Abstract
Inhaled drug delivery is a promising approach to achieving high lung drug concentrations to facilitate efficient treatment of tuberculosis (TB) and to reduce the overall duration of treatment. Rifampicin is a good candidate for delivery via the pulmonary route. There have been no clinical studies yet at relevant inhaled doses despite the numerous studies investigating its formulation and preclinical properties for pulmonary delivery. This review discusses the clinical implications of pulmonary drug delivery in TB treatment, the drug delivery systems reported for pulmonary delivery of rifampicin, animal models, and the animal studies on inhaled rifampicin formulations, and the research gaps hindering the transition from preclinical development to clinical investigation. A review of reports in the literature suggested there have been minimal attempts to test inhaled formulations of rifampicin in laboratory animals at relevant high doses and there is a lack of appropriate studies in animal models. Published studies have reported testing only low doses (≤ 20 mg/kg) of rifampicin, and none of the studies has investigated the safety of inhaled rifampicin after repeated administration. Preclinical evaluations of inhaled anti-TB drugs, such as rifampicin, should include high-dose formulations in preclinical models, determined based on allometric conversions, for relevant high-dose anti-TB therapy in humans.
Graphical abstract
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Current status of TB and approach to TB treatment
TB is caused by Mycobacterium tuberculosis (Mtb) and mainly affects the lungs, but can also affect extra-pulmonary sites. Mtb is transmitted when aerosols from an infected person are inhaled by another person. The World Health Organization (WHO), in its 2021 TB report, stated that TB was the top cause of death worldwide from a single infectious agent in 2019 [1]. There were 5.8 million new TB cases diagnosed and reported in 2020, among which approximately 1.3 million cases were fatal [1]. A decline in the number of new TB cases and an increase in the number of TB deaths, compared to those in 2019, was observed in 2020 due to the COVID-19 pandemic that resulted in reduced access to TB diagnosis and treatment [1]. The majority of TB illnesses occur in low-resource countries, and over 85% of new TB cases occur in the 30 countries with the highest TB burden [1].
The recommended treatment for drug-susceptible TB is a 6-month rifampicin-based regimen of orally administered drugs, which includes the administration of isoniazid (H), rifampicin (R), pyrazinamide (Z), and ethambutol (E) (HRZE) for 2 months (daily or thrice weekly), followed by 4 months of HR [2]. Treatment regimens have become more complicated with the rise in complications of TB, such as multi-drug resistant TB (MDR-TB), co-infection with HIV, comorbidities such as diabetes, or TB retreatment after relapse. Approaches to treating drug-resistant TB may involve the inclusion of an injectable antibiotic, such as streptomycin, kanamycin, and amikacin, or lengthening treatment duration up to 20 months with drugs to which the organism is susceptible [3]. The real-world success rates of TB treatment regimens are lower than expected due to various factors such as low patient adherence to the lengthy treatment regimen, the existence of Mtb deep inside human TB lesions, and the latency of Mtb facilitating its survival within the host by up- and down-regulation of specific genes [4].
Mtb usually resides within complex granulomatous lesions, which allow the pathogen to avoid the bactericidal effects of antibiotics by limiting their transportation from blood to the bacterial target site and contribute to treatment failure, relapse of disease, and the emergence of drug-resistant strains of the bacteria [4, 5]. This presents a hurdle for efficient drug delivery to the complex lesions of pulmonary TB via the oral route. This also requires the administration of high drug doses since only a small fraction of the orally administered dose reaches the target sites in the lungs [6]. Moreover, there is a risk of adverse effects from the chronic administration of drugs.
Among the many reasons for the spread of MDR-TB is chaotic treatment exacerbated by poor health care systems, the amplifier effect of short-course chemotherapy, community transmission, and nosocomial transmission in places with high TB burdens [7]. Moreover, exposure of the mycobacteria to sub-therapeutic levels of anti-TB drugs during treatment is a driver for the emergence of drug-resistant microbial strains [8]. One effective approach to increase the bactericidal effect of the antibiotics is to achieve a high local concentration of the drug by localized drug delivery, such as pulmonary administration.
As outlined above, TB treatment remains a challenge despite the availability of potent drugs, and this has driven a search for clinically efficacious treatment approaches that can shorten treatment duration. There has been a huge investment in the development of newer anti-TB drugs, with several drugs already in the ‘drug development pipeline’ and at various stages of clinical trials [9]. These drug candidates are promising and have novel targets to achieve the efficient bactericidal activity, but their journey from the initial drug development phase and clinical trials to clinical practice is still a few years away [9, 10]. TB treatment approaches should also focus on better delivery options for drugs that are currently available and are effective against Mtb.
Pulmonary drug delivery approach for TB treatment: evidence from preclinical and clinical studies
Pulmonary delivery of anti-TB drugs is expected to deliver high drug concentrations at the desired site, i.e., to the granulomatous TB lesions as well as the systemic circulation [11, 12]. The pulmonary route can be exploited for its advantages, such as the large surface area of the lungs, rich vascularization, and thin barrier to systemic circulation, which allows noninvasive administration of drugs for local and systemic effects while avoiding first-pass metabolism and achieving rapid onset at the same time, during TB treatment [13]. Both local and systemic effects of the drugs are desirable in inhaled TB therapy because achieving adequate drug levels in the blood, together with that in the lungs, is expected to achieve better therapeutic results in both pulmonary and extra-pulmonary TB.
