Abstract
Chronic and allergic rhinosinusitis impacts approximately 12% of the global population. Challenges in rhinosinusitis treatment include paranasal sinus inaccessibility and variability in delivery efficiency among individuals. This study addresses these challenges of drug delivery by developing a high-efficiency, low-variability protocol for nasal drop delivery to the ostiomeatal complex (OMC) and maxillary sinus. Patient-specific nasal casts were dissected to reveal the configurations of conchae and meatus, providing insights into anatomical features amendable for sinus delivery. Fluorescent dye-enhanced videos visualized the dynamic liquid translocation in transparent nasal casts, allowing real-time assessment and quick adjustment to delivery parameters. Dosimetry to the OMC and maxillary sinus were quantified as drop count and mass using a precision scale. Key delivery factors, including the device type, formulation, and head-chin orientation, were systematically investigated in a cohort of ten nasal casts. Results show that both the squeeze bottle and soft-mist nasal pump yielded notably low doses to the OMC with high variability, and no dose from these two devices was detected within the maxillary sinuses. In contrast, the proposed approach, which included a curved nozzle surpassing the nasal valve and leveraged gravity-driven liquid translocation along the lateral nasal wall, delivered significant doses to the OMC and maxillary sinus. Iterative experimentations identified the optimal head tilt to be 40° and chin tilt to be° from the lateral recumbent position. Statistical analyses established the drop count required for effective OMC/sinus delivery. The proposed delivery protocol holds the potential to enhance chronic rhinosinusitis treatment outcomes with low variability. The dual role of nasal anatomy in posing challenges and offering opportunities highlights the need for future investigations using diverse formulations in a larger cohort of nasal models.
Graphical Abstract
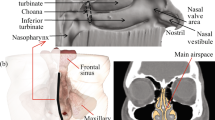
Optimized gravity-driven intranasal drop administration delivers significant doses to the ostiomeatal complex and maxillary sinus.















Similar content being viewed by others
Availability of data and materials
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Palmer JN, Messina JC, Biletch R, Grosel K, Mahmoud RA. A cross-sectional, population-based survey of U.S. adults with symptoms of chronic rhinosinusitis. Allergy Asthma Proc. 2019;40:48–56.
Albu S. Chronic rhinosinusitis—an update on epidemiology, pathogenesis and management. J Clin Med. 2020;9:2285.
Laube BL. Devices for aerosol delivery to treat sinusitis. J Aerosol Med. 2007;20(Suppl 1):S5-17.
Rudmik L. Economics of chronic rhinosinusitis. Curr Allergy Asthma Rep. 2017;17:20.
Baumann I, Blumenstock G, Zalaman IM, Praetorius M, Klingmann C, et al. Impact of gender, age, and comorbidities on quality of life in patients with chronic rhinosinusitis. Rhinology. 2007;45:268–72.
Jurlin L, Gregurić T, Baudoin T, Grgić MV, Pažanin L, et al. Cluster analysis of chronic rhinosinusitis suggests gender-based differences. ORL J Otorhinolaryngol Relat Spec. 2019;81:1–9.
Adams DR, Xu LJ, Vickery TW, Scangas GA, Bleier BS, et al. The impact of gender on long-term quality of life after sinus surgery for chronic rhinosinusitis. Laryngoscope. 2023. https://doi.org/10.1002/lary.30719.
Gill AS, Ashby S, Oakley GM, Steele TO, Menjivar D, et al. Comorbidities known to affect physical function negatively impact baseline health-related quality-of-life in patients with chronic rhinosinusitis. Am J Rhinol Allergy. 2022;36:25–32.
Jankowski R, Gallet P, Nguyen DT, Rumeau C. Chronic rhinosinusitis of adults: new definition, new diagnosis. Rev Prat. 2019;69:274–8.
