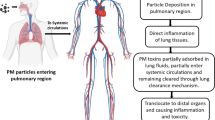
Abstract
The biological effects of airborne particulate matter (PM) in humans stimulated many research activities in recent years. One type of particles contributing to PM are those derived from combustion processes. By using in vitro test systems for assessing the toxic effects of those particles, the transfer into liquid media is necessary. Thus the question arises, how the transfer of airborne particles into liquid suspensions should be realized and how the exposure of test organisms/cells to the particles can be done and monitored with regard to concentration and stability. Further, the characterization of the airborne particles regarding their composition seems to be of importance in order to understand the induced biological effects. Hence, this review gathers and evaluates results from in vitro and ecotoxicity studies, where the introduction of airborne particles into liquids is required. The aim was to learn on the current practice and recommend on the design of toxicity studies in the future. In total, about 80 papers were retrieved by literature search that deal with the assessment of biological effects of combustion-derived particles or their constituents. The papers were evaluated in detail with regard to the methodology applied. From this evaluation and based on expert knowledge regarding nanoparticle testing, recommendations for the assessment of PM applying toxicological assays were developed. To provide guidance, essential criteria are developed that should help in preparation, testing, and during the evaluation phase of toxicological assays. We further advise whole particle testing, complemented by testing of particle extracts and washed particles.

Graphical abstract
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Particles derived from combustion processes, e.g., wood combustion or diesel exhaust, are an important source of fine particulate matter. The characteristics and the composition of combustion-derived particles depend on the sources (i.e., type of material burned) as well as on the conditions during combustion (i.e., temperature, oxygen) [1, 2]. Combustion-derived particles represent a mixture of incomplete combusted particles (diameter > 1000 nm), organic carbon particles (diameter 50–600 nm), soot particles (25–50 nm), and ash (50–125 nm), carrying variable mixtures of organic chemicals as well as heavy metals on their surface [1, 3]. The auxiliary chemicals are formed during the combustion process and deposited on the surface of the particles. Upon emission of fine dust from combustion processes into the ambient air, particles will undergo atmospheric transformation processes leading to several structural and chemical changes [4]. From the ambient air, particles are inhaled by humans with the particle size determining their fate in the lung. While the PM10 (< 10 μm) fraction reaches the upper airways such as mouth, nose, and throat, the smaller PM2.5 (< 2.5 μm) fraction is reaching the airways of the lung. The smallest particle fraction PM1 (< 1 μm) is able to enter the alveoli and is potentially able to penetrate into the blood stream [5, 6]. The deposition of particles into the lung is associated with inflammation responses and, as evidenced by numerous studies, may lead to more severe diseases such as asthma and cancer. Further, exposure to combustion-derived particles is also related to human cardiovascular diseases (reviewed by Gutierrez, 2014 [7]). It is of great interest to unravel the development of such diseases and gain a mechanistic understanding on the cause of the toxicity of combustion-derived particles. Humans are exposed to mixtures of various PM types, and from epidemiological studies, it is also difficult to gain information on the most critical type of PM. Inhalation studies involving mammals are expensive and critical from the animal welfare perspective. Hence, in vitro assays may provide a cost-effective, ethically favorable, yet informative tool to expand our knowledge on effects of PM (mechanistically as well as with regard to effective compounds). One approach is air-liquid interface (ALI) systems, which typically allow the exposure of lung cells via the gaseous phase to mimic the exposure via inhalation. Further, not only humans are exposed to combustion particles released to the ambient air, but environmental organisms as well. When assessing the hazard of combustion particles for aquatic organisms, analogous to in vitro testing, the transfer into liquids is required.
However, to date, it is difficult to correlate biological effects to specific properties or compounds of combustion-derived particles. It has to be emphasized that the testing of combustion-derived particles in in vitro assays results in a highly complex exposure situation. It is the mixture effects that need consideration but also the different distribution behavior of chemicals, metals, and particles between water and organisms [8]. Thus, for gaining more information about the particle’s toxicity, a consistent procedure regarding the preparation of filter-sampled, combustion-derived particles for biological testing in liquid media should be used. This does not yet exist. Often, only extracts of filter-sampled material are prepared and either the organic or the aqueous phase was subjected to testing for biological effects (e.g., [9–11]). By this procedure, the particles as well as other compounds are omitted from testing leading to a potential underestimation of the biological effects of combustion-derived particles [7, 12].
As it has been demonstrated in numerous studies, nanoparticles change their physical-chemical properties when brought into suspension, and the composition of the aqueous medium used and the energy input during suspension preparation will influence particle behavior (e.g., agglomeration) during the test [13–15], as well as toxicity [16]. Hence, particle characteristics and behavior need to be considered for test design and data evaluation. For combustion-derived particles, it has likewise been observed that sample collection, extraction from the filters, and resuspension in media may alter their toxicological properties [17]. The importance of considering combustion-derived particles in their entity for the evaluation of toxicity, in order to be able to also elucidate mixture effects, is emphasized in the literature [12, 18, 19], however difficult to test.
Current risk assessment of airborne particles is performed exposure-based (particle amount/volume) (1. BImSchV, Bundesimmissionsschutzverordnung). As discussed by Gutierrez [7], for setting regulatory threshold values, the particle number appears not as an adequate measure, because the relation of the particle number to particle toxicity may not be a direct one, and the composition of combustion-derived particles, their characteristics, and the content of auxiliary chemicals may be more relevant [7, 20]. The chemical analysis of selected PAHs is considered insufficient for prediction of adverse effects, as well [18, 21]. Hence, a composition-based or effect-based approach may be more reliable. But therefore, it is first of all necessary to develop recommendations and guidance for a meaningful practice for future toxicity testing of airborne particles.
Thus, we aimed to review the current approaches used to assess the ecotoxicological potential of combustion-derived particles with focus on the procedures used in bioassays and the methodology employed for particle characterization. Since only few studies analyzed the hazard of combustion-derived particles towards environmental organisms [7, 22–24], in vitro studies regarding human toxicity were considered also, thereby excluding those using ALI systems. Related to that, a specific objective of the review is to consider the transfer of particles from the air to the water phase and the implications this step may have on the whole testing procedure and ultimately on biological effects. By taking into consideration the experiences from aquatic toxicity testing of engineered nanomaterials, we aim to summarize the current practice and develop recommendations on the design of reliable toxicity and ecotoxicity studies in the future. The conclusions on physical-chemical particle behavior and its assessment and consideration during all steps of testing apply likewise to in vitro and aquatic ecotoxicity testing.
2 Literature survey on in vitro toxicity of combustion derived particles
In order to collect relevant in vitro studies assessing biological effects of combustion-derived particles, a literature research was performed in ISI Web of Science and Scopus considering ecotoxicity, as well as in vitro toxicity of combustion particles. The last search was performed in August 2017.