Pulmonary delivery of antibiotic therapy has already been used effectively in other clinical settings to treat infection and combat drug resistance. Clinical studies in respiratory intensive care unit patients have shown that aerosolized antibiotics can eradicate pathogens causing respiratory infection and reduce the incidence of newly resistant organisms [14, 15]. Mtb is a major pulmonary pathogen with a high incidence of multidrug resistance [16, 17], and inhaled therapy has the potential to reduce resistance by ensuring maximum concentration of the drug at the target site for efficient bacterial killing [18]. The pulmonary route increases drug delivery to TB lesions in the lungs and drug retention. The pulmonary route can also be used for systemic drug delivery to treat extra-pulmonary TB. Ideally, aerosol particles of the inhalable size range (1–5 µm in diameter) are desired for deep lung delivery, even though smaller particles such as the nanoparticles serve the purpose better for intracellular targeting and uptake by the macrophages. Therefore, approaches such as nano-in-micro particle systems have been used to achieve both good aerosolization as well as improved cellular uptake after deposition in the lungs [19].
Pulmonary delivery of drugs to the lungs can be achieved in several ways: by aerosolization of drug powder particles using dry powder inhalers (DPI), by nebulization of a drug solution or suspension using nebulizers, by liquid aerosolization using metered dose inhalers (MDIs), and by direct drug administration to the upper respiratory tract using insufflators [6]. Due to the requirement of high drug doses in TB treatment and the application of MDIs being limited to aerosolization of low drug doses, MDIs have not been researched for this purpose [20]. Nebulizers and DPIs of drugs for several other respiratory diseases, such as chronic obstructive pulmonary disease (COPD) and asthma, are well-established clinically. DPIs are preferred for their convenience in usage during inhalation delivery; their advantages include propellant-free formulations, minimized formulation-related problems, and a lesser need for patient coordination [21, 22]. Compared to formulations for nebulization, DPI formulations offer better stability of drug products due to their solid nature, are convenient, and allow quick administration, while nebulization requires a significant duration of time for the administration.
Several current and new anti-TB drugs have been studied for their inhalable formulation development, in vitro and in vivo evaluation, and only one of them has been studied in humans but at a subtherapeutic dose [23, 24]. Preclinical studies reporting on inhaled drugs for TB treatment suggest that inhaled delivery can ensure higher drug concentrations in the lung and achieve better therapeutic effects against TB with lower doses and toxicity, compared to oral and injectable administration. An investigation of inhaled rifampicin formulations in rats [25] suggested an increased drug concentration in the alveolar macrophages from intra-tracheal administration compared to oral administration. Similar results were observed in the case of a dry powder formulation of isoniazid [26]. In an infectious disease model [27], respirable microspheres containing rifampicin showed promise in reducing the burden of bacteria in the lungs. Another antitubercular drug, pyrazinamide, was found to achieve an absolute bioavailability of 66% when drug-loaded large porous particles were administered to guinea pigs by intra-tracheal insufflation [28]. In vitro efficacy and in vivo safety of repeated intra-tracheal administration of the same drug have also been reported [29]. Capreomycin is another promising drug candidate for inhaled delivery in the treatment of pulmonary tuberculosis. Inhaled capreomycin has been reported to have low toxicity from in vivo acute toxicity studies [30]. Another example of a promising inhaled anti-tubercular drug is ofloxacin, which has also demonstrated improved treatment efficacy against tuberculosis following pulmonary administration compared to other routes of administration, such as the intravenous and the oral route [31]. Newer drugs such as PA-824, Spectinamide-1599, All-trans-retinoic acid, and para-aminosalicylic acid also have shown promise as inhaled therapies in animal studies. Large porous particles of PA-824 were reported to achieve high drug concentrations in the lungs after pulmonary administration compared to oral administration to guinea pigs [32]. Similarly, guinea pigs with experimental TB showed lower inflammation, bacterial burden, and tissue damage when treated with a dry powder inhaled formulation of PA-824 compared to nontreated or placebo groups [33]. A study conducted in mice reported the reproducibility of dry powder administration of a spray-dried formulation of Spectinamide-1599, also demonstrating dose-dependent exposure of this promising anti-TB drug [34]. The efficacy of an inhaled formulation of all trans-retinoic-acid (ATRA) was reported in mice in which the inhaled treatment significantly reduced the bacterial burden and pulmonary pathology following just three doses [35]. In another study, direct delivery of para-aminosalicylic acid to the rat lungs was reported to achieve rapid and higher local concentration and similar systemic concentration of the drug, compared to oral delivery [36].
Only a few clinical studies on inhaled anti-TB drugs have been reported, and they include studies on rifampicin and capreomycin dry powder inhalers. A clinical study of a low dose (2 mg) of rifampicin delivered as a dry powder inhaled formulation [24] suggested that it did not provoke inflammatory cytokines and was safe to healthy subjects. In another study, the safety and pharmacokinetics of capreomycin delivered via dry powder inhaler was tested in humans up to 300 mg nominal doses [37]. The study reported safety to the lungs, rapid drug absorption after inhalation, and serum drug concentrations higher than the MIC of the drug for Mtb.