Othieno F, Schlosser RJ, Rowan NR, Storck KA, Mattos JL, et al. Taste impairment in chronic rhinosinusitis. Int Forum Allergy Rhinol. 2018;8:783–9.
Han P, Whitcroft KL, Fischer J, Gerber J, Cuevas M, et al. Olfactory brain gray matter volume reduction in patients with chronic rhinosinusitis. Int Forum Allergy Rhinol. 2017;7:551–6.
Lin S, Nie M, Wang B, Duan S, Huang Q, et al. Intrinsic brain abnormalities in chronic rhinosinusitis associated with mood and cognitive function. Front Neurosci. 2023;17:1131114.
Huang SF, Lee TJ, Lin KL. Concomitant bilateral orbital and brain abscesses–unusual complications of pediatric rhinosinusitis. Chang Gung Med J. 2005;28:51–5.
Ziegler A, Patadia M, Stankiewicz J. Neurological complications of acute and chronic sinusitis. Curr Neurol Neurosci Rep. 2018;18:5.
Whyte A, Boeddinghaus R. The maxillary sinus: physiology, development and imaging anatomy. Dentomaxillofac Radiol. 2019;48:20190205.
Harbo G, Grau C, Bundgaard T, Overgaard M, Elbrønd O, et al. Cancer of the nasal cavity and paranasal sinuses. A clinico-pathological study of 277 patients. Acta Oncol. 1997;36:45–50.
Brzost J, Czerwaty K, Dżaman K, Ludwig N, Piszczatowska K, et al. Perspectives in therapy of chronic rhinosinusitis. Diagnostics. 2022;12:2301.
Shrestha K, Wong E, Salati H, Fletcher DF, Singh N, et al. Liquid volume and squeeze force effects on nasal irrigation using volume of fluid modelling. Exp Comput Multiph Flow. 2022;4:445–64.
Farrell NF, Klatt-Cromwell C, Schneider JS. Benefits and safety of nasal saline irrigations in a pandemic-washing COVID-19 away. JAMA Otolaryngol Head Neck Surg. 2020;146:787–8.
Verma RR, Verma R. Sinonasal irrigation after endoscopic sinus surgery—past to present and future. Indian J Otolaryngol Head Neck Surg. 2023;75:1–8.
Abdullah B, Periasamy C, Ismail R. Nasal irrigation as treatment in sinonasal symptoms relief: a review of its efficacy and clinical applications. Indian J Otolaryngol Head Neck Surg. 2019;71:1718–26.
Sansila K, Eiamprapai P, Sawangjit R. Effects of self-prepared hypertonic nasal saline irrigation in allergic rhinitis: a randomized controlled trial. Asian Pac J Allergy Immunol. 2020;38:200–7.
Shi J, Tan L, Ye J, Hu L. Hypertonic saline and mannitol in patients with traumatic brain injury: a systematic and meta-analysis. Medicine. 2020;99:e21655.
Salati H, Bartley J, White DE. Nasal saline irrigation—a review of current anatomical, clinical and computational modelling approaches. Respir Physiol Neurobiol. 2020;273:103320.
Salati H, Bartley J, Yazdi SG, Jermy M, White DE. Neti pot irrigation volume filling simulation using anatomically accurate in-vivo nasal airway geometry. Respir Physiol Neurobiol. 2021;284:103580.
Inthavong K, Shang Y, Wong E, Singh N. Characterization of nasal irrigation flow from a squeeze bottle using computational fluid dynamics. Int Forum Allergy Rhinol. 2020;10:29–40.
Siddiqui Z, Tahiri M, Gupta A, Kin Nam RH, Rachmanidou A. The management of paediatric rhinosinusitis. Int J Pediatr Otorhinolaryngol. 2021;147:110786.
Medikeri G, Javer A. Optimal management of allergic fungal rhinosinusitis. J Asthma Allergy. 2020;13:323–32.
Bartier S, Coste A, Bequignon E. Management strategies for chronic rhinosinusitis with nasal polyps in adults. Rev Mal Respir. 2021;38:183–98.
