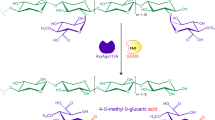
Abstract
The crude β-xylosidase from Aureobasidium pullulans CBS 135684 was immobilized on the surface of the modified spent expanded perlite via (3-aminopropyl) triethoxy-silane and glutaraldehyde cross-linking reaction. Over 80% of the initial free enzyme activity was detected on the bound enzyme. The immobilized β-xylosidase (204.7 ± 6.5 U g-1 of perlite) exhibited transxylosylation activity when sugars from black liquor xylan hydrolysate and a variety of alcohols (C1-6) were used as donor and acceptor molecules, respectively, yielding a number of alkyl xylosides. The synthesized hexyl xyloside exhibited some interesting properties comparable with those of a commercial hexyl glucoside, including an antioxidant activity (IC50 = 8.9 ± 2.0 mg mL-1), wetting time (43 s), and critical micelle concentration value (252 mmol L-1). Optimization for hexyl xyloside synthesis was performed via statistical analysis using a Box and Behnken design. The maximum predicted yield of hexyl xyloside (499.0 ± 1.1 mg g-1 xylan hydrolysate) was obtained with 10% (w/v) xylan hydrolysate, 10 U g-1 immobilized β-xylosidase, and 13% (v/v) hexanol at 70 °C, pH 6.0 for 4 h, and it was validated by the following experimental yield (498.9 ± 4.8 mg g-1 xylan hydrolysate). The maximum production yield remained relatively stable after the immobilized enzyme was reused for 12 cycles with a 471.5 ± 15.1 mg g-1 hexyl xyloside yield (94.4% of the first cycle). Thus, the immobilized β-xylosidase has a high potential as a robust catalyst for application in alkyl xyloside production.





Similar content being viewed by others
References
Leathers TD (1986) Color variants of Aureobasidium pullulans overproduce xylanase with extremely high specific activity. Appl Environ Microbiol 52(5):1026–1030
Tanaka H, Okuno T, Moriyama S, Muguruma M, Ohta K (2004) Acidophilic xylanase from Aureobasidium pullulans: efficient expression and secretion in Pichia pastoris and mutational analysis. J Biosci Bioeng 98(5):338–343. https://doi.org/10.1016/S1389-1723(04)00292-0
Goswami GK, Pathak RR (2013) Microbial xylanases and their biomedical applications: a review. Int J Basic Clin Pharmacol 2(3):237–246
Lotrakul P, Unhapattaratitikul P, Seelanan T, Prasongsuk S, Punnapayak H (2013) An aubasidan-like β-glucan produced by Aureobasidium pullulans in Thailand. ScienceAsia 39(4):363–368. https://doi.org/10.2306/scienceasia1513-1874.2013.39.363
Prasongsuk S, Ployngam S, Wacharasindhu S, Lotrakul P, Punnapayak H (2013) Effects of sugar and amino acid supplementation on Aureobasidium pullulans NRRL 58536 antifungal activity against four Aspergillus species. Appl Microbiol Biotechnol 97(17):7821–7830. https://doi.org/10.1007/s00253-013-5069-5
Prasongsuk S, Lotrakul P, Ali I, Bankeeree W, Punnapayak H (2018) The current status of Aureobasidium pullulans in biotechnology. Folia Microbiol 63(2):129–140. https://doi.org/10.1007/s12223-017-0561-4
Bankeeree W, Akada R, Lotrakul P, Punnapayak H, Prasongsuk S (2018) Enzymatic hydrolysis of black liquor xylan by a novel xylose-tolerant, thermostable β-xylosidase from a tropical strain of Aureobasidium pullulans CBS 135684. Appl Biochem Biotechnol 184(3):919–934. https://doi.org/10.1007/s12010-017-2598-x
Kurakake M, Fujii T, Yata M, Okazaki T, Komaki T (2005) Characteristics of transxylosylation by β-xylosidase from Aspergillus awamori K4. Biochim Biophys Acta, Gen Subj 1726(3):272–279. https://doi.org/10.1016/j.bbagen.2005.08.009
Muzard M, Aubry N, Plantier-Royon R, O’Donohue M, Rémond C (2009) Evaluation of the transglycosylation activities of a GH 39 β-d-xylosidase for the synthesis of xylose-based glycosides. J Mol Catal B-Ezym 58(1):1–5. https://doi.org/10.1016/j.molcatb.2008.10.004
Mamo G, Kasture S, Faryar R, Hashim S, Hatti-Kaul R (2010) Surfactants from xylan: production of n-octyl xylosides using a highly thermostable xylanase from Thermotoga neapolitana. Process Biochem 45(5):700–705. https://doi.org/10.1016/j.procbio.2010.01.005