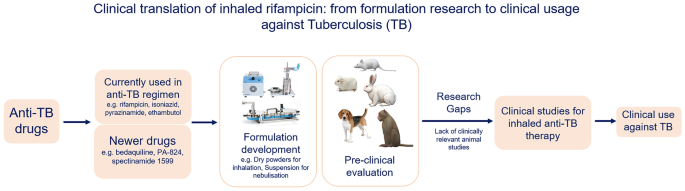
In the following sections, the formulations and animal studies reported in the literature for inhaled rifampicin are reviewed, and the considerations for preclinical studies of inhaled anti-TB drugs are discussed with a focus on inhaled rifampicin.
Rifampicin: a long-established oral anti-TB drug with new potential via the inhaled route
Rifampicin (also known as rifampin) is a semisynthetic derivative of rifamycin B that exhibits its bactericidal effect on mycobacteria by inhibiting their DNA-dependent RNA synthesis [38]. It was discovered in 1965 as a promising rifamycin derivative for oral use and was approved for oral delivery against pulmonary TB by the USFDA in 1971. Since then, rifampicin has been central to TB treatment. Together with isoniazid and pyrazinamide, it is used as the first-line agent against both pulmonary and extra-pulmonary TB at a dose of 10 mg/kg/day [39]. This dose was chosen to avoid any dose-dependent toxicity and because of its expense when it initially entered clinical use [40]; however, it is now evident that higher doses of oral rifampicin are well tolerated by humans and are more effective than the conventional 10 mg/kg/day (maximum of 600 mg/day) doses in achieving higher drug concentration in the blood [41, 42].
Rifampicin is the most promising anti-TB drug for development as inhaled therapy due to its high efficacy in sterilizing TB lesions and preventing relapse, a relatively long history of use in humans, and well-documented pharmacokinetic and pharmacodynamic properties from the oral route. Rifampicin is also considered the best candidate for improving therapeutic outcomes in TB treatment if used at a higher oral dose than in current regimens. A dose-ranging clinical trial has already demonstrated that rifampicin doses up to 35 mg/kg from the oral route are safe as well as efficient in achieving high systemic drug concentration (AUC0-24 up to 235 h·mg/L and Cmax up to 35.2 mg/L) and reducing time to sputum culture conversion with higher doses [42]. Several other clinical trials have also demonstrated the safety and/or efficacy of high-dose oral rifampicin [41, 43,44,45]. Therefore, rifampicin is likely to remain the backbone of anti-TB regimens for the years to come, and despite calls for higher oral doses, they have not yet been implemented for pulmonary TB in clinical practice [46].
High oral dose of rifampicin for TB treatment may be limited by the increased risk of systemic toxicities and there are challenges such as its unknown pharmacodynamics alone and in combination with other medications. Due to this reason, research has focused on other ways of delivering rifampicin more efficiently [46]. Delivering rifampicin by inhalation is a potential alternative to achieve high drug concentration in the lungs as well as systemic circulation with a smaller dose. After pulmonary delivery, high blood concentration can be achieved at a smaller dose than the oral dose due to rapid drug absorption through the large surface area and minimal drug metabolism in the lung [47, 48], which may reduce the likelihood of dose-dependent systemic toxicity of rifampicin. Inhaled rifampicin has a huge potential for clinical translation and could be used as an adjunct to current oral anti-TB regimens for improved therapeutic outcomes in TB patients.
Inhaled rifampicin: formulation types and preparation techniques
The dry powder formulations of anti-TB drugs for inhaled therapy against TB have been extensively reviewed previously [20, 49, 50]. Similarly, Mehanna et al. and Chae et al. have reviewed inhalable nanoparticulate systems for anti-TB drugs [51, 52]. In the present review, we have focused on the different types of inhalable formulations of rifampicin. A wide range of anti-TB drugs has been investigated for their potential as inhaled formulations suitable for lung delivery, suggesting a huge interest among researchers for inhaled anti-TB therapy (Table 1). Among these, rifampicin is the most commonly studied, either alone or in combination with other drugs, and dry powder formulations for inhalation have been studied more frequently than nebulized formulations. Spray drying is the most common technique utilized to obtain the final inhalable formulation of dry powder rifampicin with good aerodynamic properties (Table 1), although other techniques include jet milling [53] and micronization [54].
Formulations for inhaled rifampicin
Various drug delivery systems are reported in the literature for inhaled delivery of rifampicin, such as microparticle systems, liposomes, solid lipid nanoparticles (SLN), polymeric nanoparticles, porous particles, nanoaggregates, and nanocomposites (Table 2). Recently, micellar systems, such as polymeric micelles and nanomicelles, also have gained attention as a promising formulation approach for inhaled delivery of rifampicin [55, 56]. Such formulations have shown good retention after intra-tracheal administration in animal models (Table 2). Although promising in enhancing drug bioavailability and localized retention, these carrier-based formulation systems need further investigation of their systemic absorption, degradation, and clearance from the body [57].
Rifampicin is also a good drug candidate for inhaled delivery after loading into micro and nano-carrier-based systems. A variety of inhalable carrier systems for rifampicin have been reported in the literature, ranging from polymers, lipids, non-polymeric carbohydrates, and mesoporous silica [58]. A nanoparticle system for rifampicin was reported by Joshi and Prabhakar in which bovine serum albumin was used to obtain nanoparticles for rifampicin loading [59]. In another study, a spray-dried nanocomposite platform for inhaled rifampicin was reported, in which the powder formulation was obtained after spray-drying a nanosuspension prepared by antisolvent-precipitation and ultrasonication [60].