The literature research concerning the in vitro toxicity resulted in retrieval of 329 and 49 publications, respectively, and about a quarter of them were considered relevant to in vitro toxicity testing of combustion-derived particles. For instance, studies dealing with toxic effects of cigarette smoke were excluded, since often not the particle fraction was analyzed, but rather single-compound toxicity of the combustion products was considered and/or the exposure was conducted via the gaseous phase. As few as 10 out of 69 publications in Web of Science and 3 out of 17 articles in Scopus were assessing the ecotoxicity of combustion-derived particles.
Additionally, a dissertation dealing with the topic of “Toxicity Screening of Wood Combustion Fine Dust Using a Microbial Test Battery” was considered [7].
In summary, 81 publications dealing with in vitro (eco-)toxicity of combustion-derived particles were retrieved, forming the basis for this review. The different studies were analyzed in detail regarding the types of particle, the aims, the methodology, and the conclusions.
2.1 Overview on studies
In most publications, urban particulate matter was tested (57 publications), 20 papers analyzed the effects of diesel exhaust particles, and 12 publications dealt with in vitro toxicity testing of particles generated during wood combustion, while 9 publications could not be sorted in such a category and were summarized as “others.” Out of the 81 publications, 13 studies analyzed the toxic effects of more than one particle type.
Next, the objectives for analyzing the toxic effects of combustion-derived particles were summarized into five categories.
-
Objective category 1: Understanding the mechanism of health effects of combustion-derived particles is the most frequent objective (84% of studies).
-
Objective category 2: 46% of the authors wanted to compare the effects of different PM from different sources.
-
Objective category 3: 40% of studies intended to identify the effect-relevant compounds attached to the particles.
-
Objective category 4: 16% of the authors aimed to characterize the whole particles in order to identify effect-relevant parameters.
-
Objective category 5: 2 publications wanted to compare the applicability of in vivo versus in vitro methods for effect-detection of combustion-derived particles.
Several studies pursued more than one objective. Most of the publications focusing on human toxic effects employed primary cells or permanent mammalian cell lines (human cancer cell lines or mouse cell lines). In line with in vivo studies for assessing toxicity of combustion-derived particles, bioassays were used which address the following modes of action: cytotoxicity, inflammation, oxidative stress, and genotoxicity. These modes of action were investigated in 60, 25, 19, and 38 studies, respectively. The ecotoxicity of particles was analyzed in 12 publications. For analyzing ecotoxic effects of combustion-derived particles, most often the microtox assay with Vibrio fischeri was used. Barbosa et al. analyzed the effects of biomass ashes by using an ecotoxicity test battery, where they additionally analyzed the effects on Daphnia magna, Artemia franciscana, and two different algae (Selenastrum capricornutum, Phaeodactylum tricornutum) [25].
A detailed overview on the employed bioassays sorted according to the main categories “ecotoxicity,” “cytotoxicity,” “cell viability,” “mutagenicity/genotoxicity,” and “oxidative stress” is given in the Supplement. For analyzing inflammatory effects, the cytokine release was either analyzed via ELISA or via gene expression analysis of related genes.
In a next step, the studies were analyzed in detail regarding the methodology of particle generation/ sampling, subsequent sample preparation, and analysis (e.g., of chemicals attached to the particles, particle morphology, particle size). The issues that were considered in detail and the corresponding publications are summarized as a flow chart in Fig. 1.
In the upper part of the flow chart, the “Chemical and particle characterization” is displayed. About 70% of the publications characterize the chemical composition of the particle extracts [3, 7, 10, 12, 17–19, 22–70]. Out of these 55 publications in 30 articles, also particle characteristics were considered, with the “average PM concentration” mentioned most often [10, 19, 27, 29, 31, 34–36, 39, 40, 43, 46, 48, 50–53, 57–59, 63, 66, 67], the shape of the particles was analyzed in 12 publications [3, 12, 18, 28, 34, 36, 38, 43, 44, 53, 57, 71], and the surface chemistry was considered only in 4 publications [7, 18, 19, 70]. Accordingly, 24 publications did not characterize chemicals attached to the particles [9, 11, 72–93], and only 7 out of these 24 publications considered other particle characteristics [72, 76, 78, 80, 82, 87, 89], with “average PM concentration” being mentioned most often. One publication reported on the particle’s shape [80]. Overall, the particle characteristics were considered in less than 50% of the publications.
The lower part of the flow chart lists the relevant procedures and parameters regarding “experimental setup for toxicity study.” Out of the 81 publications, 47 used the suspended particles for toxicity testing [3, 7, 12, 17–19, 23, 24, 26, 28–30, 33–42, 44, 46–48, 50, 53, 57–62, 65, 70–73, 75, 78, 80, 81, 83, 85, 87, 93], while the others used extracts of the particles [9–11, 22, 25, 27, 31, 32, 43, 45, 49, 51, 52, 54–56, 63, 64, 66–69, 74, 76, 77, 79, 82, 84, 86, 88–92], i.e., organic or aqueous extracts only containing the compounds, which were attached to and/or leached from the particles. Hence, the subset of 47 studies not omitting the particles from toxicity testing was subjected to a more detailed analysis of the characterization methods employed. Out of these 47 articles in six studies, both suspended particles and extracts were tested [12, 18, 26, 29, 80, 85]; two studies also tested the particle toxicity, besides using the suspended particles, via the direct contact method [7, 57], and two studies analyzed the toxicity by using suspended particles, extracts, and additionally the washed particles [30, 70]. For suspending the particles, most often the air sampling filter was cut into small pieces and the particles were removed via ultrasonication. In detail, the preparation of the particle suspension differs in terms of sonication time, additional steps (e.g., vortexing), temperature (e.g., on ice, 40 °C), and several other aspects.
With respect to the studies assessing ecotoxicological effects of combustion particles, either extracts in water or DMSO were used for testing [25, 66–69, 91, 92] or the filters were directly used (either grounded or as inserts in well plates) for exposing the bacteria [7, 23, 24, 90, 93].
Out of the 47 publications using suspended particles for toxicity testing, only 9 publications considered the particle behavior under test conditions. Danielsen and colleagues, as well as Carero and co-workers, analyzed the size distribution in the test medium [3, 30]. Additionally, Gauggel et al. analyzed the size distribution not only in test medium, but also in water [18]. The agglomeration of the particles under test conditions was mentioned in three publications [18, 42, 71], the sedimentation of the particles under test conditions was described by Dilger et al. [71], and the interaction of the particles with the test organisms or cells is described in at least three publications [36, 44, 80]. Furthermore, Kocbach and Carero discussed the relevance of the in vitro tested particle concentrations for the human exposure scenario [30, 70]. The release of attached components (e.g., chemicals, metals) was not considered at all.
