10.2307/3583491
Trang NLN, Hoang V-T, Dinh NX, Tam LT, Le VP, Linh DT, Cuong DM, Khi NT, Anh NH, Nhung PT, Le A-T (2021) Novel eco-friendly synthesis of biosilver nanoparticles as a colorimetric probe for highly selective detection of Fe (III) ions in aqueous solution. J Nanomater 2021:5527519. https://doi.org/10.1155/2021/5527519
Laouini SE, Bouafia A, Soldatov AV, Algarni H, Tedjani ML, Ali GAM, Barhoum A (2021) Green synthesized of Ag/Ag(2)O nanoparticles using aqueous leaves extracts of phoenix dactylifera L. and their azo dye photodegradation. Membranes (Basel) 11(7). https://doi.org/10.3390/membranes11070468
Laouini SE, Bouafia A, Soldatov AV, Algarni H, Tedjani ML, Ali GAM, Barhoum A (2021) Green synthesized of Ag/Ag2O nanoparticles using aqueous leaves extracts of phoenix dactylifera L. and their azo dye photodegradation. Membranes 11(7):468
Terea H, Selloum D, Rebiai A, Bouafia A, Ben Mya O (2023) Preparation and characterization of cellulose/ZnO nanoparticles extracted from peanut shells: effects on antibacterial and antifungal activities. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-023-03959-7
Joesna G, Saravanan P, Ferin RZ, Gunachitra T, Sankar D, Tamilselvan S, Meena M, SenthilKannan K, Vimalan M, Mohamed MG (2022) Domestic microwave supported green synthesis of ZnO nanoparticles for electronic, mechano, rheological and frequency intensifying applications. J Mater Sci Mater Electron 33(17):14144–14158. https://doi.org/10.1007/s10854-022-08344-0
Al-Bayati FA (2009) Isolation and identification of antimicrobial compound from Mentha longifolia L. leaves grown wild in Iraq. Ann Clin Microbiol Antimicrob 8:20. https://doi.org/10.1186/1476-0711-8-20
Hankett JM, Liu Y, Zhang X, Zhang C, Chen Z (2013) Molecular level studies of polymer behaviors at the water interface using sum frequency generation vibrational spectroscopy. J Polym Sci B Polym Phys 51(5):311–328. https://doi.org/10.1002/polb.23221
Vijayaraghavan T, Sivasubramanian R, Hussain S, Ashok A (2017) A facile synthesis of LaFeO3-based perovskites and their application towards sensing of neurotransmitters. ChemistrySelect 2(20):5570–5577. https://doi.org/10.1002/slct.201700723
Abbas G, Kumar N, Kumar D, Pandey G (2019) Effect of reaction temperature on shape evolution of palladium nanoparticles and their cytotoxicity against A-549 lung cancer cells. ACS Omega 4(26):21839–21847. https://doi.org/10.1021/acsomega.9b02776
Vu TT, Nguyen PT, Pham NH, Le TH, Nguyen TH, Do DT, La DD (2022) Green synthesis of selenium nanoparticles using cleistocalyx operculatus leaf extract and their acute oral toxicity study. J Compos Sci 6:(10). https://doi.org/10.3390/jcs6100307
Varma R, Vasudevan S (2020) Extraction, characterization, and antimicrobial activity of chitosan from horse mussel modiolus modiolus. ACS Omega 5(32):20224–20230. https://doi.org/10.1021/acsomega.0c01903
Kordy MGM, Abdel-Gabbar M, Soliman HA, Aljohani G, BinSabt M, Ahmed IA, Shaban M (2022) Phyto-capped ag nanoparticles: green synthesis, characterization, and catalytic and antioxidant activities. Nanomaterials 12(3). https://doi.org/10.3390/nano12030373
Dobrucka R, Szymanski M, Przekop R (2019) The study of toxicity effects of biosynthesized silver nanoparticles using Veronica officinalis extract. Int J Environ Sci Technol 16(12):8517–8526. https://doi.org/10.1007/s13762-019-02441-0
