Abstract
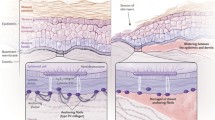
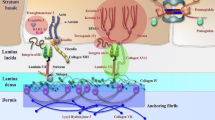
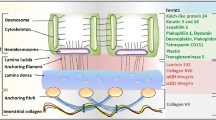
Epidermolysis bullosa (EB) is a group of rare genetic skin fragility disorders, which are hereditary. These disorders are associated with mutations in at least 16 genes that encode components of the epidermal adhesion complex. Currently, there are no effective treatments for this disorder. All current treatment approaches focus on topical treatments to prevent complications and infections. In recent years, significant progress has been achieved in the treatment of the severe genetic skin blistering condition known as EB through preclinical and clinical advancements. Promising developments have emerged in the areas of protein and cell therapies, such as allogeneic stem cell transplantation; in addition, RNA-based therapies and gene therapy approaches have also become a reality. Stem cells obtained from embryonic or adult tissues, including the skin, are undifferentiated cells with the ability to generate, maintain, and replace fully developed cells and tissues. Recent advancements in preclinical and clinical research have significantly enhanced stem cell therapy, presenting a promising treatment option for various diseases that are not effectively addressed by current medical treatments. Different types of stem cells such as primarily hematopoietic and mesenchymal, obtained from the patient or from a donor, have been utilized to treat severe forms of diseases, each with some beneficial effects. In addition, extensive research has shown that gene transfer methods targeting allogeneic and autologous epidermal stem cells to replace or correct the defective gene are promising. These methods can regenerate and restore the adhesion of primary keratinocytes in EB patients. The long-term treatment of skin lesions in a small number of patients has shown promising results through the transplantation of skin grafts produced from gene-corrected autologous epidermal stem cells. This article attempts to summarize the current situation, potential development prospects, and some of the challenges related to the cell therapy approach for EB treatment.


Similar content being viewed by others
Abbreviations
- EB:
-
Epidermolysis bullosa
- TEM:
-
Transmission electron microscopy
- ACT:
-
Autologous cell therapy
- DEB:
-
Dystrophic EB
- JEB:
-
Junctional EB
- EBS:
-
EB simplex
- KEB:
-
Kindler EB
- AR:
-
Autosomal recessive
- RDEB:
-
Recessive dystrophic epidermolysis bullosa
- DDEB:
-
Dominant dystrophic epidermolysis bullosa
- NEBR:
-
National Epidermolysis Bullosa Registry
- ECM:
-
Extracellular matrix
- HMGB1:
-
High mobility group box 1
- PDGF:
-
Platelet-derived growth factor
- T4:
-
Thymosin 4
- iPSCs:
-
Induced pluripotent stem cells
- MSCs:
-
Mesenchymal stem/stromal cells
- BMT:
-
Bone marrow transplantation
- BMZ:
-
Basement membrane zone
- BMMSCs:
-
Bone marrow mesenchymal stem cells
- BM:
-
Bone marrow
- LAM5:
-
Laminin 5
- DSB:
-
Double-strand break
- NHEJ:
-
Non-homologous end joining
- HDR:
-
Homology-directed repair
- AAVS1:
-
Adeno-associated virus integration site 1
- ZFN:
-
Zinc-finger nuclease
- EpSCs:
-
Epithelial stem cells
- IDLVs:
-
Integration-defective lentiviral vectors
- TALE:
-
Transcription activator-like effector
- gRNA:
-
Guide RNA
- KRT14:
-
Keratin 14
- TALENs:
-
Transcription activator-like effector nucleases
- COL7A1:
-
Collagen type VII alpha 1 chain
- KLHL24:
-
Kelch-like family member 24
- BPAG:
-
Bullous pemphigoid antigen
- PLEC1:
-
Plectin1
- HSV:
-
Herpes simplex virus
- CRISPR:
-
Clustered regularly interspaced palindromic repeats
- SMaRT:
-
Spliceosome-mediated RNA trans-splicing
References
Aikawa E et al (2015) Systemic high-mobility group box 1 administration suppresses skin inflammation by inducing an accumulation of PDGFRα+ mesenchymal cells from bone marrow. Sci Rep 5(1):1–13
Alexeev V, Uitto J, Igoucheva O (2011) Gene expression signatures of mouse bone marrow-derived mesenchymal stem cells in the cutaneous environment and therapeutic implications for blistering skin disorder. Cytotherapy 13(1):30–45
Alvin G (2021) Cell therapy: the new approach to der-matology and dermatologic surgery. Clin Surg 5(8):1–14
Angelova-Fischer I et al (2005) Kindler syndrome: a case report and proposal for clinical diagnostic criteria. Acta Dermatovenerol Alp Panonica Adriat 14:61–67
Arabi F, Mansouri V, Ahmadbeigi N (2022) Gene therapy clinical trials, where do we go? An overview. Biomed Pharmacother 153:113324
Arin MJ (2009) The molecular basis of human keratin disorders. Hum Genet 125(4):355–373
Ashton G (2004) Kindler syndrome. Clin Exp Dermatol 29(2):116–121
Ashton G et al (2001) α6β4 integrin abnormalities in junctional epidermolysis bullosa with pyloric atresia. Br J Dermatol 144(2):408–414