2.2 Studies’ conclusion on the potential of combustion-derived particles to induce toxic effects
About half of the studies correlate the observed biological effects with the physicochemical properties of the particles. Most often, they concluded that the effects depend on the particle composition (PAH and/or metal content) and also the particle size (e.g., [10, 40, 50, 52, 72]). In detail, about a quarter of the authors declared that the PAH content/the organic fraction is responsible for the observed toxic effects (e.g., [9, 11, 37, 88]), and fewer authors correlate the toxic effects with the inorganic water-soluble fraction (e.g., [54, 62]), while some authors concluded that both parts attribute to overall toxicity (e.g., [23, 71]). In contrast, four authors could not correlate the PAH content to the observed effects [25, 42, 57, 86]. Regarding the toxicity of the whole particles, Diociaiuti and colleagues concluded that the observed oxidative stress is mainly due to the interaction of the particle surface with the cell membrane [36].
About 20% of the studies dealt with the modulation of toxic effects due to different PM samples, e.g., collected from different sites or generated under different combustion conditions. For example, Turoczi and co-workers analyzed the ecotoxicity of different PMs and concluded that “wood smoke was found to be more ecotoxic than particulates emitted from modern diesel engines.” [24].
In five publications, conclusions on the asset of using bioassays for toxicity testing of combustion-derived particles were made. In general, it is recognized that neither the amount of PM emission nor chemical analysis alone provides sufficient information to estimate the overall toxicity of combustion-derived particles [7, 18, 29, 51]. Accordingly, the use of effect-based methods (e.g., in vitro tests) is recommended in order to allow a more complete view on the toxic effects combustion particle may exert [7, 22, 29, 51, 92].
Especially in terms of correlating the analyzed toxicity to the particle’s physicochemical properties, no clear conclusions can be drawn so far. In order to facilitate our understanding on the relation between properties and behavior of combustion particles with their biological effects towards environmental organisms, a suitable and adaptable strategy for particle characterization in relation to toxicological testing is needed. Therefore, the ambition of the review was to provide guidance for the design of future ecotoxicity studies. Specific attention is paid to the transfer of particles from the air to the water phase and the implications this step may have on biological effects and the testing procedure in general.
3 Developing recommendations—the way towards suitable and adaptable criteria
While analyzing the 81 selected publications, it became obvious that there is no common understanding on a suitable methodology regarding toxicity testing of combustion-derived particles. Half of the studies only tested the toxicity of extracts, the other part tested suspended particles, and in six studies the toxicity of suspended particles was compared to extracts’ toxicity [12, 18, 26, 29, 80, 85].
But in order to understand the mechanism of particle toxicity or to compare toxic effects, e.g., of different combustion conditions, a suitable yet adaptable practice is needed. For developing such an approach, it is necessary to compile which procedures and parameters should be characterized.
3.1 Need for physical-chemical characterization in relation to the studies’ aims
For developing an applicable approach for toxicity testing of combustion-derived particles, first of all important issues that should be included in characterization of the particles and toxicity testing were summarized. In a first step, arguments were collected regarding the need for characterization in relation to the five objective categories (Table 1).
Most of the studies aimed to understand the mechanisms responsible for particle-induced health effects (objective category 1). To tackle this question, it is necessary to use an adequate bioassay system able to analyze the effects of the test compounds as well as of particles. Additionally, the validation of the test results by appropriate positive and negative controls is important. For understanding the mechanism of the particle-induced toxicity, it seems necessary to consider the exposure type, i.e., only testing the extracts did not represent the possible toxic effects of the whole particle, as it occurred in the environment.
The second objective category relates to the comparison of toxic effects with different PM sources. For this purpose, a thorough description of the different sources (e.g., sampling site, combustion conditions) is required as well as a detailed analysis of the different particles, including physicochemical characterization of the particles (e.g., particle concentration, component analysis, size, shape, surface area). When using reliable standardized bioassays, it may be appropriate to reduce the effort in particle characterization and focus on the effect side.
Forty percent of the studies aimed at identification of the effect-relevant components attached to the particles (objective category 3). Therefore, it seems reasonable only analyzing and testing particles’ extracts. But it should be kept in mind that the bioavailability of attached compounds may be different from that of extract, e.g., whole particles may act in a Trojan horse-like manner, and compounds attached to the particles may enter organisms and cells faster in higher concentrations (e.g., [94, 95]). Thus, the particle characteristics (e.g., size, shape, surface) and the particle behavior under test conditions should be considered as well when determining the effect-relevant components.
In the fourth objective category, the studies aimed at assessing the toxicity of the whole PM. For this purpose, it seems necessary to characterize the particles in detail regarding their physicochemical properties (e.g., components, size, shape). Additionally, the behavior of the particles under test conditions in the medium (e.g., agglomeration) as well as over the test duration (e.g., release of attached compounds, particle sedimentation) should be analyzed. In principle, attention needs to be paid to the modifications particles undergo upon transfer from the gas to the liquid phase (e.g., resulting in detachment of chemicals from the particles).
Some of the authors wanted to compare the results of in vivo studies with those of in vitro studies, i.e., wanted to show the applicability of in vitro methods for particle-induced toxicity (objective category 5). The different exposure routes should be kept in mind, since in in vivo studies, the test organisms were exposed via the gaseous phase, while for in vitro tests it is more common to use exposure via the liquid phase. The more detailed the differences of airborne particles are compared to suspended particles, the more reliable the comparison between in vivo and in vitro results will be. Appropriate dosimetry is important to allow a comparison of air-based measures (i.e., particle number/volume) to liquid-based measured (i.e., mass/volume). With regard to this aspect, it seems necessary to describe the preparation of test suspensions in detail. Additionally, the particle behavior under test conditions and over test duration should be characterized, since the physicochemical parameters of the particles may change.
As demonstrated by the different motivations to study effects of PM, it is obvious that for objective categories 1–5 the focus of the physical-chemical characterization may be different (see Table 1). Based on these considerations in a next step, a criterion catalog listing all the essential parameters and processes is developed.
3.2 Development of a list of relevant characterization criteria
The criterion catalog was developed based on the issues considered for the testing methodology in the flow chart (Fig. 1) as well as on requirements for in vitro testing that were formulated in 4.1 in relation to the five objective categories. In addition, existing knowledge on toxicity testing of nanoparticles was used, specifically by considering the characterization criteria developed within the DaNa project (http://www.nanopartikel.info/en/nanoinfo/methods/991-literature-criteria-checklist, [96, 97]). All relevant parameters and requirements were listed according to the five subsections “general methodology,” “chemical characterization,” “particle characterization,” “exposure conditions,” and “consideration of particle behavior under test conditions.” The final list is summarized in Table 2 and presents a compilation of issues that may be considered by experimenters during planning and conducting an in vitro toxicity study involving combustion particles. The list aims to provide guidance in the sense that it gives an overview of criteria, and experimenters may select the relevant ones for the purpose of their study.
