Abstract
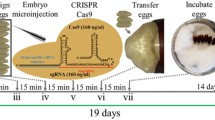
Current methods for gene editing in insects rely on embryonic microinjection, which can be challenging for non-specialist laboratories. Recently, an alternative method known as “direct parental” CRISPR (DIPA-CRISPR) was developed. This method involves injecting commercial Cas9 protein and single-guide RNA into adult females, which can efficiently introduce mutations into developing oocytes. However, its versatility has not been fully explored, particularly in insects that have the most derived, polytrophic meroistic ovaries. In this study, we successfully applied DIPA-CRISPR to the yellow fever mosquito Aedes aegypti, which has polytrophic meroistic ovaries. Following adult injection of Cas9 ribonucleoproteins (Cas9 RNPs) targeting the kynurenine 3-monooxygenase gene, we recovered gene-edited G0 individuals. Injection at 24 h after blood-feeding resulted in the highest gene editing efficiency (3.5%), confirming that a key parameter of DIPA-CRISPR is the stage in which the adult females are injected. Together with our previous study, we demonstrated that DIPA-CRISPR is applicable to all three types of insect ovaries (i.e., panoistic, telotrophic, and polytrophic), which indicates that DIPA-CRISPR is a generalizable approach for insect gene editing.



Similar content being viewed by others
Data availability
Data will be made available on request.
References
Basu S, Aryan A, Overcash JM et al (2015) Silencing of end-joining repair for efficient site-specific gene insertion after TALEN/CRISPR mutagenesis in Aedes aegypti. Proc Natl Acad Sci USA 112:4038–4043. https://doi.org/10.1073/pnas.1502370112
Chaverra-Rodriguez D, Macias VM, Hughes GL et al (2018) Targeted delivery of CRISPR-Cas9 ribonucleoprotein into arthropod ovaries for heritable germline gene editing. Nat Commun 9:1–11. https://doi.org/10.1038/s41467-018-05425-9
Chaverra-Rodriguez D, Dalla Benetta E, Heu CC et al (2020) Germline mutagenesis of Nasonia vitripennis through ovarian delivery of CRISPR-Cas9 ribonucleoprotein. Insect Mol Biol 29:569–577. https://doi.org/10.1111/imb.12663
Daimon T, Uchibori M, Nakao H et al (2015) Knockout silkworms reveal a dispensable role for juvenile hormones in holometabolous life cycle. Proc Natl Acad Sci USA 112:E4226–E4235. https://doi.org/10.1073/pnas.1506645112
Dermauw W, Jonckheere W, Riga M et al (2020) Targeted mutagenesis using CRISPR-Cas9 in the chelicerate herbivore Tetranychus urticae. Insect Biochem Mol Biol 120:103347. https://doi.org/10.1016/j.ibmb.2020.103347
Gantz VM, Akbari OS (2018) Gene editing technologies and applications for insects. Curr Opin Insect Sci 28:66–72. https://doi.org/10.1016/j.cois.2018.05.006
Heu CC, McCullough FM, Luan J, Rasgon JL (2020) CRISPR-Cas9-based genome editing in the silverleaf whitefly (Bemisia tabaci). CRISPR J 3:89–96. https://doi.org/10.1089/crispr.2019.0067
Hwang WY, Fu Y, Reyon D et al (2013) Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat Biotechnol 31:227–229. https://doi.org/10.1038/nbt.2501
Koller CN, Raikhel AS (1991) Initiation of vitellogenin uptake and protein synthesis in the mosquito (Aedes aegypti) ovary in response to a blood meal. J Insect Physiol 37:703–711. https://doi.org/10.1016/0022-1910(91)90048-5
Koller CN, Dhadialla TS, Raikhel AS (1989) Selective endocytosis of vitellogenin by oocytes of the mosquito, Aedes aegypti: An in vitro study. Insect Biochem 19:693–702. https://doi.org/10.1016/0020-1790(89)90106-6
Li X, Xu Y, Zhang H et al (2021) ReMOT control delivery of CRISPR-Cas9 ribonucleoprotein complex to induce germline mutagenesis in the disease vector mosquitoes Culex pipiens pallens (Diptera: Culicidae). J Med Entomol 58:1202–1209. https://doi.org/10.1093/jme/tjab016
Macias VM, McKeand S, Chaverra-Rodriguez D et al (2020) Cas9-mediated gene-editing in the malaria mosquito Anopheles stephensi by ReMOT control. G3-Genes Genom Genet 10:1353–1360. https://doi.org/10.1534/g3.120.401133
Matthews BJ, Vosshall LB (2020) How to turn an organism into a model organism in 10 “easy” steps. J Exp Biol 223:218198. https://doi.org/10.1242/jeb.218198
Mclaughlin JM, Bratu DP (2015) Drosophila melanogaster oogenesis: an overview. Methods Mol Biol 1328:1–20. https://doi.org/10.1007/978-1-4939-2851-4_1
Raikhel AS, Dhadialla TS (1992) Accumulation of yolk proteins in insect oocytes. Annu Rev Entomol 37:217–251. https://doi.org/10.1146/annurev.ento.37.1.217
Sharma A, Pham MN, Reyes JB et al (2022) Cas9-mediated gene editing in the black-legged tick, Ixodes scapularis, by embryo injection and ReMOT Control. iScience 25:103781. https://doi.org/10.1016/j.isci.2022.103781
Shirai Y, Daimon T (2020) Mutations in cardinal are responsible for the red-1 and peach eye color mutants of the red fl our beetle Tribolium castaneum. Biochem Biophys Res Commun 529:372–378. https://doi.org/10.1016/j.bbrc.2020.05.214
Shirai Y, Piulachs M-D, Belles X, Daimon T (2022) DIPA-CRISPR is a simple and accessible method for insect gene editing. Cell Rep. Methods 2:100215. https://doi.org/10.1016/j.crmeth.2022.100215
Acknowledgements
We thank Takahiro Ohde, Toshiya Ando, and the laboratory members for helpful suggestions and discussion, and Misaki Masuda for assistance with mosquito rearing. This research was supported by JSPS KAKENHI (nos. 20K21311and 22K19179 to T.D.) and Moonshot Research and Development Program for Agriculture, Forestry, and Fisheries (Funding Agency: Bio-oriented Technology Research Advancement Institution) (no. JPJ009237 to T.D. and M.O.). Y.S. is a JSPS Research Fellow with a research grant (DC1, 21J20658).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing or financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shirai, Y., Takahashi, M., Ote, M. et al. DIPA-CRISPR gene editing in the yellow fever mosquito Aedes aegypti (Diptera: Culicidae). Appl Entomol Zool 58, 273–278 (2023). https://doi.org/10.1007/s13355-023-00831-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13355-023-00831-y