Atkinson SD et al (2011) Development of allele-specific therapeutic siRNA for keratin 5 mutations in epidermolysis bullosa simplex. J Investig Dermatol 131(10):2079–2086
Aumailley M et al (2005) A simplified laminin nomenclature. Matrix Biol 24(5):326–332
Aushev M et al (2017) Traceless targeting and isolation of gene-edited immortalized keratinocytes from epidermolysis bullosa simplex patients. Mol Ther Methods Clin Dev 6:112–123
Baardman R et al (2021) Novel insights into the epidemiology of epidermolysis bullosa (EB) from the Dutch EB Registry: EB more common than previously assumed? J Eur Acad Dermatol Venereol 35(4):995–1006
Balistreri CR et al (2020) Stem cell therapy: old challenges and new solutions. Mol Biol Rep 47(4):3117–3131
Bardhan A et al (2020) Epidermolysis bullosa. Nat Rev Dis Primers 6(1):1–27
Bartel RL (2015) Stem cells and cell therapy: autologous cell manufacturing. In: Translational regenerative medicine. Elsevier, pp 107–112
Bartoszewski R, Sikorski AF (2019) Editorial focus: understanding off-target effects as the key to successful RNAi therapy. Cell Mol Biol Lett 24(1):1–23
Bauer J et al (2017) Closure of a large chronic wound through transplantation of gene-corrected epidermal stem cells. J Invest Dermatol 137(3):778–781
Baum C et al (2003) Side effects of retroviral gene transfer into hematopoietic stem cells. Blood, J Am Soc Hematol 101(6):2099–2113
Benati D et al (2018) CRISPR/Cas9-mediated in situ correction of LAMB3 gene in keratinocytes derived from a junctional epidermolysis bullosa patient. Mol Ther 26(11):2592–2603
Bennett CF, Swayze EE (2010) RNA targeting therapeutics: molecular mechanisms of antisense oligonucleotides as a therapeutic platform. Annu Rev Pharmacol Toxicol 50:259–293
Bolling M et al (2011) Mutations in KRT5 and KRT14 cause epidermolysis bullosa simplex in 75% of the patients. Br J Dermatol 164(3):637–644
Bonaventura G et al (2021) Stem cells: innovative therapeutic options for neurodegenerative diseases? Cells 10(8):1992
Bordet T, Behar-Cohen F (2019) Ocular gene therapies in clinical practice: viral vectors and nonviral alternatives. Drug Discov Today 24(8):1685–1693
Bordignon C et al (1999) Cell therapy: achievements and perspectives. Haematologica 84(12):1110–1149
Bornert O et al (2016) Analysis of the functional consequences of targeted exon deletion in COL7A1 reveals prospects for dystrophic epidermolysis bullosa therapy. Mol Ther 24(7):1302–1311
Bornert O et al (2021) QR-313, an antisense oligonucleotide, shows therapeutic efficacy for treatment of dominant and recessive dystrophic epidermolysis bullosa: a preclinical study. J Investig Dermatol 141(4):883–893
Bremer J et al (2016) Antisense oligonucleotide-mediated exon skipping as a systemic therapeutic approach for recessive dystrophic epidermolysis bullosa. Mol Ther Nucleic Acids:5
Bruckner AL et al (2020) The challenges of living with and managing epidermolysis bullosa: insights from patients and caregivers. Orphanet J Rare Dis 15(1):1–14
Bruckner-Tuderman L et al (2013) Progress in epidermolysis bullosa research: summary of DEBRA International Research Conference 2012. J Investig Dermatol 133(9):2121–2126
Carter DM et al (1987) Treatment of junctional epidermolysis bullosa with epidermal autografts. J Am Acad Dermatol 17(2):246–250
Chino T et al (2008) Bone marrow cell transfer into fetal circulation can ameliorate genetic skin diseases by providing fibroblasts to the skin and inducing immune tolerance. Am J Pathol 173(3):803–814
Christiano AM et al (1994) Cloning of human type VII collagen. Complete primary sequence of the alpha 1 (VII) chain and identification of intragenic polymorphisms. J Biol Chem 269(32):20256–20262
Cogan J et al (2014) Aminoglycosides restore full-length type VII collagen by overcoming premature termination codons: therapeutic implications for dystrophic epidermolysis bullosa. Mol Ther 22(10):1741–1752
Coluccio A et al (2013) Targeted gene addition in human epithelial stem cells by zinc-finger nuclease-mediated homologous recombination. Mol Ther 21(9):1695–1704
Condrat I et al (2019) Junctional epidermolysis bullosa: allelic heterogeneity and mutation stratification for precision medicine. Front Med 5:363
Cong L et al (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339(6121):819–823
Conget P et al (2010) Replenishment of type VII collagen and re-epithelialization of chronically ulcerated skin after intradermal administration of allogeneic mesenchymal stromal cells in two patients with recessive dystrophic epidermolysis bullosa. Cytotherapy 12(3):429–431
Coulombe PA, Kerns ML, Fuchs E (2009) Epidermolysis bullosa simplex: a paradigm for disorders of tissue fragility. J Clin Invest 119(7):1784–1793
Cutlar L, Greiser U, Wang W (2014) Gene therapy: pursuing restoration of dermal adhesion in recessive dystrophic epidermolysis bullosa. Exp Dermatol 23(1):1–6