3.3 Studies considering particle characteristics and particle behavior under test conditions
By generating the flow chart presented in Fig. 1, a number of studies were identified that (1) characterized the particles, (2) applied whole particles to toxicological testing, and (3) provided information on the particle behavior during the testing (most noticeable studies are those that passed all decision points with “YES” and hence fulfilled the essential criteria with regard to “chemical and physical characterization” (upper part) and “Experimental setup for toxicity tests” (lower part)). This set of seven studies [3, 18, 36, 44, 70, 71, 80] was subjected to an in-depth evaluation of the particle characterization. Regarding the objectives of these most noticeable studies interestingly, most of them dealt primarily with the objective category 2 by comparing different types of particles (either generated by different combustion conditions, from different locations or different sources), which requires knowledge on particle characteristics. In addition, also objectives 1, 3, and 4 were addressed. However, none of these seven publications considered more than one or two of the criteria related to the particle behavior under test conditions, i.e., none of the papers reported on all three parameters on particle characteristics (upper part) or all five parameters on particle behavior (lower part). The issues that were analyzed by these authors are summarized in Table 3.
Out of these seven publications, the work of Gauggel and co-workers (2012) has to be highlighted [18]. The authors stressed out the importance of particle characterization within the cell culture medium: “Upon suspension in liquids the physico-chemical characteristics of PM may change, due to aggregation, wetting or dissolution, with ensuing consequences for the bioavailability of particulates and/or adsorbed toxins and thus for the type and intensity of the effects observed. Hence, toxicological studies of PM must always be accompanied by a characterization of particles in liquids.” [18]. Therefore, they used a systematic approach, consisting of a combination of methods to characterize different particle types in liquids, i.e., in cell culture medium and in ultrapure water. With this methodology, the authors could demonstrate that PAH bound to the particles has a stronger potency than the extracted PAH alone. Hence, the authors concluded that the bioavailability of toxins strongly depends on the physico-chemical composition of the PM.
In addition to the particle characterization, the methodology of Kocbach and co-workers as well as Carero et al. seems convincing, since in these studies in addition to suspended particles, also the extracts and the washed particles were used for toxicity tests [30, 70]. The idea was “to verify whether the effects were the result either of the particles or of organic compounds that could be extracted from the particles (…)” [30]. In line with this, Kocbach and colleagues observed that the sum effect “induced by the organic extract and the washed particles was considerably lower than for the corresponding native particles.” Thus, they concluded that the effect (inflammatory potential) of the organic fraction depends on the adsorption to the particle [70]. In the study of Carero et al., two cell lines were used for toxicity testing. When A549 cells were exposed to extracts or the washed particles, no effects were detected, thus the authors concluded that “both structure and composition probably play a role in the genotoxicity of these particles in A549 cells.” In contrast, when THP-1 cells were used for toxicity testing, washed particles of carbon black and extracts of diesel exhaust particles induced significant DNA damage, similar to the effects observed by using the original particles [30]. Furthermore, both authors discussed the relevance of the in vitro tested particle concentrations for human exposure and concluded that the lower test concentrations (100 μg/mL in Kocbach et al. [70]) could be representative for human exposure via the air.
In addition, two more publications were examined in two more publications which were identified during literature research were examined in detail, which were identified during the literature research, but were excluded from the first analysis since no combustion-derived particles were analyzed [98, 99]. But due to their noteworthy methodology, i.e., related to particle characterization, these publications were now discussed in detail. Van Landuyt and colleagues analyzed the “Interaction of rat alveolar macrophages with dental composite dust” [99]. For this purpose, they analyzed the size distribution of the original unfiltered particle preparation in water and in cell culture medium and could show that the particles agglomerate under cell culture conditions. Furthermore, the particle behavior was analyzed via phase contrast microscopy and it was demonstrated that filtered particles underwent rapid sedimentation during the incubation time. In this publication, macrophages were used as the test system. It is a special model, as the cells were mobile, not attaching to the bottom of test vessels and actively taking up the particles. Using light microscopy, the authors could show, that “some cells were completely loaded with engulfed composite dust, while others only contained 2–3 phagosomes” and further calculated the mean cellular load, which ranged under experimental conditions from 15 to 120 pg per cell. In a next step, the authors compared this dosage to those typically achieved in inhalation studies on rats and concluded that this in vitro study reflects an in vivo experimental scenario (details in van Landuyt et al. [97]). Additionally, the real exposure situation was reflected and the authors concluded, that “(…) the dose range used in this study should be regarded as relevant for the work place exposure scenario (…)”. To sum up, in this study all of the items regarding the particle behavior under test conditions were considered allowing to draw more reliable conclusions on the biological effects of different types of respirable dust as well as on the transferability of in vitro results to the in vivo situation.
In the publication of Gminski et al., black toner powders were analyzed concerning their genotoxicity [98]. The authors analyzed the particles via scanning electron microscopy in order to characterize the particles shape. Additionally, they described the particle surface as “slightly rough and (..) covered by rounded submicrometer-sized particles with a diameter of 30–200 nm,” which they identified as magnetite particles using X-ray diffractometry. After suspending the particles, the authors observed that the “particles still exhibit the same size and remain covered by magnetite particles.” In contrast, after sonication some of the magnetite particles were separated and formed agglomerates. The genotoxicity of the toner particles was examined in A549 human cell line using the Comet Assay. For two black toner powders, significant genotoxicity was found. Gminski and co-workers concluded that the effects “(…) cannot be linked to one particular substance or mechanism, but to a complex interaction of particles, their metal content and their PAH contaminants.”
4 Recommendation for future toxicity studies
Out of 81 publications which formed the basis of this review, no consistent methodology regarding toxicity testing of combustion-derived particles could be derived. Half of the studies tested the toxicity of extracts; the other part studied suspended particles. In as few as nine studies, the particle behavior under test conditions was examined. In the interest of understanding the mechanism of particle toxicity, to compare toxic effects, e.g., of different combustion conditions, or to allow better transferability between in vitro results and exposure to airborne particles, a suitable yet adaptable practice is preferable. In order to allow the design of toxicity studies that cover these particle relevant issues, a list of criteria is suggested. These criteria were developed according to the following:
-
1.
Need to describe exposure conditions and respective particle behavior and composition over exposure time to allow identification of hazard of combustion derived particles
-
2.
Need to assess potential alterations in particle behavior and composition upon transfer in liquids and to take these alterations into account for final hazard assessment
Furthermore, we conclude from the evaluation of the current literature that whole particle testing is preferable, because it will cover mixture effects, kinetic effects, and “Trojan horse”-like effects. Mixture effects may arise from the combination of various chemicals, metals, and the particles when applied together in a toxicity test. Additionally, the toxicity may depend on Trojan horse-like effects, which means that the uptake of combustion particles by the test organisms/cells may increase the amount of toxic organic or metal compounds internalized by organisms/cells. In relation to this effect, the uptake of particles may facilitate the emergence of effects of the attached auxiliary compounds (kinetic effect). The testing of whole particles will ideally be complemented by testing the extracts and washed particles (remaining particles after the extraction procedure). This will allow elucidating potential mixture effects.