Amjadimanesh H, Faramarzi M, Sadrizadeh S, Abouali O. Micro-particle deposition in maxillary sinus for various sizes of opening in a virtual endoscopic surgery. Exp Comput Multiph Flow. 2023;5:262–71.
Alsaleh S, Manji J, Javer A. Optimization of the surgical field in endoscopic sinus surgery: an evidence-based approach. Curr Allergy Asthma Rep. 2019;19:8.
Li Y, Konuthula N, Humphreys IM, Moe K, Hannaford B, et al. Real-time virtual intraoperative CT in endoscopic sinus surgery. Int J Comput Assist Radiol Surg. 2022;17:249–60.
Zhao K, Craig JR, Cohen NA, Adappa ND, Khalili S, et al. Sinus irrigations before and after surgery—visualization through computational fluid dynamics simulations. Laryngoscope. 2016;126:E90-96.
Wu Z, Craig JR, Maza G, Li C, Otto BA, et al. Peak sinus pressures during sneezing in healthy controls and post-skull base surgery patients. Laryngoscope. 2020;130:2138–43.
Sun Q, Dong J, Zhang Y, Tian L, Tu J. Numerical study of the effect of nasopharynx airway obstruction on the transport and deposition of nanoparticles in nasal airways. Exp Comput Multiph Flow. 2022;4:399–408.
Xi J, Lei LR, Zouzas W, April Si X. Nasally inhaled therapeutics and vaccination for COVID-19: developments and challenges. MedComm. 2021;2:569–86.
Xi J. Development and challenges of nasal spray vaccines for short-term COVID-19 protection. Curr Pharm Biotechnol. 2022;23:1671–7.
Sélam JL. Inhaled insulin: Promises and concerns. J Diabetes Sci Technol. 2008;2:311–5.
Djupesland PG, Skretting A, Winderen M, Holand T. Bi-directional nasal delivery of aerosols can prevent lung deposition. J Aerosol Med. 2004;17:249–59.
Xi J, Wang Z, Nevorski D, White T, Zhou Y. Nasal and olfactory deposition with normal and bidirectional intranasal delivery techniques: in vitro tests and numerical simulations. J Aerosol Med Pulm Drug Deliv. 2017;30:118–31.
Xi J, Wang Z, Si XA, Zhou Y. Nasal dilation effects on olfactory deposition in unilateral and bi-directional deliveries: in vitro tests and numerical modeling. Eur J Pharm Sci. 2018;118:113–23.
Seifelnasr A, Talaat M, Ramaswamy P, Si XA, Xi J. A supine position and dual-dose applications enhance spray dosing to the posterior nose: paving the way for mucosal immunization. Pharmaceutics. 2023;15:359.
Djupesland PG. Nasal drug delivery devices: Characteristics and performance in a clinical perspective-a review. Drug Deliv Transl Res. 2013;3:42–62.
Leclerc L, Pourchez J, Aubert G, Leguellec S, Vecellio L, et al. Impact of airborne particle size, acoustic airflow and breathing pattern on delivery of nebulized antibiotic into the maxillary sinuses using a realistic human nasal replica. Pharm Res. 2014;31:2335–43.
Maniscalco M, Sofia M, Weitzberg E, Lundberg JO. Sounding airflow enhances aerosol delivery into the paranasal sinuses. Eur J Clin Invest. 2006;36:509–13.
Durand M, Pourchez J, Aubert G, Le Guellec S, Navarro L, et al. Impact of acoustic airflow nebulization on intrasinus drug deposition of a human plastinated nasal cast: new insights into the mechanisms involved. Int J Pharm. 2011;421:63–71.
Xi J, Si XA, Peters S, Nevorski D, Wen T, et al. Understanding the mechanisms underlying pulsating aerosol delivery to the maxillary sinus: in vitro tests and computational simulations. Int J Pharm. 2017;520:254–66.