Dang N, Murrell DF (2008) Mutation analysis and characterization of COL7A1 mutations in dystrophic epidermolysis bullosa. Exp Dermatol 17(7):553–568
Das BB, Sahoo S (2004) Dystrophic epidermolysis bullosa. J Perinatol 24(1):41–47
Dasgeb B et al (2018) Colchicine: an ancient drug with novel applications. Br J Dermatol 178(2):350–356
De Luca M et al (2019) Advances in stem cell research and therapeutic development. Nat Cell Biol 21(7):801–811
De Rosa L et al (2020) Toward combined cell and gene therapy for genodermatoses. Cold Spring Harb Perspect Biol 12(5):a035667
Dias N, Stein C (2002) Antisense oligonucleotides: basic concepts and mechanisms. Mol Cancer Ther 1(5):347–355
Dominici M et al (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8(4):315–317
Eady R, Dopping-Hepenstal (2010) Transmission electron microscopy for the diagnosis of epidermolysis bullosa. Dermatol Clin 28(2):211–222
Ebens C et al (2019) Bone marrow transplant with post-transplant cyclophosphamide for recessive dystrophic epidermolysis bullosa expands the related donor pool and permits tolerance of nonhaematopoietic cellular grafts. Br J Dermatol 181(6):1238–1246
Eichstadt S et al (2019a) Phase 1/2a clinical trial of gene-corrected autologous cell therapy for recessive dystrophic epidermolysis bullosa. JCI insight 4(19)
Eichstadt S et al (2019b) Phase 1/2a clinical trial of gene-corrected autologous cell therapy for recessive dystrophic epidermolysis bullosa. JCI. Insight 4(19)
Eisenberg M et al (1987) Successful engraftment of cultured human epidermal allograft in a child with recessive dystrophic epidermolysis bullosa. Med J Aust 147(10):520–521
Farokhforghani S et al (2021) Epidermolysis bullosa registry data in Iran. World J Plast Surg 10(3):99
Felts KA et al (2002) Functional cloning using pFB retroviral cDNA expression libraries. Mol Biotechnol 22(1):25–32
Fine J-D (2010) Inherited epidermolysis bullosa. Orphanet J Rare Dis 5(1):1–17
Fine J-D (2016) Epidemiology of inherited epidermolysis bullosa based on incidence and prevalence estimates from the National Epidermolysis Bullosa Registry. JAMA Dermatol 152(11):1231–1238
Fine J-D et al (1991) Revised clinical and laboratory criteria for subtypes of inherited epidermolysis bullosa: a consensus report by the subcommittee on diagnosis and classification of the National Epidermolysis Bullosa Registry. J Am Acad Dermatol 24(1):119–135
Fine J-D et al (2008) The classification of inherited epidermolysis bullosa (EB): report of the third international consensus meeting on diagnosis and classification of EB. J Am Acad Dermatol 58(6):931–950
Fine J-D et al (2014) Inherited epidermolysis bullosa: updated recommendations on diagnosis and classification. J Am Acad Dermatol 70(6):1103–1126
Firing C, Bygum A (2017) Epidermolysis bullosa. Ugeskr Laeger 179(47)
Fontao L et al (2004) Molecular consequences of deletion of the cytoplasmic domain of bullous pemphigoid 180 in a patient with predominant features of epidermolysis bullosa simplex. J Investig Dermatol 122(1):65–72
Foster L, Holmes Y (2007) Transition from paediatric to adult service in epidermolysis bullosa. Br J Nurs 16(4):244–246
Fry LE et al (2020) RNA editing as a therapeutic approach for retinal gene therapy requiring long coding sequences. Int J Mol Sci 21(3):777
Fujita Y et al (2021) Intravenous allogeneic multilineage-differentiating stress-enduring cells in adults with dystrophic epidermolysis bullosa: a phase 1/2 open-label study. J Eur Acad Dermatol Venereol 35(8):e528
Gallico GG III et al (1984) Permanent coverage of large burn wounds with autologous cultured human epithelium. N Engl J Med 311(7):448–451
Gear R et al (2022) Epidemiological, clinical, pathological and genetic characteristics of epidermolysis bullosa in New Zealand. Australas J Dermatol 63(1):62–67
Goldschneider KR et al (2014) Pain care for patients with epidermolysis bullosa: best care practice guidelines. BMC Med 12(1):1–23
Gostyńska KB et al (2019) Allogeneic haematopoietic cell transplantation for epidermolysis bullosa: the Dutch experience. Acta Derm Venereol 99(3):347–348
Goto M et al (2006) Targeted skipping of a single exon harboring a premature termination codon mutation: implications and potential for gene correction therapy for selective dystrophic epidermolysis bullosa patients. J Investig Dermatol 126(12):2614–2620
Green H, Kehinde O, Thomas J (1979) Growth of cultured human epidermal cells into multiple epithelia suitable for grafting. Proc Natl Acad Sci 76(11):5665–5668
Guleria I et al (2022) How do I: evaluate the safety and legitimacy of unproven cellular therapies? Transfusion 62(3):518–532
Guo C et al (2023) Off-target effects in CRISPR/Cas9 gene editing. Front Bioeng Biotechnol 11:1143157
Gurevich I et al (2022) In vivo topical gene therapy for recessive dystrophic epidermolysis bullosa: a phase 1 and 2 trial. Nat Med 28(4):780–788