While formulations based on all of the above delivery systems can be prepared into a dry powder formulation, some liposomal formulations are also suitable for delivery by nebulization of the suspension without subsequent drying [61, 62]. Similarly, a pressurized packed system of rifampicin liposomal formulation in chlorofluorocarbon aerosol propellants has also been reported [63]. Liposomal formulations in pulmonary delivery are desired for targeted delivery of drugs to specific cells such as the alveolar macrophages, to formulate poorly soluble drugs, to control drug release after administration, and to improve drug bioavailability in the lung tissue [61, 64, 65]. Since TB treatment usually requires a high dose of the antibiotic, liposomal systems may not be suitable for formulating anti-TB drugs because of challenges in loading a high amount of drug into the liposomes. On the other hand, liposomes can be utilized for targeted delivery of rifampicin to the alveolar macrophages in the lung. Solid lipid nanoparticles and polymeric nanoparticles are other delivery systems both suitable for pulmonary drug delivery to achieve controlled and targeted delivery of anti-TB drugs [66]. However, both these systems are unsuitable when high-dose delivery of a drug to the lungs is desired. Therefore, among the delivery systems, the microparticle system is most suitable to achieve high-dose delivery of drugs via inhalation since it can be prepared with minimal or no excipients using common techniques like spray drying. Spray drying allows the engineering of particles by manipulation of its process variables, such as the feed characteristics and the operating conditions to obtain small inhalable drug particles (1–5 µm) [67, 68]. Therefore, it is a method of choice for producing both carrier-free dry powder formulations as well as powder formulations composed of engineered particles such as nano-in-micro formulations and porous particles.
Animal studies of inhaled rifampicin
Studies in animals have been conducted to evaluate the alveolar macrophage uptake, safety and tolerability, toxicity, systemic exposure, pharmacokinetics and bio-distribution, and assessment of antibacterial efficacy after inhaled delivery of rifampicin (Table 3). In vivo studies have mostly used small animals such as mice, rats, and guinea pigs. The dose of delivered drug varies from one animal study to another, but it has consistently been found that inhaled delivery of anti-TB drugs was superior to the oral or intravenous route in achieving higher uptake by alveolar macrophages, higher systemic drug concentrations, and better antibacterial efficacy with minimal or no toxicity to the local lung tissues (Table 3).
Evaluation of the safety and toxicity of high-dose inhaled rifampicin is necessary because a higher dose may result in an increased risk of toxicity to the lung due to potential drug accumulation in the lungs [69]. Rifampicin has proven its potential for inhaled anti-TB therapy, with its safety demonstrated after the pulmonary administration of different types of formulations in laboratory animals [25, 54, 70]. Results from small animal studies have reported that high rifampicin concentrations can be achieved in the lungs (65% dose recovered from the lungs 1 h after administration) as well as in the systemic circulation (AUC0-24 of 131 ± 5 µg/mL/h) when delivered via the pulmonary route, further supporting the potential of inhaled rifampicin in TB treatment [71]. Up to 30 and 65% aerosolized rifampicin dose was recovered within 30 min in the serum and the lung, respectively, in a rat study in which various formulations were administered by intra-tracheal delivery [63]. The lack of studies in large animals is due to the challenges associated with experimental complexities, cost, and biosafety requirements. Moreover, most of the anti-TB drugs are well-known compounds rather than new drugs and have well-documented safety and efficacy profiles in humans, albeit via the oral route.
Several studies on inhaled rifampicin have investigated its pulmonary pharmacokinetics, tissue distribution, or toxicity after delivery to the lungs of animals (Table 3). The pulmonary pharmacokinetics of rifampicin have been reported for various formulations to evaluate the efficacy of delivering systemic drug concentrations via the pulmonary route. Comparisons between inhaled and oral rifampicin pharmacokinetics in animals suggest that inhaled rifampicin can achieve similar systemic drug concentrations, resulting in similar bioavailability (AUC0-∞ 19.8 ± 7.5 ug·h/mL from inhaled rifampicin compared to AUC0-∞ 27.8 ± 10.6 ug·h/mL from oral rifampicin) to that achieved by orally administered rifampicin but at half of the oral dose [48].