So far, whole particle testing in ecotoxicity assays was only performed in bacterial assays and exposure was performed in the presence of the filter material used for collection of PM. This approach may not be applicable for other ecotoxicity tests involving, e.g., algae and daphnids; further, we suspect that the material (even though filter material without particles was used as control) will affect the assay by, e.g., sorption of media components. In the future, the development of standardized toxicological test systems may allow to reduce the effort in particle characterization for at least some of the criteria mentioned in Table 2.
Concerning the risk assessment of airborne particles, a database might be useful, where the particle characteristics, attached compounds, exposure conditions, and the analyzed effects are summarized. Further, the proposed list of essential criteria for toxicity testing will hopefully foster first steps towards the standardization of testing methodology.
References
Kocbach Bølling A, Pagels J, Yttri KE et al (2009) Health effects of residential wood smoke particles: the importance of combustion conditions and physicochemical particle properties. Part Fibre Toxicol 6:29. https://doi.org/10.1186/1743-8977-6-29
Naeher LP, Brauer M, Lipsett M et al (2007) Woodsmoke health effects: a review. Inhal Toxicol 19:67–106. https://doi.org/10.1080/08958370600985875
Danielsen PH, Moller P, Jensen KA et al (2011) Oxidative stress, DNA damage, and inflammation induced by ambient air and wood smoke particulate matter in human A549 and THP-1 cell lines. Chem Res Toxicol 24:168–184. https://doi.org/10.1021/tx100407m
Lundstedt S, White PA, Lemieux CL et al (2007) Sources, fate, and toxic hazards of oxygenated polycyclic aromatic hydrocarbons (PAHs) at PAH-contaminated sites. Source AMBIO A J Hum Environ 36:475–485. https://doi.org/10.1579/0044-7447(2007)36[475:SFATHO]2.0.CO;2
Kreyling WG, Semmler-Behnke M, Takenaka S, Möller W (2013) Differences in the biokinetics of inhaled nano- versus micron- sized particles. Acc Chem Res 46:714–722. https://doi.org/10.1021/ar300043r
Oberdörster G, Oberdörster E, Oberdörster J (2005) Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environ Health Perspect 113:823–839. https://doi.org/10.1289/ehp.7339
Gutiérrez IR (2014) Toxicity screening of wood combustion fine dust using a microbial test battery. PhD thesis, University of Konstanz
Praetorius A, Tufenkji N, Goss K-U et al (2014) The road to nowhere: equilibrium partition coefficients for nanoparticles. Environ Sci Nano 1:317. https://doi.org/10.1039/C4EN00043A
Binkova B, Vesely D, Vesela D et al (1999) Genotoxicity and embryotoxicity of urban air particulate matter collected during winter and summer period in two different districts of the Czech Republic. Mutat Res Toxicol Environ Mutagen 440:45–58. https://doi.org/10.1016/s1383-5718(99)00011-x
Bonetta S, Gianotti V, Bonetta S et al (2009) DNA damage in A549 cells exposed to different extracts of PM2.5 from industrial, urban and highway sites. Chemosphere 77:1030–1034. https://doi.org/10.1016/j.chemosphere.2009.07.076
Hamers T, van Schaardenburg MD, Felzel EC et al (2000) The application of reporter gene assays for the determination of the toxic potency of diffuse air pollution. Sci Total Environ 262:159–174. https://doi.org/10.1016/s0048-9697(00)00600-8
Landkocz Y, Ledoux F, André V et al (2017) Fine and ultrafine atmospheric particulate matter at a multi-influenced urban site: physicochemical characterization, mutagenicity and cytotoxicity. Environ Pollut 221:130–140. https://doi.org/10.1016/j.envpol.2016.11.054
Meißner T, Kühnel D, Busch W et al (2010) Physical-chemical characterization of tungsten carbide nanoparticles as a basis for toxicological investigations. Nanotoxicology 4:196–206. https://doi.org/10.3109/17435391003605455
Tantra R, Sikora A, Hartmann NB et al (2015) Comparison of the effects of different protocols on the particle size distribution of TiO2 dispersions. Particuology 19:35–44. https://doi.org/10.1016/j.partic.2014.03.017
Meißner T, Oelschlägel K, Potthoff A (2014) Dispersion of nanomaterials used in toxicological studies: a comparison of sonication approaches demonstrated on TiO2 P25. J Nanopart Res 16:2228. https://doi.org/10.1007/s11051-013-2228-7
Potthoff A, Weil M, Meißner T, Kühnel D (2015) Towards sensible toxicity testing for nanomaterials: proposal for the specification of test design. Sci Technol Adv Mater 16:65006. https://doi.org/10.1088/1468-6996/16/6/065006
Jalava PI, Happo MS, Kelz J et al (2012) In vitro toxicological characterization of particulate emissions from residential biomass heating systems based on old and new technologies. Atmos Environ 50:24–35. https://doi.org/10.1016/j.atmosenv.2012.01.009
Gauggel S, Derreza-Greeven C, Wimmer J et al (2012) Characterization of biologically available wood combustion particles in cell culture medium. ALTEX Altern Tierexperimenten 29:183–200
Lepers C, Dergham M, Armand L et al (2014) Mutagenicity and clastogenicity of native airborne particulate matter samples collected under industrial, urban or rural influence. Toxicol Vitr 28:866–874. https://doi.org/10.1016/j.tiv.2014.03.011
Valberg PA (2004) Is PM more toxic than the sum of its parts? Risk-assessment toxicity factors vs. PM-mortality “effect functions”. Inhal Toxicol 16:19–29. https://doi.org/10.1080/08958370490442935
Mumtaz, M., George J (1995) Toxicological profile for polycyclic aromatic hydrocarbons. Agency for Toxic Substances and Disease Registry. http://www.atsdr.cdc.gov/toxprofiles/tp69.pdf
Roig N, Sierra J, Rovira J et al (2013) In vitro tests to assess toxic effects of airborne PM10 samples. Correlation with metals and chlorinated dioxins and furans. Sci Total Environ 443:791–797. https://doi.org/10.1016/j.scitotenv.2012.11.022
Ács A, Ferincz Á, Kovács A et al (2013) Ecotoxicological characterisation of exhaust particulates from diesel-powered light-duty vehicles. Cent Eur J Chem 11:1954–1958. https://doi.org/10.2478/s11532-013-0326-0
Turóczi B, Hoffer A, Tóth Á et al (2012) Comparative assessment of ecotoxicity of urban aerosol. Atmos Chem Phys 12:7365–7370. https://doi.org/10.5194/acp-12-7365-2012