Haniffa MA et al (2007) Adult human fibroblasts are potent immunoregulatory cells and functionally equivalent to mesenchymal stem cells. J Immunol 179(3):1595–1604
Haniffa MA et al (2009) Mesenchymal stem cells: the fibroblasts’ new clothes? Haematologica 94(2):258–263
Has C (2017) The “Kelch” surprise: KLHL24, a new player in the pathogenesis of skin fragility. J Investig Dermatol 137(6):1211–1212
Has C, South A, Uitto J (2020b) Molecular therapeutics in development for epidermolysis bullosa: update 2020. Mol Diagn Ther 24(3):299–309
Has C et al (2018) Epidermolysis bullosa: molecular pathology of connective tissue components in the cutaneous basement membrane zone. Matrix Biol 71:313–329
Has C et al (2020a) Consensus reclassification of inherited epidermolysis bullosa and other disorders with skin fragility. Br J Dermatol 183(4):614–627
Hauschild-Quintern J et al (2013) Gene knockout and knockin by zinc-finger nucleases: current status and perspectives. Cell Mol Life Sci 70(16):2969–2983
Heagerty A et al (1987) Rapid prenatal diagnosis of epidermolysis bullosa letalis using GB3 monoclonal antibody. Br J Dermatol 117(3):271–275
Herz C et al (2006) Kindlin-1 is a phosphoprotein involved in regulation of polarity, proliferation, and motility of epidermal keratinocytes. J Biol Chem 281(47):36082–36090
Hirsch T et al (2017) Regeneration of the entire human epidermis using transgenic stem cells. Nature 551(7680):327–332
Hou P-C et al (2021) Investigational treatments for epidermolysis bullosa. Am J Clin Dermatol 22:801–817
Hou Y et al (2015) Intravenously administered recombinant human type VII collagen derived from Chinese hamster ovary cells reverses the disease phenotype in recessive dystrophic epidermolysis bullosa mice. J Investig Dermatol 135(12):3060–3067
Hourd P et al (2021) Regulatory challenges for the manufacture and scale-out of autologous cell therapies. Int J Biomed Health Sci 10(4)
Hünefeld C et al (2013) One goal, different strategies–molecular and cellular approaches for the treatment of inherited skin fragility disorders. Exp Dermatol 22(3):162–167
Ito K et al (2009) Keratinocyte-/fibroblast-targeted rescue of Col7a1-disrupted mice and generation of an exact dystrophic epidermolysis bullosa model using a human COL7A1 mutation. Am J Pathol 175(6):2508–2517
Kaipe H et al (2015) Immunogenicity of decidual stromal cells in an epidermolysis bullosa patient and in allogeneic hematopoietic stem cell transplantation patients. Stem Cells Dev 24(12):1471–1482
Kashpur O et al (2019) Differentiation of diabetic foot ulcer–derived induced pluripotent stem cells reveals distinct cellular and tissue phenotypes. FASEB J 33(1):1262–1277
Kaviarasan P, Prasad PV, Shradda PV (2005) Kindler syndrome. Indian J Dermatol Venereol Leprol 71:348
Kazmi B, Inglefield CJ, Lewis M (2009) Autologous cell therapy: current treatments and future prospects. Wounds 21(9):234–242
Keith AR et al (2020) Leading edge: emerging drug, cell, and gene therapies for junctional epidermolysis bullosa. Expert Opin Biol Ther 20(8):911–923
Kern JS et al (2009) Mechanisms of fibroblast cell therapy for dystrophic epidermolysis bullosa: high stability of collagen VII favors long-term skin integrity. Mol Ther 17(9):1605–1615
Khaniani MS et al (2015) One novel frameshift mutation on exon 64 of COL7A1 gene in an Iranian individual suffering recessive dystrophic epidermolysis bullosa. Ann Clin Lab Sci 45(5):582–584
Kho YC et al (2010) Epidemiology of epidermolysis bullosa in the antipodes: the Australasian Epidermolysis Bullosa Registry with a focus on Herlitz junctional epidermolysis bullosa. Arch Dermatol 146(6):635–640
Kiritsi D et al (2011) Molecular mechanisms of phenotypic variability in junctional epidermolysis bullosa. J Med Genet 48(7):450–457
Kittridge A et al (2014) Herlitz junctional epidermolysis bullosa with a novel mutation in LAMB 3. Pediatr Dermatol 31(4):530–532
Kligys KR et al (2012) α6β4 integrin, a master regulator of expression of integrins in human keratinocytes. J Biol Chem 287(22):17975–17984
Kocher T, Koller U (2021) Advances in gene editing strategies for epidermolysis bullosa. Prog Mol Biol Transl Sci 182:81–109
Kocher T et al (2017) Cut and paste: efficient homology-directed repair of a dominant negative KRT14 mutation via CRISPR/Cas9 nickases. Mol Ther 25(11):2585–2598
Kocher T et al (2019) Improved double-nicking strategies for COL7A1-editing by homologous recombination. Mol Ther Nucleic Acids 18:496–507
Kocher T et al (2020) Predictable CRISPR/Cas9-mediated COL7A1 reframing for dystrophic epidermolysis bullosa. J Investig Dermatol 140(10):1985–1993
Koller U et al (2014) Considerations for a successful RNA trans-splicing repair of genetic disorders. Mol Ther Nucleic Acids:3