Xi J, Yuan JE, Si XA, Hasbany J. Numerical optimization of targeted delivery of charged nanoparticles to the ostiomeatal complex for treatment of rhinosinusitis. Int J Nanomedicine. 2015;10:4847–61.
Möller W, Schuschnig U, Bartenstein P, Meyer G, Häussinger K, et al. Drug delivery to paranasal sinuses using pulsating aerosols. J Aerosol Med Pulm Drug Deliv. 2014;27:255–63.
Möller W, Schuschnig U, Celik G, Münzing W, Bartenstein P, et al. Topical drug delivery in chronic rhinosinusitis patients before and after sinus surgery using pulsating aerosols. PLoS ONE. 2013;8:e74991.
Foo MY, Cheng YS, Su WC, Donovan MD. The influence of spray properties on intranasal deposition. J Aerosol Med. 2007;20:495–508.
Cheng YS, Holmes TD, Gao J, Guilmette RA, Li S, et al. Characterization of nasal spray pumps and deposition pattern in a replica of the human nasal airway. J Aerosol Med. 2001;14:267–80.
Pourmehran O, Zarei K, Pourchez J, Vreugde S, Psaltis A, et al. Advancements in acoustic drug delivery for paranasal sinuses: A comprehensive review. Int J Pharm. 2023;644:123277.
Tai J, Lee K, Kim TH. Current perspective on nasal delivery systems for chronic rhinosinusitis. Pharmaceutics. 2021;13:246.
Xi J, Yuan JE, Zhang Y, Nevorski D, Wang Z, et al. Visualization and quantification of nasal and olfactory deposition in a sectional adult nasal airway cast. Pharm Res. 2016;33:1527–41.
Seifelnasr A, Talaat M, Si XA, Xi J. Delivery of agarose-aided sprays to the posterior nose for mucosa immunization and short-term protection against infectious respiratory diseases. Curr Pharm Biotechnol. 2024;25:In press.
Cankurtaran M, Celik H, Coşkun M, Hizal E, Cakmak O. Acoustic rhinometry in healthy humans: accuracy of area estimates and ability to quantify certain anatomic structures in the nasal cavity. Ann Otol Rhinol Laryngol. 2007;116:906–16.
Avrunin OG, Nosova YV, Abdelhamid IY, Pavlov SV, Shushliapina NO, et al. Research active posterior rhinomanometry tomography method for nasal breathing determining violations. Sensors. 2021;21:8508.
Kwah JH, Peters AT. Nasal polyps and rhinosinusitis. Allergy Asthma Proc. 2019;40:380–4.
Seifelnasr A, Si XA, Xi J. Visualization and estimation of nasal spray delivery to olfactory mucosa in an image-based transparent nasal model. Pharmaceutics. 2023;15:1657.
Xi J, Si XA. A next-generation vaccine for broader and long-lasting COVID-19 protection. MedComm. 2022;3:e138.
Talaat M, Si X, Liu X, Xi J. Count-and mass-based dosimetry of MDI spray droplets with polydisperse and monodisperse size distributions. Int J Pharm. 2022;623:121920.
Xi J, Talaat M, Si XA. Two-way coupling and Kolmogorov scales on inhaler spray plume evolutions from Ventolin, ProAir, and Qvar. Math Biosci Eng. 2022;19:10915–40.
Zhao K, Malhotra P, Rosen D, Dalton P, Pribitkin EA. Computational fluid dynamics as surgical planning tool: a pilot study on middle turbinate resection. Anat Rec (Hoboken). 2014;297:2187–95.
Kim K, Otto BA, Farag AA, Zhao K. Topical irrigation against gravity may lead to better sinus penetration. Int Forum Allergy Rhinol. 2021;11:198–200.
Lepley TJ, Kim K, Ardizzone M, Kelly KM, Otto BA, et al. 3D printing as a planning tool to optimize sinonasal irrigation. Ann Otol Rhinol Laryngol. 2023:34894221149242.