Belaiche Y, Khelef A, Laouini SE, Bouafia A, Tedjani ML, Barhoum A (2021) Green synthesis and characterization of silver/silver oxide nanoparticles using aqueous leaves extract of Artemisia herba-alba as reducing and capping agents. Rom J Mater 51(3):342–352
Tedjani ML, Khelef A, Laouini SE, Bouafia A, Albalawi N (2022) Optimizing the antibacterial activity of iron oxide nanoparticles using central composite design. J Inorg Organomet Polym Mater 32(9):3564–3584. https://doi.org/10.1007/s10904-022-02367-0
Nanaei M, Nasseri MA, Allahresani A, Kazemnejadi M (2019) Phoenix dactylifera L. extract: antioxidant activity and its application for green biosynthesis of Ag nanoparticles as a recyclable nanocatalyst for 4-nitrophenol reduction. SN Appl Sci 1(8):853. https://doi.org/10.1007/s42452-019-0895-4
Jose Luis A-T, du Wessel T (2018) The role of UV-visible spectroscopy for phenolic compounds quantification in Winemaking. In: Rosa Lidia S-O, Ángel de la Cruz P-C (eds) Frontiers and new trends in the science of fermented food and beverages. IntechOpen, Rijeka, p Ch. 3. https://doi.org/10.5772/intechopen.79550
Dai J, Mumper RJ (2010) Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules 15(10):7313–7352. https://doi.org/10.3390/molecules15107313
Pfost D, Vardeny Z, Tauc J (1985) Pfost, vardeny, and tauc respond. Phys Rev Lett 54(3):251–251. https://doi.org/10.1103/PhysRevLett.54.251
El-Ghmari B, Farah H, Ech-Chahad A (2021) A new approach for the green biosynthesis of silver oxide nanoparticles Ag2O, characterization and catalytic application. 2021:10. https://doi.org/10.9767/bcrec.16.3.11577.651-660
He L, He T, Farrar S, Ji L, Liu T, Ma X (2017) Antioxidants maintain cellular redox homeostasis by elimination of reactive oxygen species. Cell Physiol Biochem 44(2):532–553. https://doi.org/10.1159/000485089
Temraz A, El-Tantawy WH (2008) Characterization of antioxidant activity of extract from Artemisia vulgaris. Pak J Pharm Sci 21(4):321–326
Baliyan S, Mukherjee R, Priyadarshini A, Vibhuti A, Gupta A, Pandey RP, Chang CM (2022) Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of ficus religiosa. Molecules 27(4). https://doi.org/10.3390/molecules27041326
Ahmad S, Rehman T, Abbasi W, Zaman M (2017) Analysis of antioxidant activity and total phenolic content of some homoeopathic mother tinctures. Indian J Res Homoeopathy 11(1):21–25
Kumar B, Smita K, Cumbal L, Debut A (2017) Green synthesis of silver nanoparticles using Andean blackberry fruit extract. Saudi J Biol Sci 24(1):45–50. https://doi.org/10.1016/j.sjbs.2015.09.006
Daoudi H, Bouafia A, Meneceur S, Laouini SE, Belkhalfa H, Lebbihi R, Selmi B (2022) Secondary metabolite from nigella sativa seeds mediated synthesis of silver oxide nanoparticles for efficient antioxidant and antibacterial activity. J Inorg Organomet Polym Mater 32(11):4223–4236. https://doi.org/10.1007/s10904-022-02393-y
Mathew S, Abraham TE, Zakaria ZA (2015) Reactivity of phenolic compounds towards free radicals under in vitro conditions. J Food Sci Technol 52(9):5790–5798. https://doi.org/10.1007/s13197-014-1704-0
Lava MB, Muddapur UM, Basavegowda N, More SS, More VS (2021) Characterization, anticancer, antibacterial, anti-diabetic and anti-inflammatory activities of green synthesized silver nanoparticles using Justica wynaadensis leaves extract. Mater Today: Proc 46:5942–5947. https://doi.org/10.1016/j.matpr.2020.10.048
Selem E, Mekky AF, Hassanein WA, Reda FM, Selim YA (2022) Antibacterial and antibiofilm effects of silver nanoparticles against the uropathogen Escherichia coli U12. Saudi J Biol Sci 29(11):103457. https://doi.org/10.1016/j.sjbs.2022.103457