Papadopoulou E, Hatjiissaak A, Estrine B, Marinkovic S (2011) Novel use of biomass derived alkyl-xylosides in wetting agent for paper impregnation suitable for the wood-based industry. Eur J Wood Wood Prod 69(4):579–585. https://doi.org/10.1007/s00107-010-0513-z
Oldham ED, Seelam S, Lema C, Aguilera RJ, Fiegel J, Rankin SE, Knutson BL, Lehmler H-J (2013) Synthesis, surface properties, and biocompatibility of 1,2,3-triazole-containing alkyl β-d-xylopyranoside surfactants. Carbohydr Res 379:68–77. https://doi.org/10.1016/j.carres.2013.06.020
Xu W, Osei-Prempeh G, Lema C, Davis Oldham E, Aguilera RJ, Parkin S, Rankin SE, Knutson BL, Lehmler H-J (2012) Synthesis, thermal properties, and cytotoxicity evaluation of hydrocarbon and fluorocarbon alkyl β-d-xylopyranoside surfactants. Carbohydr Res 349:12–23. https://doi.org/10.1016/j.carres.2011.11.020
Söderlind E, Wollbratt M, von Corswant C (2003) The usefulness of sugar surfactants as solubilizing agents in parenteral formulations. Int J Pharm 252(1):61–71. https://doi.org/10.1016/S0378-5173(02)00599-9
Jain I, Kumar V, Satyanarayana T (2014) Applicability of recombinant β-xylosidase from the extremely thermophilic bacterium Geobacillus thermodenitrificans in synthesizing alkylxylosides. Bioresour Technol 170:462–469. https://doi.org/10.1016/j.biortech.2014.07.113
Ochs M, Muzard M, Plantier-Royon R, Estrine B, Rémond C (2011) Enzymatic synthesis of alkyl β-d-xylosides and oligoxylosides from xylans and from hydrothermally pretreated wheat bran. Green Chem 13(9):2380–2388. https://doi.org/10.1039/C1GC15719A
Smaali I, Rémond C, Skhiri Y, O’Donohue MJ (2009) Biocatalytic conversion of wheat bran hydrolysate using an immobilized GH43 β-xylosidase. Bioresour Technol 100(1):338–344. https://doi.org/10.1016/j.biortech.2008.06.041
Benassi VM, Silva TM, Pessela BC, Guisan JM, Mateo C, Lima MS, Jorge JA, Polizeli MLTM (2013) Immobilization and biochemical properties of a β-xylosidase activated by glucose/xylose from Aspergillus niger USP-67 with transxylosylation activity. J Mol Catal B Enzym 89:93–101. https://doi.org/10.1016/j.molcatb.2012.12.010
Terrasan CRF, Trobo-Maseda L, Moreno-Pérez S, Carmona EC, Pessela BC, Guisan JM (2016) Co-immobilization and stabilization of xylanase, β-xylosidase and α-l-arabinofuranosidase from Penicillium janczewskii for arabinoxylan hydrolysis. Process Biochem 51(5):614–623. https://doi.org/10.1016/j.procbio.2016.02.014
Ivankovic T, Hrenovic J, Sekovanic L (2010) Influence of the degree of perlite expansion on immobilization of Acinetobacter junii. Biochem Eng J 51(3):117–123. https://doi.org/10.1016/j.bej.2010.06.004
Zhang J, Liu Y, Feng T, Zhou M, Zhao L, Zhou A, Li Z (2017) Immobilizing bacteria in expanded perlite for the crack self-healing in concrete. Constr Build Mater 148:610–617. https://doi.org/10.1016/j.conbuildmat.2017.05.021
Shavisi Y, Sharifnia S, Hosseini SN, Khadivi MA (2014) Application of TiO2/perlite photocatalysis for degradation of ammonia in wastewater. J Ind Eng Chem 20(1):278–283. https://doi.org/10.1016/j.jiec.2013.03.037
Liffourrena AS, Lucchesi GI (2018) Alginate-perlite encapsulated Pseudomonas putida A (ATCC 12633) cells: preparation, characterization and potential use as plant inoculants. J Biotechnol 278:28–33. https://doi.org/10.1016/j.jbiotec.2018.04.019
Rodriguez J, Soria F, Geronazzo H, Destefanis H (2016) Modification and characterization of natural aluminosilicates, expanded perlite, and its application to immobilise α–amylase from A. oryzae. J Mol Catal B Enzym 133:259–270. https://doi.org/10.1016/j.molcatb.2017.01.012
Atlas RM (2010) Handbook of microbiological media. CRC Press, Florida
Torabi SF, Khajeh K, Ghasempur S, Ghaemi N, Siadat SOR (2007) Covalent attachment of cholesterol oxidase and horseradish peroxidase on perlite through silanization: activity, stability and co-immobilization. J Biotechnol 131(2):111–120. https://doi.org/10.1016/j.jbiotec.2007.04.015