Barbosa R, Dias D, Lapa N et al (2012) Chemical and ecotoxicological properties of size fractionated biomass ashes. Fuel Process Technol 109:124–132. https://doi.org/10.1016/j.fuproc.2012.09.048
Akhtar US, McWhinney RD, Rastogi N et al (2010) Cytotoxic and proinflammatory effects of ambient and source-related particulate matter (PM) in relation to the production of reactive oxygen species (ROS) and cytokine adsorption by particles. Inhal Toxicol 22:37–47. https://doi.org/10.3109/08958378.2010.518377
Argyropoulos G, Besis A, Voutsa D et al (2016) Source apportionment of the redox activity of urban quasi-ultrafine particles (PM0.49) in Thessaloniki following the increased biomass burning due to the economic crisis in Greece. Sci Total Environ 568:124–136. https://doi.org/10.1016/j.scitotenv.2016.05.217
Baulig A, Poirault JJ, Ausset P et al (2004) Physicochemical characteristics and biological activities of seasonal atmospheric particulate matter sampling in two locations of Paris. Environ Sci Technol 38:5985–5992. https://doi.org/10.1021/es049476z
Brits E, Schoeters G, Verschaeve L (2004) Genotoxicity of PM10 and extracted organics collected in an industrial, urban and rural area in Flanders, Belgium. Environ Res 96:109–118. https://doi.org/10.1016/j.envres.2004.03.006
Carero ADP, Hoet PHM, Verschaeve L et al (2001) Genotoxic effects of carbon black particles, diesel exhaust particles, and urban air particulates and their extracts on a human alveolar epithelial cell line (A549) and a human monocytic cell line (THP-1). Environ Mol Mutagen 37:155–163
Chen SJ, Cheng SY, Shue MF et al (2006) The cytotoxicities induced by PM10 and particle-bound water-soluble species. Sci Total Environ 354:20–27. https://doi.org/10.1016/j.scitotenv.2004.11.012
Chiarolini A, Donate MT, GomezLechon MJ et al (1997) Comparison of rat hepatocyte and differentiated hepatoma cell line cultures as bio-indicators of CYP 1A1 inducers in urban air. Biomarkers 2:279–285
Corsini E, Budello S, Marabini L et al (2013) Comparison of wood smoke PM2.5 obtained from the combustion of FIR and beech pellets on inflammation and DNA damage in A549 and THP-1 human cell lines. Arch Toxicol 87:2187–2199. https://doi.org/10.1007/s00204-013-1071-z
De Vizcaya-Ruiz A, Gutierrez-Castillo ME, Uribe-Ramirez M et al (2006) Characterization and in vitro biological effects of concentrated particulate matter from Mexico City. Atmos Environ 40:S583–S592. https://doi.org/10.1016/j.atmosenv.2005.12.073
Ding X, Wang ML, Chu HY et al (2014) Global gene expression profiling of human bronchial epithelial cells exposed to airborne fine particulate matter collected from Wuhan, China. Toxicol Lett 228:25–33. https://doi.org/10.1016/j.toxlet.2014.04.010
Diociaiuti M, Balduzzi M, De Berardis B et al (2001) The two PM2.5 (fine) and PM2.5-10 (coarse) fractions: evidence of different biological activity. Environ Res 86:254–262. https://doi.org/10.1006/enrs.2001.4275
Dumax-Vorzet AF, Tate M, Walmsley R et al (2015) Cytotoxicity and genotoxicity of urban particulate matter in mammalian cells. Mutagenesis 30:621–633. https://doi.org/10.1093/mutage/gev025
Durga M, Nathiya S, Rajasekar A, Devasena T (2014) Effects of ultrafine petrol exhaust particles on cytotoxicity, oxidative stress generation, DNA damage and inflammation in human A549 lung cells and murine RAW 264.7 macrophages. Environ Toxicol Pharmacol 38:518–530. https://doi.org/10.1016/j.etap.2014.08.003
Fox JR, Cox DP, Drury BE et al (2015) Chemical characterization and in vitro toxicity of diesel exhaust particulate matter generated under varying conditions. Air Qual Atmos Heal 8:507–519. https://doi.org/10.1007/s11869-014-0301-8
Ghauri B, Mansha M, Khalil C (2012) Characterization of cytotoxicity of airborne particulates from urban areas of Lahore. J Environ Sci 24:2028–2034. https://doi.org/10.1016/S1001-0742(11)61040-1
Jalava PI, Hirvonen M-R, Sillanpaa M et al (2009) Associations of urban air particulate composition with inflammatory and cytotoxic responses in RAW 246.7 cell line. Inhal Toxicol 21:994–1006. https://doi.org/10.1080/08958370802695710
Jalava PI, Salonen RO, Nuutinen K et al (2010) Effect of combustion condition on cytotoxic and inflammatory activity of residential wood combustion particles. Atmos Environ 44:1691–1698. https://doi.org/10.1016/j.atmosenv.2009.12.034
Jayasekher T (2009) Aerosols near by a coal fired thermal power plant: chemical composition and toxic evaluation. Chemosphere 75:1525–1530. https://doi.org/10.1016/j.chemosphere.2009.02.001
Karlsson HL, Nilsson L, Moller L (2005) Subway particles are more genotoxic than street particles and induce oxidative stress in cultured human lung cells. Chem Res Toxicol 18:19–23. https://doi.org/10.1021/tx049723c
Landreman AP, Shafer MM, Hemming JC et al (2008) A macrophage-based method for the assessment of the reactive oxygen species (ROS) activity of atmospheric particulate matter (PM) and application to routine (daily-24 h) aerosol monitoring studies. Aerosol Sci Technol 42:946–957. https://doi.org/10.1080/02786820802363819
Manzano-Leon N, Serrano-Lomelin J, Sanchez BN et al (2016) TNF alpha and IL-6 responses to particulate matter in vitro: variation according to PM size, season, and polycyclic aromatic hydrocarbon and soil content. Environ Health Perspect 124:406–412. https://doi.org/10.1289/ehp.1409287
Matzenbacher CA, Garcia ALH, dos Santos MS et al (2017) DNA damage induced by coal dust, fly and bottom ash from coal combustion evaluated using the micronucleus test and comet assay in vitro. J Hazard Mater 324:781–788. https://doi.org/10.1016/j.jhazmat.2016.11.062
Naimabadi A, Ghadiri A, Idani E et al (2016) Chemical composition of PM10 and its in vitro toxicological impacts on lung cells during the Middle Eastern Dust (MED) storms in Ahvaz, Iran. Environ Pollut 211:316–324. https://doi.org/10.1016/j.envpol.2016.01.006
Oh SM, Chung KH (2006) Identification of mammalian cell genotoxins in respirable diesel exhaust particles by bioassay-directed chemical analysis. Toxicol Lett 161:226–235. https://doi.org/10.1016/j.toxlet.2005.09.008