Kopp J et al (2005) Hematopoietic stem cell transplantation and subsequent 80% skin exchange by grafts from the same donor in a patient with Herlitz disease. Transplantation 79(2):255–256
Kulkarni JA et al (2021) The current landscape of nucleic acid therapeutics. Nat Nanotechnol 16(6):630–643
Kulus M et al (2021) Mesenchymal stem/stromal cells derived from human and animal perinatal tissues—origins, characteristics, signaling pathways, and clinical trials. Cells 10(12):3278
Kumar S et al (2014) Autologous therapies in dermatology. J Clin Aesthet Dermatol 7(12):38
Lai-Cheong J et al (2009a) Kindler syndrome: a focal adhesion genodermatosis. Br J Dermatol 160(2):233–242
Lai-Cheong JE, McGrath JA (2010) Kindler syndrome. Dermatol Clin 28(1):119–124
Lai-Cheong JE et al (2009b) Loss-of-function FERMT1 mutations in kindler syndrome implicate a role for fermitin family homolog-1 in integrin activation. Am J Pathol 175(4):1431–1441
Lane E, McLean W (2004) Keratins and skin disorders. J Pathol: A J Pathol Soc Great Britain and Ireland 204(4):355–366
Larouche J et al (2018) Immune regulation of skin wound healing: mechanisms and novel therapeutic targets. Adv Wound Care 7(7):209–231
Latella MC et al (2017) Correction of recessive dystrophic epidermolysis bullosa by transposon-mediated integration of COL7A1 in transplantable patient-derived primary keratinocytes. J Investig Dermatol 137(4):836–844
Lee SE et al (2021) Intravenous allogeneic umbilical cord blood–derived mesenchymal stem cell therapy in recessive dystrophic epidermolysis bullosa patients. JCI insight 6(2)
Lehn P et al (1998) Gene delivery systems: bridging the gap between recombinant viruses and artificial vectors. Adv Drug Deliv Rev 30(1-3):5–11
Lei Y et al (2013) Generation of gene disruptions by transcription activator-like effector nucleases (TALENs) in Xenopus tropicalis embryos. Cell Biosci 3(1):1–10
Lukomska B et al (2019) Challenges and controversies in human mesenchymal stem cell therapy. Stem Cells Int 2019
March OP, Kocher T, Koller U (2020) Context-dependent strategies for enhanced genome editing of genodermatoses. Cells 9(1):112
Mariath LM et al (2020) Inherited epidermolysis bullosa: update on the clinical and genetic aspects. An Bras Dermatol 95:551–569
Marinkovich MP, Bauer E (2008) Epidermolysis bullosa. Fitzpatrick’s dermatology in general medicine, 7th edn. McGraw-Hill, New York, pp 505–516
Marinkovich MP, Tang JY (2019) Gene therapy for epidermolysis bullosa. J Investig Dermatol 139(6):1221–1226
Maseda R et al (2020) Beneficial effect of systemic allogeneic adipose derived mesenchymal cells on the clinical, inflammatory and immunologic status of a patient with recessive dystrophic epidermolysis bullosa: a case report. Front Med 7:576558
Mathes DW et al (2014) Simultaneous transplantation of hematopoietic stem cells and a vascularized composite allograft leads to tolerance. Transplantation 98(2):131
Mavilio F et al (2006) Correction of junctional epidermolysis bullosa by transplantation of genetically modified epidermal stem cells. Nat Med 12(12):1397–1402
McAllister TN, Audley D, L’Heureux N (2012) Autologous cell therapies: challenges in US FDA regulation. Regen Med 7(6 Suppl):94–97
McGrath J, Eady R, Pope F (2004) Anatomy and organization of human skin. Rook’s Textbook Dermatol 1:3.2-3.80
McGrath JA et al (1993) Cultured keratinocyte allografts and wound healing in severe recessive dystrophic epidermolysis bullosa. J Am Acad Dermatol 29(3):407–419
McLean WI, Smith FJ, Cassidy AJ (2005) Insights into genotype–phenotype correlation in pachyonychia congenita from the human intermediate filament mutation database. In: Journal of Investigative Dermatology Symposium Proceedings. Elsevier
Melo SP et al (2014) Somatic correction of junctional epidermolysis bullosa by a highly recombinogenic AAV variant. Mol Ther 22(4):725–733
Mencía Á et al (2018) Deletion of a pathogenic mutation-containing exon of COL7A1 allows clonal gene editing correction of RDEB patient epidermal stem cells. Mol Ther Nucleic Acids 11:68–78
Michalak A et al (2018) A rare case of skin blistering and esophageal stenosis in the course of epidermolysis bullosa-case report and literature review. BMC Gastroenterol 18(1):1–5
Moravvej H et al (2016) Comparison of the application of allogeneic fibroblast and autologous mesh grafting with the conventional method in the treatment of third-degree burns. J Burn Care Res 37(1):e90–e95
Mousaei Ghasroldasht M et al (2022) Stem cell therapy: from idea to clinical practice. Int J Mol Sci 23(5):2850
Mullard A (2023) FDA approves first topical gene therapy. Nature reviews. Drug Discov
Murauer EM et al (2013) A reporter-based screen to identify potent 3’trans-splicing molecules for endogenous RNA repair. Hum Gene Ther Methods 24(1):19–27
Murauer EM et al (2015) Advances in gene/cell therapy in epidermolysis bullosa. Keio J Med:2014-0013-RE