Pandey et al. and Suarez et al. reported better therapeutic effects and higher bioavailability from inhaled rifampicin compared to oral rifampicin, suggesting the superiority of inhaled therapy to oral therapy in TB treatment [27, 72]. In the study by Pandey et al. the AUC0-∞ of rifampicin from the nebulized formulation was 107 ± 8 mg·h/L, about 12.7 times higher than that from oral rifampicin [72]. In the same study, the nebulized rifampicin required only one administration every 10 days for similar efficacy (Log10cfu < 1) in the lung compared to the daily administration required for oral rifampicin. In the study by Suarez et al. nebulized rifampicin microspheres were found to significantly reduce the viable microorganism count (log cfu/mL of 3.8 ± 0.4) in the lung compared to that from the nebulized unformulated rifampicin (log cfu/mL of 4.8 ± 0.1), 4–5 weeks post-infection [27]. However, both studies utilized rifampicin formulations based on poly lactic-co-glycolic acid (PLGA), which were designed for controlled release of the drug and targeted delivery or retention within the lungs. These formulations are not able to deliver high doses of inhaled rifampicin. Moreover, the safety issues specific to formulation excipients or carriers restrict the progress of a formulation to a clinical study. Although a high dose of rifampicin is a requirement for the efficient treatment of TB, studies focusing on a high dose of rifampicin are rare in the literature. Toxicity of inhaled rifampicin to rat lungs was reported previously in two different studies [25, 70]. However, only a low-doses of rifampicin (200 µg and 5 mg/kg) were evaluated in both cases, which have little relevance to clinical conditions because the safety and toxicity evaluation of high-dose inhaled rifampicin in animals is required to determine a safe high-dose for human inhalation during TB treatment. Administration of 5 mg/kg rifampicin via the intra-tracheal route in rats was found to be nontoxic to the liver, as shown by similar serum glutamic-oxalacetic transaminase (SGOT) and serum glutamic-pyruvic transaminase (SGPT) levels before (SGOT: 10.5 ± 0.2 unit/L; SGPT: 62.4 ± 0.9 unit/L) and 240 min after the administration (SGOT: 11.4 ± 0.2 unit/L; SGPT: 72.5 ± 0.8 unit/L), contrary to the significant increase in the enzyme levels observed after the oral administration of rifampicin [25]. Administration of chitosan microparticles equivalent to 200 µg rifampicin by intratracheal instillation resulted only in mild changes in the lung histopathology compared to the severe peribronchiolar infiltration of the inflammatory cells and septal thickening resulted by administration of free unformulated rifampicin at the same dose and route of administration [70]. Another study reported the use of a high-dose (314 mg/kg) rifampicin in rats, in which a passive lung dosing method was adopted for single-dose administration, and the actual amount of drug deposited in the rat lungs was not reported [54]. Nevertheless, the study reported only mild changes in the lung histopathology, suggesting the absence of toxicity after the administration of rifampicin as an inhaled dry powder in rats. In bioavailability studies [48, 73], low-dose rifampicin (5–20 mg/kg) was administered via the inhaled route to rats and guinea pigs, which demonstrated up to two-fold higher bioavailability of rifampicin compared to that from the oral route. In the guinea pig study, administration of 20 mg/kg rifampicin as porous particles via the inhaled route led to a relative bioavailability of 0.87 ± 0.33 compared to the intravenous rifampicin (10 mg/kg), while administration of the same via the oral route at 40 mg/kg dose achieved a relative bioavailability of only 0.61 ± 0.23 [48]. In the rat study, intratracheal administration of 5 mg/kg rifampicin powder formulation was reported to achieve AUC0-∞ of 211 μg·h/mL, while the reported AUC0-∞ achieved by oral administration of the free drug was 12.0 μg·h/mL. Despite the studies discussed above, the pharmacokinetics of rifampicin after inhaled administration of higher doses (> 20 mg/kg) remained unknown.
These observations suggested a gap in the literature regarding the formulation and animal studies related to inhaled high-dose rifampicin. To address these gaps, a recent study was conducted in laboratory rats to evaluate the safety and pharmacokinetics of high-dose inhaled rifampicin in which the animal doses were determined based on allometric scaling from the estimated human inhalation doses. It was observed that repeated intra-tracheal administration of high-dose rifampicin (up to 50 mg/kg) powder formulations were safe to rat lungs and liver, and intra-tracheal rifampicin decreased the drug burden on the liver compared to oral rifampicin, as suggested by no rise in the serum alanine transaminase (ALT) activity in repeated dose 25 mg/kg and single dose 50 mg/kg intratracheal administration groups compared to the nontreated control group [74]. Similarly, rifampicin powder formulations delivered via the inhaled route resulted in significantly higher systemic bioavailabilities of rifampicin (193.1 ± 37.9 and 126.3 ± 20.3 μg·h/mL) compared to that from oral rifampicin (87.4 ± 64.7 and 71.5 ± 11.0 μg·h/mL) at the same dose after a single administration of 50 mg/kg rifampicin for each formulation, respectively [47].
The following sections will discuss the key considerations for the formulation requirement and preclinical development of inhaled rifampicin for TB treatment. The research gap in the preclinical development of inhalable formulations for TB, along with their challenges, will be discussed.
Considerations for designing preclinical studies on inhaled rifampicin
Selection of animal models for preclinical assessment
The selection of appropriate animal species in the preclinical evaluation of inhaled formulations is affected by a number of key factors, such as the anatomical and physiological differences between the animal respiratory system to that of humans, the pathological differences in TB infection between humans and animals, ethical considerations, and financial and zootechnical issues [75]. Moreover, an important difference to animal studies involving drug administration through other routes of administration is that the formulations for pulmonary delivery need to be aerosolized before being administered into the animal body [76]. Therefore, the selection of animal models in pulmonary testing is often driven by the exposure technology available for drug delivery. The animal models used to evaluate formulations for respiratory delivery can mainly be categorized into two groups: small rodents, including mice, rats, guinea pigs, and large mammals, which include rabbits, dogs, sheep, and monkeys [77].
