Abstract
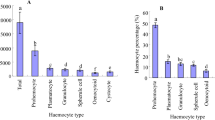
Recognition of foreign targets by insect hemocytes is a crucial first step for insect immunity against invading multicellular organisms in the hemocoel. To understand the mechanism of recognition, we observed the hemocyte behavior of Galleria mellonella Linnaeus (Lepidoptera: Pyralidae) larvae against beads and the nonparasitic nematode Caenorhabditis elegans (Maupas) (Rhabditida: Rhabditidae) in the presence of plasma ex vivo using time-lapse microscopy. Both granular cells and plasmatocytes adhered to and spread on the surface of beads and nematodes. In addition, the spread plasmatocytes actively moved over the beads and nematodes. These results suggest that not only granular cells but also plasmatocytes can recognize foreign targets in the presence of insect plasma and that spread plasmatocytes can actively search for foreign targets. Hemocyte adhesion to beads and nematodes ex vivo was similar to that of the in vivo 1 h after injection. A divalent cation chelator inhibited the spreading and adhesion of plasmatocytes ex vivo, but it did not affect the adhesion of granular cells. The present method enables the analysis of acute hemocyte response against foreign targets in the presence of plasma.




Similar content being viewed by others
References
Bedick JC, Pardy RL, Howard RW, Stanley DW (2000) Insect cellular reactions to the lipopolysaccharide component of the bacterium Serratia marcescens are mediated by eicosanoids. J Insect Physiol 46:1481–1487
Bird DM, Jones JT, Opperman CH, Kikuchi T, Danchin EG (2014) Signatures of adaptation to plant parasitism in nematode genomes. Parasitology 142:S71–S84
Blaxter M, Koutsovoulos G (2014) The evolution of parasitism in Nematoda. Parasitology 142:S26–S39
Brivio MF, Pagani M, Restelli S (2002) Immune suppression of Galleria mellonella (Insecta, Lepidoptera) humoral defenses induced by Steinernema feltiae (Nematoda, Rhabditida): involvement of the parasite cuticle. Exp Parasitol 101:149–156
Horohov DW, Dunn PE (1983) Phagocytosis and nodule formation by hemocytes of Manduca sexta larvae following injection of Pseudomonas aeruginosa. J Invertebr Pathol 41:203–213
Lackie AM (1988) Haemocyte behavior. Adv Insect Physiol 21:85–178
Lavine MD, Strand MR (2001) Surface characteristics of foreign targets that elicit an encapsulation response by the moth Pseudoplusia includens. J Insect Physiol 47:965–974
Lavine MD, Strand MR (2002) Insect haemocyte and their role in immunity. Insect Biochem Mol Biol 32:295–309
Lemaitre B, Hoffmann J (2007) The host defense of Drosophila melanogaster. Annu Rev Immunol 25:697–743
Li X, Cowles EA, Cowles RS, Gaugler R, Cox-Foster DL (2009) Characterization of immunosuppressive surface coat proteins from Steinernema glaseri that selectively kill blood cells in susceptible hosts. Mol Biochem Parasitol 165:162–169
Matsumura T, Taya H, Matsumoto H, Hayakawa Y (2019) Repeated phenotypic selection for cuticular blackness of armyworm larvae decreased stress resistance. J Insect Physiol 117:103889
Ono M, Yoshiga T (2018) Cellular immunity in the insect Galleria mellonella against insect non-parasitic nematodes. Parasitology 146:708–715
Pech LL, Strand MR (1995) Encapsulation of foreign targets by hemocytes of the moth Pseudoplusia includens (Lepidoptera: Noctuidae) involves an RGD-dependent cell adhesion mechanism. J Insect Physiol 41:481–488
Pech LL, Strand MR (1996) Granular cells are required for encapsulation of foreign targets by insect haemocytes. J Cell Sci 109:2053–2060
Ratcliffe NA (1993) Cellular defense responses of insects: unresolved problems. In: Beckage NE, Thompson SN, Federici BA (eds) Parasites, parasites and pathogens of insects, vol I. Academic Press. San Diego, CA, pp 267–304
Sato S, Akai H, Sawada H (1976) An ultrastructrural study of capsule formation by Bombyx hemocytes. Ann Zool Jpn 49:177–188
Schmit AR, Ratcliffe NA (1977) The encapsulation of foreign tissue implants in Galleria mellonella larvae. J Insect Physiol 23:175–184