Perrone MG, Gualtieri M, Ferrero L et al (2010) Seasonal variations in chemical composition and in vitro biological effects of fine PM from Milan. Chemosphere 78:1368–1377. https://doi.org/10.1016/j.chemosphere.2009.12.071
Schiliro T, Alessandria L, Bonetta S et al (2016) Inflammation response and cytotoxic effects in human THP-1 cells of size-fractionated PM10 extracts in a polluted urban site. Chemosphere 145:89–97. https://doi.org/10.1016/j.chemosphere.2015.11.074
Schiliro T, Bonetta S, Alessandria L et al (2015) PM10 in a background urban site: chemical characteristics and biological effects. Environ Toxicol Pharmacol 39:833–844. https://doi.org/10.1016/j.etap.2015.02.008
Senlin L, Zhenkun Y, Xiaohui C et al (2008) The relationship between physicochemical characterization and the potential toxicity of fine particulates (PM2.5) in Shanghai atmosphere. Atmos Environ 42:7205–7214. https://doi.org/10.1016/j.atmosenv.2008.07.030
Shafer MM, Perkins DA, Antkiewicz DS et al (2010) Reactive oxygen species activity and chemical speciation of size-fractionated atmospheric particulate matter from Lahore, Pakistan: an important role for transition metals. J Environ Monit 12:704–715. https://doi.org/10.1039/B915008K
Sijan Z, Antkiewicz DS, Heo J et al (2015) An in vitro alveolar macrophage assay for the assessment of inflammatory cytokine expression induced by atmospheric particulate matter. Environ Toxicol 30:836–851. https://doi.org/10.1002/tox.21961
Song CL, Huang RJ, Wang YQ et al (2006) Determination of in vitro biotoxicity in exhaust particulate matter from heavy-duty diesel engine. Bull Environ Contam Toxicol 76:24–32. https://doi.org/10.1007/s00128-005-0885-7
Soto KF, Garza KM, Shi Y, Murr LE (2008) Direct contact cytotoxicity assays for filter-collected, carbonaceous (soot) nanoparticulate material and observations of lung cell response. Atmos Environ 42:1970–1982. https://doi.org/10.1016/j.atmosenv.2007.12.027
Steenhof M, Gosens I, Strak M et al (2011) In vitro toxicity of particulate matter (PM) collected at different sites in the Netherlands is associated with PM composition, size fraction and oxidative potential - the RAPTES project. Part Fibre Toxicol. https://doi.org/10.1186/1743-8977-8-26
Thomson EM, Breznan D, Karthikeyan S et al (2015) Cytotoxic and inflammatory potential of size-fractionated particulate matter collected repeatedly within a small urban area. Part Fibre Toxicol. https://doi.org/10.1186/s12989-015-0099-z
Thomson EM, Breznan D, Karthikeyan S et al (2016) Contrasting biological potency of particulate matter collected at sites impacted by distinct industrial sources. Part Fibre Toxicol. https://doi.org/10.1186/s12989-016-0176-y
Totlandsdal AI, Lag M, Lilleaas E et al (2015) Differential proinflammatory responses induced by diesel exhaust particles with contrasting PAH and metal content. Environ Toxicol 30:188–196. https://doi.org/10.1002/tox.21884
Van Den Heuvel R, Den Hond E, Govarts E et al (2016) Identification of PM10 characteristics involved in cellular responses in human bronchial epithelial cells (Beas-2B). Environ Res 149:48–56. https://doi.org/10.1016/j.envres.2016.04.029
Velali E, Papachristou E, Pantazaki A et al (2016) Redox activity and in vitro bioactivity of the water-soluble fraction of urban particulate matter in relation to particle size and chemical composition. Environ Pollut 208:774–786. https://doi.org/10.1016/j.envpol.2015.10.058
Verma MK, Poojan S, Sultana S, Kumar S (2014) Mammalian cell-transforming potential of traffic-linked ultrafine particulate matter PM0.056 in urban roadside atmosphere. Mutagenesis 29:335–340. https://doi.org/10.1093/mutage/geu021
Wagner W, Sachrajda I, Pulaski L et al (2011) Application of cellular biosensors for analysis of bioactivity associated with airborne particulate matter. Toxicol Vitr 25:1132–1142. https://doi.org/10.1016/j.tiv.2011.03.019
Filep Á, Drinovec L, Palágyi A et al (2015) Source specific cyto-and genotoxicity of atmospheric aerosol samples. Aerosol Air Qual Res 15:2325–2331. https://doi.org/10.4209/aaqr.2015.03.0131
Wang W, Shi C, Yan Y et al (2016) Eco-toxicological bioassay of atmospheric fine particulate matter (PM2.5) with Photobacterium Phosphoreum T3. Ecotoxicol Environ Saf 133:226–234. https://doi.org/10.1016/j.ecoenv.2016.07.024
Vouitsis E, Ntziachristos L, Pistikopoulos P et al (2009) An investigation on the physical, chemical and ecotoxicological characteristics of particulate matter emitted from light-duty vehicles. Environ Pollut 157:2320–2327. https://doi.org/10.1016/j.envpol.2009.03.028
Sheesley RJ, Schauer JJ, Hemming JD et al (2004) Toxicity of ambient atmospheric particulate matter from the Lake Michigan (USA) airshed to aquatic organisms. Environ Toxicol Chem 23:133–140. https://doi.org/10.1897/02-636
Kocbach A, Namork E, Schwarze PE (2008) Pro-inflammatory potential of wood smoke and traffic-derived particles in a monocytic cell line. Toxicology 247:123–132. https://doi.org/10.1016/j.tox.2008.02.014
Dilger M, Orasche J, Zimmermann R et al (2016) Toxicity of wood smoke particles in human A549 lung epithelial cells: the role of PAHs, soot and zinc. Arch Toxicol 90:3029–3044. https://doi.org/10.1007/s00204-016-1659-1
Alfaro-Moreno E, Martínez L, García-Cuellar C et al (2002) Biologic effects induced in vitro by PM10 from three different zones of Mexico City. Environ Health Perspect 110:715–720. https://doi.org/10.1289/ehp.02110715
Breznan D, Karthikeyan S, Phaneuf M et al (2016) Development of an integrated approach for comparison of in vitro and in vivo responses to particulate matter. Part Fibre Toxicol. https://doi.org/10.1186/s12989-016-0152-6
Bünger J, Krahl J, Baum K et al (2000) Cytotoxic and mutagenic effects, particle size and concentration analysis of diesel engine emissions using biodiesel and petrol diesel as fuel. Arch Toxicol 74:490–498. https://doi.org/10.1007/s002040000155
Cheng YW, Lee WW, Li CH et al (2004) Genotoxicity of motorcycle exhaust particles in vivo and in vitro. Toxicol Sci 81:103–111. https://doi.org/10.1093/toxsci/kfh173