Small animals such as rodents are relatively easier to handle, require small quantities of test material for pulmonary dosing, and more numbers are available for a lower cost and in small-scale facilities. Rodents are more suitable for lung deposition and safety studies of inhaled formulations and have been used for evaluating inhaled pharmacokinetics of drugs. However, due to anatomical differences in their respiratory system compared to that of humans, the results obtained from a rodent study face challenges of extrapolation to human results [78]. Mice, rats, and guinea pigs are all obligatory nose breathers [78], and thus, it is necessary to administer drug formulations directly to their lungs by intra-tracheal administration to assess drug products that are intended to be orally inhaled by humans. The intra-tracheal delivery technique, however, has a limitation in the testing of anti-TB drug formulations or TB disease models due to the requirement of multiple administration of the antibiotic that may last for several weeks and increases the risk of tracheal irritation as well as effects on animal welfare resulting from multiple anesthesia [79]. Long-term testing of anti-TB inhaled formulations in small animals is not feasible, and they are best suited for lung deposition and safety studies and a short-term evaluation of inhaled pharmacokinetics or efficacy after intra-tracheal delivery of anti-TB drugs. For pharmacokinetic studies, repeated blood sampling is necessary, which requires blood vessel cannulation to be performed in animals. The maximum blood volume that can be collected per sample in blood vessel cannulated animals is 0.02 mL, 0.2 mL, and 0.5 mL for mice, rats, and guinea pigs, respectively (Table 4) [80,81,82]. Considering the volume of samples that can be collected, the use of rats and guinea pigs is more feasible for pharmacokinetic studies in which repeated blood sampling is usually required.
Large animals for pulmonary drug delivery testing include rabbits, dogs, sheep, and monkeys, which are used for complex or long-term studies when the establishment of preclinical data is necessary with large doses and lengthy regimens, and analysis is performed on a large volume of biological samples [83]. They tend to have similar respiratory anatomy to humans compared to the smaller animals, although the data obtained from the large animal species still cannot be completely extrapolated to humans due to the influence of laboratory procedures and environments on experimental results, the lack of congruence between human diseases and animal models of diseases, and the interspecies differences in physiology and genetics [84]. Testing of new inhalable formulations and devices in larger species may be required to fulfill regulatory needs when the safety or toxicity of such new entities need to be investigated in at least two different species, one of them being a non-rodent [85]. Due to the technical complexity and requirement of dedicated laboratory facilities, conducting preclinical testing in large animal species is difficult, and as a result, studies reported on in vivo testing of inhaled anti-TB drug formulations in larger species are rare. Nevertheless, preclinical testing of inhaled formulations of drugs of different classes has been conducted in mammals, and the use of rabbits [86, 87], beagle dogs [88,89,90], and baboons [91] have also been reported.
For preclinical testing of inhaled rifampicin formulations, small animal models are more suitable for lung deposition and safety studies. Since rifampicin has been in clinical use as an anti-TB treatment for several decades and has well-documented systemic safety for its current oral delivery, respiratory tract safety is the primary concern for inhaled delivery. The animal studies are therefore aimed at studying the safety and toxicity to local lung tissues after inhaled delivery. Assessment of the safety of inhaled rifampicin formulations to lung tissues can be conducted in small animals such as mice, rats, and guinea pigs, which allows drug distribution, alveolar macrophage uptake, and pharmacokinetic studies to be conducted.
Anatomical and physiological differences between human and animal respiratory system
The respiratory differences between humans and laboratory animals in terms of both anatomical layout as well as their physiological parameters have been well discussed in the literature [75, 78, 83, 92,93,94]. The key differences in the respiratory anatomy and physiology of mammalian species used for preclinical testing in pulmonary drug delivery in comparison to humans are summarized in Table 5.
The mammalian respiratory tract is known to vary from species to species, with obvious differences in the nasal anatomy and significant differences in the tracheobronchial tree and the lower respiratory system, which affect aerosol deposition, clearance, as well as absorption after inhalation of drug particles [95]. The breathing pattern is also different between species, with rodents (mice, rats, and guinea pigs) and rabbits able to breathe only through their nose, while humans, monkeys, and dogs are capable of breathing through both nose and mouth [93]. This anatomical difference is more likely to be relevant when aerosol delivery to animals involves whole-body exposure or nose-only exposure in which the aerosolized particles need to enter the animal lung via the nasal opening. Anatomical differences further down the respiratory tract include differences in airway branching, lung weight and symmetry, tracheal and tracheobronchial length and diameters, lung volume, the number and size of alveoli, and number of alveolar macrophages [78]. Similarly, the physiological differences between species include the differences in their respiratory rates, tidal volumes, and ventilation rates [93].
Consideration of the differences in respiratory anatomy and physiology between the selected animal model and humans is important for inhaled drug and formulations. This allows allometric conversion of the effect observed and responses recorded in animals into that for humans.
Methods for pulmonary drug administration to animals
The complexity of the respiratory system makes it challenging to deliver drug aerosols efficiently to the desired site in the respiratory tract of laboratory animals. In most cases, anesthetizing or restraining the animals is inevitable, which may alter the normal respiratory phenomena or induce stress in animals. Nevertheless, the development of state-of-the-art tools and techniques has allowed the precise delivery of drug aerosols to laboratory animals with minimized risk while maintaining animal welfare to the best possible standard.
The pulmonary drug delivery technique to animals depends on the nature of the study and its goals. The delivery techniques are categorized into either direct or passive techniques depending on whether aerosol is administered to the animal or the animal inhales the aerosol by breathing, respectively [78, 92]. Direct delivery techniques are suitable when a predetermined amount of drug is to be delivered to the lungs, while passive techniques are preferable to study the aerodynamics of drug particles in the respiratory tract together with the drug effects after deposition.