Murrell DF (2009) Life with epidermolysis bullosa (EB): etiology, diagnosis, multidisciplinary care and therapy. J Am Acad Dermatol 61(6):1092–1093
Nagy N et al (2011) HB-EGF induces COL7A1 expression in keratinocytes and fibroblasts: possible mechanism underlying allogeneic fibroblast therapy in recessive dystrophic epidermolysis bullosa. J Investig Dermatol 131(8):1771–1774
Naso G, Petrova A (2020) Cellular therapy options for genetic skin disorders with a focus on recessive dystrophic epidermolysis bullosa. Br Med Bull 136(1):30–45
Natsuga K et al (2021) Current topics in epidermolysis bullosa: pathophysiology and therapeutic challenges. J Dermatol Sci 104(3):164–176
Niti A, Koliakos G, Michopoulou A (2023) Stem cell therapies for epidermolysis bullosa treatment. Bioengineering 10(4):422
Nourian Dehkordi A et al (2019) Skin tissue engineering: wound healing based on stem-cell-based therapeutic strategies. Stem Cell Res Ther 10(1):1–20
Nyström A, Bernasconi R, Bornert O (2018) Therapies for genetic extracellular matrix diseases of the skin. Matrix Biol 71:330–347
Nyström A, Bruckner-Tuderman L, Kern JS (2013) Cell-and protein-based therapy approaches for epidermolysis bullosa. In: Molecular Dermatology. Springer, pp 425–440
Nyström A et al (2015) Losartan ameliorates dystrophic epidermolysis bullosa and uncovers new disease mechanisms. EMBO Mol Med 7(9):1211–1228
Oever MV et al (2016) miR-29 regulates type VII collagen in recessive dystrophic epidermolysis bullosa. J Investig Dermatol 136(10):2013–2021
Ormond KE et al (2019) The clinical application of gene editing: ethical and social issues. Pers Med 16(4):337–350
Osborn MJ et al (2020) Base editor correction of COL7A1 in recessive dystrophic epidermolysis bullosa patient-derived fibroblasts and iPSCs. J Investig Dermatol 140(2):338–347
Pânzaru M-C et al (2022) Epidermolysis bullosa—a different genetic approach in correlation with genetic heterogeneity. Diagnostics 12(6):1325
Patel PM et al (2021) A review of acquired autoimmune blistering diseases in inherited epidermolysis bullosa: implications for the future of gene therapy. Antibodies 10(02):19
Peking P et al (2017) An RNA-targeted therapy for dystrophic epidermolysis bullosa. Nucleic Acids Res 45(17):10259–10269
Peking P et al (2019) An ex vivo RNA trans-splicing strategy to correct human generalized severe epidermolysis bullosa simplex. Br J Dermatol 180(1):141–148
Petrof G et al (2013) Fibroblast cell therapy enhances initial healing in recessive dystrophic epidermolysis bullosa wounds: results of a randomized, vehicle-controlled trial. Br J Dermatol 169(5):1025–1033
Petrof G et al (2015) Potential of systemic allogeneic mesenchymal stromal cell therapy for children with recessive dystrophic epidermolysis bullosa. J Invest Dermatol 135(9):2319
Petrof G et al (2022) The epidemiology of epidermolysis bullosa in England and Wales: data from the national epidermolysis bullosa database. Br J Dermatol 186(5):843–848
Petrova A, Ilic D, McGrath J (2010) Stem cell therapies for recessive dystrophic epidermolysis bullosa. Br J Dermatol 163(6):1149–1156
Pfendner E, Uitto J (2005) Plectin gene mutations can cause epidermolysis bullosa with pyloric atresia. J Investig Dermatol 124(1):111–115
Pfendner, E.G. and A.W. Lucky, Junctional epidermolysis bullosa. 2018a.
Pfendner, E.G. and A.W. Lucky, Dystrophic epidermolysis bullosa. 2018b.
Piipponen M, Li D, Landén NX (2020) The immune functions of keratinocytes in skin wound healing. Int J Mol Sci 21(22):8790
Prasongchean W, Ferretti (2012) Autologous stem cells for personalised medicine. New Biotechnol 29(6):641–650
Prodinger C et al (2019) Epidermolysis bullosa: advances in research and treatment. Exp Dermatol 28(10):1176–1189
Rashidghamat E, McGrath JA (2017) Novel and emerging therapies in the treatment of recessive dystrophic epidermolysis bullosa. Intractable Rare Dis Res 6(1):6–20
Rashidghamat E et al (2020) Phase I/II open-label trial of intravenous allogeneic mesenchymal stromal cell therapy in adults with recessive dystrophic epidermolysis bullosa. J Am Acad Dermatol 83(2):447–454
Remington J et al (2009) Injection of recombinant human type VII collagen corrects the disease phenotype in a murine model of dystrophic epidermolysis bullosa. Mol Ther 17(1):26–33
Ren D et al (2022) Immune responses to gene editing by viral and non-viral delivery vectors used in retinal gene therapy. Pharmaceutics 14(9):1973
Rheinwatd JG, Green H (1975) Seria cultivation of strains of human epidemal keratinocytes: the formation keratinizin colonies from single cell is. Cell 6(3):331–343
Riedl J et al (2021) ABCB5+ dermal mesenchymal stromal cells with favorable skin homing and local immunomodulation for recessive dystrophic epidermolysis bullosa treatment. Stem Cells 39(7):897–903