Stiernagle T (1999) Maintenance of C. elegans. In: Hope I (ed) C. elegans: a practical approach. Oxford University Press, Oxford, pp 51–67
Strand MR (2008) The host defense of Drosophila melanogaster. Insect Sci 15:1–14
Strand MR, Pech LL (1995) Immunological basis for compatibility in parasitoid–host relationships. Annu Rev Entomol 40:31–56
Sudhaus W (2008) Evolution of insect parasitism in rhabditid and diplogastrid nematodes. In: Makarov SE, Dimitrijevic RN (eds) Advances in arachnology and developmental biology. Institute of Zoology, Belgrade, pp 143–161
Tojo S, Naganuma F, Arakawa K, Yokoo S (2000) Involvement of both granular cells and plasmatocytes in phagocytic reactions in the greater wax moth, Galleria mellonella. J Insect Physiol 46:1129–1135
Toubarro D, Avila MM, Montiel R, Simoes N (2013) A pathogenic nematode targets recognition proteins to avoid insect defenses. PLoS One 8:e75691
Tsuzuki S, Matsumoto H, Furihata S, Ryuda M, Tanaka H, Sung EJ, Bird GS, Zhou Y, Shears SB, Hayakawa Y (2014) Switching between humoral and cellular immune responses in Drosophila is guided by the cytokine GBP. Nat Commun 5:4628
Wang X, Fuchs JF, Infanger LC, Rocheleau TA, Hillyer JF, Chen CC, Christensen BM (2005) Mosquito innate immunity: involvement of beta 1,3-glucan recognition protein in melanotic encapsulation immune responses in Armigeres subalbatus. Mol Biochem Parasitol 139:65–73
Wang P, Zhuo XR, Tang L, Liu XS, Wang YF, Wang GX, Yu XQ, Wang JL (2017) C-type lectin interacting with β-integrin enhances hemocytic encapsulation in the cotton bollworm, Helicoverpa armigera. Insect Biochem Mol Biol 86:29–40
Willott E, Hallberg CA, Tran HQ (2002) Influence of calcium on Manduca sexta plasmatocyte spreading and network formation. Arch Insect Biochem Physiol 49:187–202
Wu G, Liu Y, Ding Y, Yi Y (2016) Ultrastructural and functional characterization of circulating hemocytes from Galleria mellonella larva: cell types and their role in the innate immunity. Tissue Cell 48:297–304
Yi YH, Wu GQ, Lv JL, Li M (2016) Eicosanoids mediate Galleria mellonella immune response to hemocoel injection of entomopathogenic nematode cuticles. Parasitol Res 11:597–608
Yokoo S, Gotz P, Tojo S (1995) Phagocytic activities of haemocytes separated by two simple methods from larvae of two lepidopteran species, Agrotis segetum and Galleria mellonella. Appl Entomol Zool 30:342–350
Yu XQ, Kanost MR (2004) Immulectin-2, a pattern recognition receptor that stimulates hemocyte encapsulation and melanization in the tobacco hornworm, Manduca sexta. Dev Comp Immunol 9:891–900
Acknowledgements
The authors would like to thank the Caenorhabditis Genetics Center for providing nematode strain and Enago (www.enago.jp) for the English language review of the manuscript. This work was supported by a Grant-in-Aid for Challenging Exploratory Research (T.Y., Grant Number 23658051) and a Grant-in-Aid for Scientific Research (C) (T.Y, Grant Number 18K05676).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Movie 1. Hemocyte behavior on a Dowex bead. The focus is on the hemocytes on the top of the bead. Granular cells (black arrowheads) and plasmatocytes (white arrowheads) spread soon after descending onto the bead and plasmatocytes actively move over the bead. (MP4 5241 kb)
Movie 2. Hemocyte behavior on a nematode. Granular cells (black arrowheads) remained stationary at the places, where they adhered, but the spread plasmatocytes (white arrowheads) actively moved over the nematode. (MP4 6761 kb)
Movie 3. Hemocyte behavior on a nematode in the presence of EDTA. While some granular cells adhered on the nematode, no spreading or adhesion of plasmatocytes was observed. The black arrowheads indicate granular cells. (MP4 8076 kb)
Rights and permissions
About this article
Cite this article
Ono, M., Arimatsu, C. & Yoshiga, T. Ex vivo observation of insect hemocyte behavior against beads and nematodes in the presence of insect plasma. Appl Entomol Zool 55, 65–70 (2020). https://doi.org/10.1007/s13355-019-00654-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13355-019-00654-w