Direct pulmonary administration
Direct administration is often carried out under anesthesia. This technique allows the delivery of desired doses of drugs with minimum loss while being accurately measured. The common methods of direct aerosol administration in animals are liquid instillation, liquid spray instillation, and dry powder insufflation to deliver a liquid bolus, liquid spray, and powder aerosol, respectively. The delivery devices commonly employed for direct aerosol delivery to small animals are known as sprayers or insufflators (Fig. 1). Recently, a Venturi-effect device for direct delivery of aerosolized anti-TB inhalable powders for uniform lung distribution was reported by Hirota et al. in which the Venturi-effect administration showed three times higher drug administration compared to a conventional insufflator [96]. The animals need to be anesthetized and laid in a supine position on a suitable platform to visualize the vocal cords and the trachea with the help of a laryngoscope [93]. The tip of the aerosol device is then inserted into the trachea up to the carina, and the device is actuated to release the aerosol. A small animal laryngoscope (manufactured by Harvard Apparatus, Holliston, MA, USA) and a prototype rat intubation platform, designed in-house, are shown in Fig. 2. Direct administration of drug formulations to the airways of small animals can also be undertaken in anesthetized, tracheotomized, and mechanically ventilated animals using a purpose-built jet nebulizer or a dry powder delivery apparatus [97]. For direct aerosol delivery to larger animals, anesthetic and surgical procedures are often required [92]. Delivery of liquid drug formulations and powder aerosols to large animals is performed with dedicated sprayers and dry powder inhaler devices, respectively, via oro-tracheal intubation [75].
The use of direct delivery methods in animals is invasive and requires the use of anesthesia and surgical procedures. This can cause irritation to the animal respiratory tract and induces stress in animals. Therefore, repeated or long-term pulmonary administration of drug formulations by this method is challenging.
Passive pulmonary administration
Passive administration of aerosols to animals is undertaken by exposing them to an aerosol chamber where both conscious and unconscious animals can breathe in the aerosolized drugs following either a whole-body, head-only, nose-only, or facemask exposure (Fig. 3). The limitations of this method include variation in dose delivered to the respiratory tract, a requirement of large doses to compensate for drug loss, the possibility of drug absorption by cutaneous or oral routes, the requirement of an aerosol generator, and size limitations of exposure chamber for larger animals [78, 92].
After whole body exposure, the dose delivered to the animal can be calculated using Eq. 1, where D refers to the delivered dose (mg/kg), C is the drug concentration in air (mg/L), RMV is the respiratory minute volume of the animal (L/min), T is the duration of exposure (min), RF is the respirable fraction of the formulation, and BW is the weight of the animal (kg) [83, 98].
Passive pulmonary administration of drug formulations to large mammals can be carried out via face mask inhalation, but this technique also has limitations due to the requirement of animal training and difficulties in predicting lung deposition and thus can be used only for lung tolerance or regulatory toxicity assessment for long-term studies [75].
Between the two different delivery techniques, direct aerosol delivery is preferable in testing inhaled powder or liquid formulations in animals since it allows precise delivery of higher doses to animal lungs, which is a key requirement for rifampicin. The passive delivery technique is not suitable for studies on high-dose rifampicin formulations since the animals are not able to inhale large doses of aerosolized drug particles on their own [93]. Toxicity evaluation of low rifampicin doses, on the other hand, will not be relevant for extrapolation to human responses, which is expected to involve the use of high inhaled rifampicin doses for TB treatment.
Assessment of the in vivo properties of the aerosol
Assessment of in vivo properties of drug formulations delivered via the pulmonary route mainly includes an assessment of the particle deposition, safety, and toxicity to the lung and other organs, absorption and disposition after pulmonary delivery, or the efficacy after inhaled therapy, often known as the endpoints of a preclinical study in animals (Fig. 4).
The drug deposited after pulmonary delivery to laboratory animals can be determined either by imaging techniques such as gamma scintigraphy, single-photon emission computed tomography (SPECT), and positron emission tomography (PET) or by bronchoalveolar lavage (BAL) procedure [83]. For imaging techniques, the drug particles often need to be conjugated with radioligands to be imaged. The BAL procedure is an invasive method in which normal saline is injected into the lung, followed by its aspiration to collect deposited drug along with cells, soluble proteins, lipids, and other chemical constituents from the epithelial region [99]. The procedures to measure drug deposition after pulmonary delivery should be performed early enough to assure accurate measurement of the deposited drug before subsequent drug absorption, clearance, or metabolism [83].
The endpoint of safety and toxicity evaluation of inhaled drug formulation in animals is histopathology, which includes histopathologic examination of lung tissues and a range of organs for evaluation of local and systemic toxicities, respectively [100]. Similarly, markers of pulmonary inflammation such as lactate dehydrogenase activity for tissue injury, protein levels for permeability alteration and inflammatory cytokines such as TNF-α can be examined from BAL fluid to assess the safety of the inhaled formulations [93].