Robbins PD, Ghivizzani SC (1998) Viral vectors for gene therapy. Pharmacol Ther 80(1):35–47
Rossi S et al (2021) Immunofluorescence mapping, electron microscopy and genetics in the diagnosis and sub-classification of inherited epidermolysis bullosa: a single-centre retrospective comparative study of 87 cases with long-term follow-up. J Eur Acad Dermatol Venereol 35(4):1007–1016
Saunderson RB et al (2019) A retrospective cohort study evaluating the accuracy of clinical diagnosis compared with immunofluorescence and electron microscopy in children with inherited epidermolysis bullosa. Br J Dermatol 180(5):1258–1259
Sayed N et al (2022) Gene therapy: comprehensive overview and therapeutic applications. Life Sci 294:120375
Schwieger-Briel A et al (2015) A COL7A1 variant leading to in-frame skipping of exon 15 attenuates disease severity in recessive dystrophic epidermolysis bullosa. Br J Dermatol 173(5):1308–1311
Sfeir A, Symington LS (2015) Microhomology-mediated end joining: a back-up survival mechanism or dedicated pathway? Trends Biochem Sci 40(11):701–714
Shahryari A et al (2019) Development and clinical translation of approved gene therapy products for genetic disorders. Front Genet 10:868
Shamis Y et al (2012) iPSC-derived fibroblasts demonstrate augmented production and assembly of extracellular matrix proteins. In Vitro Cell Dev Biol Anim 48(2):112–122
Shams F et al (2021) The utility of dermal fibroblasts in treatment of skin disorders: a paradigm of recessive dystrophic epidermolysis bullosa. Dermatol Ther 34(4):e15028
Sharma P et al (2021) Stem cells and growth factors-based delivery approaches for chronic wound repair and regeneration: a promise to heal from within. Life Sci 268:118932
Shinkuma S (2015) Dystrophic epidermolysis bullosa: a review. Clin Cosmet Investig Dermatol 8:275
Siprashvili Z et al (2010) Long-term type VII collagen restoration to human epidermolysis bullosa skin tissue. Hum Gene Ther 21(10):1299–1310
Siprashvili Z et al (2016) Safety and wound outcomes following genetically corrected autologous epidermal grafts in patients with recessive dystrophic epidermolysis bullosa. Jama 316(17):1808–1817
Siprashvili Z et al (2017) Phase I/IIa clinical trial for recessive dystrophic epidermolysis bullosa using genetically corrected autologous keratinocytes. J Invest Dermatol 137(5 Suppl. 1):S89
Solis DC et al Classification of two distinct wound types in recessive dystrophic epidermolysis bullosa: a retrospective and cohort natural history study. J Am Acad Dermatol 85(5):1296–1298
Sorg H et al (2017) Skin wound healing: an update on the current knowledge and concepts. Eur Surg Res 58(1-2):81–94
Stublar A, Dragos V, Dolenc-Voljc M (2021) Inherited epidermolysis bullosa: epidemiology and patient care in Slovenia with a review of the updated classification. Acta Dermatovenerol Alp Panon Adriat 30(2):63–67
Sugawara K et al (2008) Laminin-332 and-511 in skin. Exp Dermatol 17(6):473–480
Takeichi T et al (2015) Founder mutation in dystonin-e underlying autosomal recessive epidermolysis bullosa simplex in Kuwait. Br J Dermatol 172(2):527–531
Tamai K, Uitto J (2016) Stem cell therapy for epidermolysis bullosa—does it work? J Investig Dermatol 136(11):2119–2121
Tamai K et al (2011) PDGFRα-positive cells in bone marrow are mobilized by high mobility group box 1 (HMGB1) to regenerate injured epithelia. Proc Natl Acad Sci 108(16):6609–6614
Tang JY et al (2021) A systematic literature review of the disease burden in patients with recessive dystrophic epidermolysis bullosa. Orphanet J Rare Dis 16(1):1–25
Techanukul T et al (2011) Novel and recurrent FERMT1 gene mutations in Kindler syndrome. Acta Derm Venereol 91(3):267
Titeux M et al (2020) Emerging drugs for the treatment of epidermolysis bullosa. Expert Opin Emerg Drugs 25(4):467–489
Tockner B et al (2016) Construction and validation of an RNA trans-splicing molecule suitable to repair a large number of COL7A1 mutations. Gene Ther 23(11):775–784
Tolar J, Blazar BR, Wagner JE (2011) Concise review: transplantation of human hematopoietic cells for extracellular matrix protein deficiency in epidermolysis bullosa. Stem Cells 29(6):900–906
Tolar J, Wagner JE (2012) Management of severe epidermolysis bullosa by haematopoietic transplant: principles, perspectives and pitfalls. Exp Dermatol 21(12):896–900
Tolar J, Wagner JE (2015) A biologic Velcro patch. N Engl J Med 372(4):382–384
Tolar J et al (2009) Amelioration of epidermolysis bullosa by transfer of wild-type bone marrow cells. Blood, J Am Soc Hematol 113(5):1167–1174
Tong Y, Zuo J, Yue D (2021) Application prospects of mesenchymal stem cell therapy for bronchopulmonary dysplasia and the challenges encountered. Biomed Res Int 2021
Turczynski, S., et al., Antisense-mediated exon skipping to reframe transcripts. Exon Skipping: Methods and Protocols, 2012: 221-238.