Colacci A, Vaccari M, Mascolo MG et al (2014) Alternative testing methods for predicting health risk from environmental exposures. Sustainability 6:5265–5283. https://doi.org/10.3390/su6085265
Jiang LP, Dai H, Sun QH et al (2011) Ambient particulate matter on DNA damage in HepG2 cells. Toxicol Ind Health 27:87–95. https://doi.org/10.1177/0748233710387001
Karlsson HL, Ljungman AG, Lindbom J, Moller L (2006) Comparison of genotoxic and inflammatory effects of particles generated by wood combustion, a road simulator and collected from street and subway. Toxicol Lett 165:203–211. https://doi.org/10.1016/j.toxlet.2006.04.003
Liu YQ, Keane M, Ensell M et al (2005) In vitro genotoxicity of exhaust emissions of diesel and gasoline engine vehicles operated on a unified driving cycle. J Environ Monit 7:60–66. https://doi.org/10.1039/b407375d
Orona NS, Astort F, Maglione GA et al (2014) Direct and indirect air particle cytotoxicity in human alveolar epithelial cells. Toxicol Vitr 28:796–802. https://doi.org/10.1016/j.tiv.2014.02.011
Poma A, Limongi T, Pisani C et al (2006) Genotoxicity induced by fine urban air particulate matter in the macrophages cell line RAW 264.7. Toxicol Vitr 20:1023–1029. https://doi.org/10.1016/j.tiv.2006.01.014
Schiliro T, Alessandria L, Degan R et al (2010) Chemical characterisation and cytotoxic effects in A549 cells of urban-air PM10 collected in Torino, Italy. Environ Toxicol Pharmacol 29:150–157. https://doi.org/10.1016/j.etap.2009.12.005
Seagrave J, Mauderly JL, Seilkop SK (2003) In vitro relative toxicity screening of combined particulate and semivolatile organic fractions of gasoline and diesel engine emissions. J Toxicol Environ Heal A 66:1113–1132. https://doi.org/10.1080/15287390390213881
Traversi D, Degan R, De Marco R et al (2008) Mutagenic properties of PM2.5 air pollution in the Padana Plain (Italy) before and in the course of XX Winter Olympic Games of “Torino 2006.”. Environ Int 34:966–970. https://doi.org/10.1016/j.envint.2008.02.007
Turner J, Hernandez M, Snawder JE et al (2015) A toxicology suite adapted for comparing parallel toxicity responses of model human lung cells to diesel exhaust particles and their extracts. Aerosol Sci Technol 49:599–610. https://doi.org/10.1080/02786826.2015.1053559
Vaccari M, Mascolo MG, Rotondo F et al (2015) Identification of pathway-based toxicity in the BALB/c 3T3 cell model. Toxicol Vitr 29:1240–1253. https://doi.org/10.1016/j.tiv.2014.10.002
Watterson TL, Hamilton B, Martin R, Coulombe RA (2009) Urban particulate matter causes ER stress and the unfolded protein response in human lung cells. Toxicol Sci 112:111–122. https://doi.org/10.1093/toxsci/kfp186
Xu HJ, Wang XM, Poesch U et al (2008) Genotoxicity of total and fractionated extractable organic matter in fine air particulate matter from urban Guangzhou: comparison between haze and nonhaze episodes. Environ Toxicol Chem 27:206–212. https://doi.org/10.1897/07-095.1
Yamaguchi T, Yamazaki H (2001) Cytotoxicity of airborne particulates sampled roadside in rodent and human lung fibroblasts. J Heal Sci 47:272–277. https://doi.org/10.1248/jhs.47.272
Kováts N, Ács A, Kovács A et al (2012) Direct contact test for estimating the ecotoxicity of aerosol samples. Environ Toxicol Pharmacol 33:284–287. https://doi.org/10.1016/j.etap.2011.12.021
Anwar K, Ejaz S, Ashraf M et al (2013) Cytotoxic, phytotoxic, and mutagenic appraisal to ascertain toxicological potential of particulate matter emitted from automobiles. Environ Sci Pollut Res 20:4817–4830. https://doi.org/10.1007/s11356-012-1431-x
Olivares A, Van Drooge BL, Casado M et al (2013) Developmental effects of aerosols and coal burning particles in zebrafish embryos. Environ Pollut 178:72–79. https://doi.org/10.1016/j.envpol.2013.02.026
Kováts N, Ács A, Ferincz Á et al (2013) Ecotoxicity and genotoxicity assessment of exhaust particulates from diesel-powered buses. Environ Monit Assess 185:8707–8713. https://doi.org/10.1007/s10661-013-3206-3
Canesi L, Ciacci C, Balbi T (2015) Interactive effects of nanoparticles with other contaminants in aquatic organisms: friend or foe? Mar Environ Res 111:128–134. https://doi.org/10.1016/j.marenvres.2015.03.010
Limbach LK, Wick P, Manser P et al (2007) Exposure of engineered nanoparticles to human lung epithelial cells: influence of chemical composition and catalytic activity on oxidative stress. Environ Sci Technol 41:4158–4163. https://doi.org/10.1021/es062629t
Steinbach C, Bohmer N, Krug HF et al (2017) DaNa 2.0 - verlässliche Informationen zur Sicherheit von marktüblichen Nanomaterialien. DaNa 2.0 - reliable information on the safety of commercially available nanomaterials. Chemie Ing Tech 89:232–238. https://doi.org/10.1002/cite.201600074
Nau K, Bohmer N, Kühnel D, Marquardt C, Paul F, Steinbach C, Krug HF (2016) Communicating current nanosafety research based on evaluated literature data. J Mater Educ 38:93–108
Gminski R, Decker K, Heinz C et al (2011) Genotoxic effects of three selected black toner powders and their dimethyl sulfoxide extracts in cultured human epithelial A549 lung cells in vitro. Environ Mol Mutagen 52:296–309. https://doi.org/10.1002/em.20621
Van Landuyt KL, Cokic SM, Asbach C et al (2016) Interaction of rat alveolar macrophages with dental composite dust. Part Fibre Toxicol. 13:62. https://doi.org/10.1186/s12989-016-0174-0
Acknowledgements
The authors want to thank Prof. Dr.-Ing. Martin Kaltschmitt for pointing us to the issue of toxicity testing of combustion-derived particles and thereby initiating the work on this review.
Funding
This study was partly funded by the German Federal Ministry for Education and Research (BMBF) in the frame of the DaNa2.0 project (Data and knowledge on nanomaterials—Evaluation of socially relevant scientific facts), grant no. 03X0131 (Aug. 2013–July 2017).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 162 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Schmidt, S., Altenburger, R. & Kühnel, D. From the air to the water phase: implication for toxicity testing of combustion-derived particles. Biomass Conv. Bioref. 9, 213–225 (2019). https://doi.org/10.1007/s13399-017-0295-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-017-0295-1