Skóra B, Krajewska U, Nowak A, Dziedzic A, Barylyak A, Kus-Liśkiewicz M (2021) Noncytotoxic silver nanoparticles as a new antimicrobial strategy. Sci Rep 11(1):13451. https://doi.org/10.1038/s41598-021-92812-w
Sondi I, Salopek-Sondi B (2004) Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for Gram-negative bacteria. J Colloid Interface Sci 275(1):177–182. https://doi.org/10.1016/j.jcis.2004.02.012
Lee HJ, Yeo SY, Jeong SH (2003) Antibacterial effect of nanosized silver colloidal solution on textile fabrics. J Mater Sci 38(10):2199–2204. https://doi.org/10.1023/A:1023736416361
Recordati C, De Maglie M, Bianchessi S, Argentiere S, Cella C, Mattiello S, Cubadda F, Aureli F, D’Amato M, Raggi A, Lenardi C, Milani P, Scanziani E (2016) Tissue distribution and acute toxicity of silver after single intravenous administration in mice: nano-specific and size-dependent effects. Part Fibre Toxicol 13(1):12. https://doi.org/10.1186/s12989-016-0124-x
Patlolla AK, Hackett D, Tchounwou PB (2015) Silver nanoparticle-induced oxidative stress-dependent toxicity in Sprague-Dawley rats. Mol Cell Biochem 399(1):257–268. https://doi.org/10.1007/s11010-014-2252-7
Cerqueira MA, Vicente AA, Pastrana LM (2018) Chapter 1 - nanotechnology in food packaging: opportunities and challenges. In: Cerqueira MÂPR, Lagaron JM, Pastrana Castro LM, de Oliveira Soares Vicente AAM (eds) Nanomaterials for food packaging. Elsevier, pp 1–11. https://doi.org/10.1016/B978-0-323-51271-8.00001-2
Funding
The authors, Prof. Ahmed Barhoum and Prof. Ali Alsalme, would like to acknowledge the support received from the Egypt–France Joint Program (Imhotep, Project No. 43990SF, 2020-2022), Joint Egyptian Japanese Scientific Cooperation (JEJSC, Project No. 42811) and the Researchers Supporting Project (RSP-2023R78) at King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Conceptualization, M.A., I.M., I.T., A.BOUAFIA., G.G.H., H.A.M., S.M., S.E.L., A.B.; methodology, M.A., I.M., I.T., A.BOUAFIA., G.G.H., H.A.M., S.M., S.E.L., A.B.; validation, M.A., I.M., I.T., A.BOUAFIA., G.G.H., H.A.M., S.M., S.E.L., A.B.; investigation, M.A., I.M., I.T., A.BOUAFIA., G.G.H., H.A.M., S.M., S.E.L., A.B.; resources, M.A., I.M., I.T., A.BOUAFIA., G.G.H., H.A.M., S.M., S.E.L., A.A., A.B.; data curation, M.A., I.M., I.T., A.BOUAFIA., G.G.H., H.A.M., S.M., S.E.L., A.B.; writing—original draft preparation, M.A., I.M., I.T., A.BOUAFIA., G.G.H., H.A.M., S.M., S.E.L., A.B.; writing—review and editing, M.A., I.M., I.T., A.BOUAFIA., G.G.H., H.A.M., S.M., S.E.L., A.A., A.B.; supervision, M.A., I.M., I.T., A.BOUAFIA., G.G.H., H.A.M., S.M., S.E.L., A.B. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Ethical approval statements for animal studies of this work were obtained for a study from the University of El Oued, El Oued 39000, Algeria.
Competing interests
All co-authors have seen and agree with the contents of the manuscript and there is no financial interest to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Azzi, M., Medila, I., Toumi, I. et al. Plant extract-mediated synthesis of Ag/Ag2O nanoparticles using Olea europaea leaf extract: assessing antioxidant, antibacterial, and toxicological properties. Biomass Conv. Bioref. (2023). https://doi.org/10.1007/s13399-023-05093-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-023-05093-w