Uitto J et al (2010) Progress in epidermolysis bullosa research: toward treatment and cure. J Investig Dermatol 130(7):1778–1784
Uitto J et al (2016) Progress toward treatment and cure of epidermolysis bullosa: summary of the DEBRA International Research Symposium EB2015. J Investig Dermatol 136(2):352–358
Uitto J et al (2018) EB2017—progress in epidermolysis bullosa research toward treatment and cure. J Investig Dermatol 138(5):1010–1016
Vahidnezhad H et al (2017) Dystrophic epidermolysis bullosa: COL7A1 mutation landscape in a multi-ethnic cohort of 152 extended families with high degree of customary consanguineous marriages. J Investig Dermatol 137(3):660–669
Vahidnezhad H et al (2019) Phenotypic spectrum of epidermolysis bullosa: the paradigm of syndromic versus non-syndromic skin fragility disorders. J Investig Dermatol 139(3):522–527
Vanden Oever M et al (2018) Inside out: regenerative medicine for recessive dystrophic epidermolysis bullosa. Pediatr Res 83(1):318–324
Vander Beken S et al (2019) Newly defined ATP-binding cassette subfamily B member 5 positive dermal mesenchymal stem cells promote healing of chronic iron-overload wounds via secretion of interleukin-1 receptor antagonist. Stem Cells 37(8):1057–1074
Varki R et al (2006) Epidermolysis bullosa. I. Molecular genetics of the junctional and hemidesmosomal variants. J Med Genet 43(8):641–652
Večerić-Haler Ž et al (2021) Multiorgan failure with fatal outcome after stem cell tourism. Eur J Med Res 26(1):1–5
Venugopal SS et al (2013) A phase II randomized vehicle-controlled trial of intradermal allogeneic fibroblasts for recessive dystrophic epidermolysis bullosa. J Am Acad Dermatol 69(6):898–908
Virtanen I et al (1996) Laminin chains in the basement membranes of human thymus. Histochem J 28:643–650
Vivinus-Nebot MN et al (2004) Mature human thymocytes migrate on laminin-5 with activation of metalloproteinase-14 and cleavage of CD44. J Immunol 172(3):1397–1406
Wagner JE et al (2010) Bone marrow transplantation for recessive dystrophic epidermolysis bullosa. N Engl J Med 363(7):629–639
Wally V et al (2008) 5′ trans-splicing repair of the PLEC1 gene. J Investig Dermatol 128(3):568–574
Wang R et al (2022) Novel variants in LAMA3 and COL7A1 and recurrent variant in KRT5 underlying epidermolysis bullosa in five Chinese families. Front Med:1–7
Wang X (2014) The roles of type VII collagen in wound healing and scar reduction. University of Southern California
Weber F et al (2001) Squamous cell carcinoma in junctional and dystrophic epidermolysis bullosa. Acta Derm Venereol 81(3)
Wong T et al (2008) Potential of fibroblast cell therapy for recessive dystrophic epidermolysis bullosa. J Investig Dermatol 128(9):2179–2189
Woodley DT et al (2004) Injection of recombinant human type VII collagen restores collagen function in dystrophic epidermolysis bullosa. Nat Med 10(7):693–695
Woodley DT et al (2007) Intravenously injected human fibroblasts home to skin wounds, deliver type VII collagen, and promote wound healing. Mol Ther 15(3):628–635
Woodley DT et al (2013) Intravenously injected recombinant human type VII collagen homes to skin wounds and restores skin integrity of dystrophic epidermolysis bullosa. J Invest Dermatol 133(7):1910
Wright JT, Fine J, Johnson L (1993) Hereditary epidermolysis bullosa: oral manifestations and dental management. Pediatr Dent 15:242–242
Wu T-L, Ertl HC (2009) Immune barriers to successful gene therapy. Trends Mol Med 15(1):32–39
Wu Y et al (2007) Bone marrow-derived stem cells in wound healing: a review. Wound Repair Regen 15:S18–S26
Yamada N, Shioya N, Kuroyanagi Y (1995) Evaluation of an allogeneic cultured dermal substitute composed of fibroblasts within a spongy collagen matrix as a wound dressing. Scand J Plast Reconstr Surg Hand Surg 29(3):211–219
Yenamandra V et al (2018) Cardiomyopathy in patients with epidermolysis bullosa simplex with mutations in KLHL24. Br J Dermatol 179(5):1181–1183
Youssefian, L., H. Vahidnezhad, and J. Uitto, Kindler syndrome synonym: congenital bullous poikiloderma. 2016, Seattle: Pagon RA, Adam MP, Ardinger HH, et al.
Youssefian, L., H. Vahidnezhad, and J. Uitto, Kindler syndrome. 2022.
Youssefian L et al (2015) The Kindler syndrome: a spectrum of FERMT1 mutations in Iranian families. J Invest Dermatol 135(5):1447–1450
Zhang X et al (2017) CRISPR/Cas9 system: a powerful technology for in vivo and ex vivo gene therapy. Sci China Life Sci 60(5):468–475
Zidorio A et al (2018) Effectiveness of gastrostomy for improving nutritional status and quality of life in patients with epidermolysis bullosa: a systematic review. Br J Dermatol 179(1):42–49
Availability of data and material
Not applicable.
Funding
This study is financially supported by the student research committee of Behbahan Faculty of Medical Sciences (Grant Number: 402042).
Author information
Authors and Affiliations
Contributions
Writing and drafting: Fatemeh Gila, Vahab Alamdari-Palangi, Maedeh Rafiee, Arezoo Jokar, and Sajad Ehtiaty. Data curation: Aria Dianatinasab, Seyyed Hossein Khatami, and Mortaza Taheri-Anganeh. Conceptualization: Jafar Fallahi. Supervision: Ahmad Movahedpour.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by: Ewa Ziętkiewicz
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gila, F., Alamdari-Palangi, V., Rafiee, M. et al. Gene-edited cells: novel allogeneic gene/cell therapy for epidermolysis bullosa. J Appl Genetics (2024). https://doi.org/10.1007/s13353-024-00839-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13353-024-00839-2