Pezzella C, Russo ME, Marzocchella A, Salatino P, Sannia G (2014) Immobilization of a Pleurotus ostreatus laccase mixture on perlite and its application to dye decolourisation. Biomed Res Int 308613(11):1–12. https://doi.org/10.1155/2014/308613
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Bankeeree W, Prasongsuk S, Imai T, Lotrakul P, Punnapayak H (2016) A novel xylan-polyvinyl alcohol hydrogel bead with laccase entrapment for decolorization of reactive black 5. BioResources 11:6984–7000
Jiang Z, Zhu Y, Li L, Yu X, Kusakabe I, Kitaoka M, Hayashi K (2004) Transglycosylation reaction of xylanase B from the hyperthermophilic Thermotoga maritima with the ability of synthesis of tertiary alkyl β-d-xylobiosides and xylosides. J Biotechnol 114(1):125–134
Rosenthal KS, Koussaie F (1983) Critical micelle concentration determination of nonionic detergents with Coomassie brilliant blue G-250. Anal Chem 55(7):1115–1117
Chen G, Li Z, Chen L, Ji S, Shen W (2016) Synthesis and properties of alkyl β-D-galactopyranoside. J Surfactant Deterg 19(6):1095–1105
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol 28(1):25–30
Box GEP, Behnken DW (1960) Some new three level designs for the study of quantitative variables. Technometrics 2(4):455–475
Kumar RR, Cho JY (2014) Reuse of hydroponic waste solution. Environ Sci Pol 21:9569–9577
Briga-Sá A, Nascimento D, Teixeira N, Pinto J, Caldeira F, Varum H, Paiva A (2013) Textile waste as an alternative thermal insulation building material solution. Constr Build Mater 38:155–160
Kotwica Ł, Pichór W, Kapeluszna E, Różycka A (2017) Utilization of waste expanded perlite as new effective supplementary cementitious material. J Clean Prod 140:1344–1352. https://doi.org/10.1016/j.jclepro.2016.10.018
Zhang D, Hegab HE, Lvov Y, Dale Snow L, Palmer J (2016) Immobilization of cellulase on a silica gel substrate modified using a 3-APTES self-assembled monolayer. SpringerPlus 5(1):48
Shinoyama H, Kamiyama Y, Yasui T (1988) Enzymatic synthesis of alkyl β-xylosides from xylobiose by application of the transxylosyl reaction of Aspergillus niger β-xylosidase. Agric Biol Chem 52(9):2197–2202
Rather MY, Mishra S (2013) β-Glycosidases: an alternative enzyme based method for synthesis of alkyl-glycosides. Sustain chem process 1(1):7–22
Nieto-Domínguez M, de Eugenio LI, Barriuso J, Prieto A, de Toro BF, Canales-Mayordomo Á, Martínez MJ (2015) Characterization of a novel pH-stable GH3 β-xylosidase from Talaromyces amestolkiae: an enzyme displaying regioselective transxylosylation. Appl Environ Microbiol 81:6380–6392
Matsumura S, Sakiyama K, Toshima K (1997) One-pot synthesis of alkyl β-D-xylobioside from xylan and alcohol by acetone powder of Aureobasidium pullulans. Biotechnol Lett 19(12):1249–1253
Rosen MJ, Kunjappu JT (2014) Surfactants and interfacial phenomena. Wiley, Hoboken. NJ, USA
Gargouri M, Smaali I, Maugard T, Legoy MD, Marzouki N (2004) Fungus β-glycosidases: immobilization and use in alkyl-β-glycoside synthesis. J Mol Catal B-Enzym 29(1):89–94
Acknowledgments
We are thankful for ACK Hydro Farm Co., Ltd. and Double A (1991) Public Co., Ltd. for providing the materials in this study and Dr. Rico Ramadhan, Universitas Airlangga, for the helpful NMR interpretation.
Funding
This research was performed under funding from the Japan-ASEAN Science, Technology and Innovation Platform (JASTIP) and Grant for Development of New Faculty Staff, Ratchadapiseksomphot Endowment Fund, Chulalongkorn University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 61 kb)
Rights and permissions
About this article
Cite this article
Bankeeree, W., Watanabe, T., Punnapayak, H. et al. Alkyl β-D-xyloside synthesis from black liquor xylan using Aureobasidium pullulans CBS 135684 β-xylosidases immobilized on spent expanded perlite. Biomass Conv. Bioref. 12, 2677–2686 (2022). https://doi.org/10.1007/s13399-020-00755-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-020-00755-5