Alfaifi A, Hosseini S, Esmaeili AR, Walenga R, Babiskin A, et al. Anatomically realistic nasal replicas capturing the range of nasal spray drug delivery in adults. Int J Pharm. 2022;622:121858.
Maaz A, Blagbrough IS, De Bank PA. In vitro evaluation of nasal aerosol depositions: an insight for direct nose to brain drug delivery. Pharmaceutics. 2021;13.
Williams G, Suman JD. In vitro anatomical models for nasal drug delivery. Pharmaceutics. 2022;14:1353.
Xi J, Si XA, Kim J, Zhang Y, Jacob RE, et al. Anatomical details of the rabbit nasal passages and their implications in breathing, air conditioning, and olfaction. Anat Rec. 2016.
Xi J, Yuan J, Alshaiba M, Cheng D, Firlit Z, et al. Design and testing of electric-guided delivery of charged particles to the olfactory region: experimental and numerical studies. Curr Drug Deliv. 2015;13:1–10.
Si XA, Talaat M, Xi J. Effects of guiding vanes and orifice jet flow of a metered-dose inhaler on drug dosimetry in human respiratory tract. Exp Comput Multiph Flow. 2023;5:247–61.
Si X, Xi J, Kim J. Effect of laryngopharyngeal anatomy on expiratory airflow and submicrometer particle deposition in human extrathoracic airways. Open J Fluid Dyn. 2013;3:286–301.
Fedorov A, Beichel R, Kalpathy-Cramer J, Finet J, Fillion-Robin JC, et al. 3D Slicer as an image computing platform for the ouantitative imaging network. Magn Reson Imaging. 2012;30:1323–41.
Xi J, Kim J, Si XA, Corley RA, Zhou Y. Modeling of inertial deposition in scaled models of rat and human nasal airways: towards in vitro regional dosimetry in small animals. J Aerosol Sci. 2016;99:78–93.
Xi J, Si X, Kim J, Su G, Dong H. Modeling the pharyngeal anatomical effects on breathing resistance and aerodynamically generated sound. Med Biol Eng Comput. 2014;52:567–77.
Kabilan S, Suffield SR, Recknagle KP, Jacob RE, Einstein DR, et al. Computational fluid dynamics modeling of Bacillus anthracis spore deposition in rabbit and human respiratory airways. J Aerosol Sci. 2016;99:64–77.
Zare F, Aalaei E, Zare F, Faramarzi M, Kamali R. Targeted drug delivery to the inferior meatus cavity of the nasal airway using a nasal spray device with angled tip. Comput Methods Programs Biomed. 2022;221:106864.
Lehr C-M, Bouwstra JA, Boddé HE, Junginger HE. A surface energy analysis of mucoadhesion: contact angle measurements on polycarbophil and pig intestinal mucosa in physiologically relevant fluids. Pharm Res. 1992;9:70–5.
Acknowledgements
Dr. Chantal Darquenne at UCSD and Dr. Richard Corley at Greek Creek Toxicokinetics Consulting were gratefully acknowledged for sharing the M2 nasal model used in this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Amr Seifelnasr, Farhad Zare, Xiuhua April Si, and Jinxiang Xi. The first draft of the manuscript was written by Amr Seifelnasr, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This is an observational study. The UMass Lowell Research Ethics Committee has confirmed that no ethical approval is required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Authors AS FZ and XSA declare they have no financial interests. Author JX has received research funding from AmPhastar Pharmaceuticals Inc. and MedSpray Inc.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Seifelnasr, A., Zare, F., Si, X.A. et al. Optimized gravity-driven intranasal drop administration delivers significant doses to the ostiomeatal complex and maxillary sinus. Drug Deliv. and Transl. Res. (2023). https://doi.org/10.1007/s13346-023-01488-4
Accepted:
Published:
DOI: https://doi.org/10.1007/s13346-023-01488-4