The measurement of drug absorption and disposition after pulmonary delivery in animals is possible by measurement of drug concentration in the plasma as well as the tissues. The concentration in the tissues provides a tissue distribution profile of the drug after pulmonary delivery. Absorption and elimination profiles of the drug in plasma after inhaled delivery can be studied by pharmacokinetic evaluation, which is essential for drugs intended for systemic action, as well as in bioequivalence studies for comparison between two inhaled formulations. The pharmacokinetic evaluation permits an understanding of the fate of the drug from its inhaled formulation in terms of its area under the plasma concentration–time curve (AUC), the maximum plasma concentration (Cmax), the time to Cmax (Tmax), absorption and elimination rates, and bioavailability of the inhaled drug. Estimation of pharmacokinetics after pulmonary administration of a drug formulation is often carried out using compartmental, non-compartmental, and physiologically based pharmacokinetic (PBPK) models [83]. A compartmental model assumes a system composed of a group of arbitrarily sized compartments, where one of them is assigned as the central compartment to which absorption of the drug occurs and is also the site from which the drug gets excreted. The non-compartmental approach is simpler and usually followed when data are limited to the drug concentration versus time in a single compartment. Similarly, the PBPK model consists of multiple compartments in which each compartment is a defined space within the body and considers the anatomy, physiology, direction of blood flow, rates of drug extraction, or metabolism relevant to the actual physiological conditions for each compartment.
Among the key assessments of in vivo properties of inhaled drugs in animals, the efficacy assessment is the most important. While endpoints for assessment of efficacy are different based on the type of the disease, the efficacy endpoint for TB is either the reduction in bacterial burden or complete eradication of the bacteria. Comparison of efficacy between inhaled and orally administered anti-TB drugs in an animal model is rare in the literature, possibly due to complexity in the experimental requirements as well as the pathogenesis of the disease itself. TB pathogenesis is known to be different in humans and animals, with distinct pathological features such as primary and secondary granuloma formation, cavity formation with necrosis, and caseation being prominent in humans while absent in some laboratory animals [76]. This makes the extrapolation of animal efficacy results in humans more difficult. Nevertheless, assessment of the efficacy of inhaled drugs against TB in animal models is necessary to strengthen the hypothesis that pulmonary delivery of anti-TB drugs is potentially effective for TB treatment. This can be best achieved by selecting the animal model with the highest similarity to humans in terms of TB pathogenesis. For this purpose, guinea pigs have been widely used as the model of choice because their TB infection features human-like pathological changes such as granulomas with central necrosis within the immune cells and a fibrotic capsule [101].
Selection of the right inhaled formulation for in vivo testing
The formulations used for testing in laboratory animals via the pulmonary route include dry powder and liquid formulations for nebulization intended for human use. Animal experiments do not take into account the effect of inhaler devices that are used in clinical practice. This might have an effect on the assessment of drug deposition patterns in the respiratory tract of animals. However, this does not hinder the studies intended for the evaluation of safety, pharmacokinetics, drug distribution, or efficacy. The formulations are therefore designed with the intended features for clinical use in humans regardless of the technical difficulties in animal studies. Due to this reason, drugs can sometimes be administered in liquid form to animals for safety evaluation, even though the intended formulation for human use may be a dry powder inhaler [25, 70].
Dose calculation for in vivo testing
In the case of passive pulmonary administration of aerosolized formulations to animals, the dose delivered to the airways mainly depends on the factors associated with Eq. 1. Among all the variables, the respiratory minute volume (RMV) is the animal-dependent variable that affects the dose of the aerosol deposited. RMV is calculated using an allometric equation based on body weight (BW in kg) (Eq. 2) [98].
For studies dealing with direct pulmonary administration, the selection of inhaled dose varies based on the study design and the experimental feasibility. As a result, there is a lack of consistency in the dose of a specific drug delivered to the animal lungs in the reported studies (Table 3). While there are no criteria developed for the calculation of inhaled doses to be delivered in animal studies, the current approach is based on individual experimental design and feasibility. The maximum dose and duration in toxicological studies via the pulmonary route may be set depending on the anatomy and physiology of the animal respiratory system. However, one approach to determine the prospective animal inhaled doses is to consider the allometric scaling for dose conversion. Allometric scaling is based on the normalization of dose-to-body surface area and is often used for dose extrapolation in determining maximum starting human doses for clinical studies. This has been established as a guidance by the USFDA for dose conversion between humans and animals to calculate equivalent human or animal doses [102]. Based on the allometric scaling, the human equivalent dose (HED) can be calculated as shown in Eq. 3.
The exponent for body surface area (0.67) in Eq. 3 accounts for the difference in metabolic rates and body surface area between animals and humans. Usually, the animal dose in Eq. 3 is the no observed adverse effect level (NOAEL) in animals determined from the toxicological study to calculate HED. The maximum starting dose in humans is usually one-tenth of the HED calculated based on NOAEL.
For determining animal inhaled doses in preclinical studies, the animal doses can be determined from Eq. 3 by estimating the expected dose in humans. This approach can be helpful in preclinical safety and toxicity studies of inhaled drugs, in which the safety or toxicity profile of the relevant animal dose needs to be established with respect to the estimated human dose. For pharmacokinetics and efficacy studies of inhaled drugs, using a relevant dose in animals can be helpful in establishing reliable preclinical data for future clinical studies.
Regulatory considerations on the development of inhaled formulations
The guidelines for manufacturing and quality control of inhalation drug products have been developed by the FDA and the European Medicines Agency (EMA) for metered dose inhalers, dry powder inhalers, as well as solutions and suspensions for nebulization [103,104,105]. The preclinical studies on the inhaled formulations, however, need to establish evidence of safety and efficacy in animal models through the pulmonary route. The safety assessment for a new drug requires studies in two different species, including rodent and a non-rodent model [85]. Development of inhalable formulations against TB usually includes an approved anti-TB drug, conventionally administered via the oral or parenteral routes. However, when an approved drug is to be administered via a different route other than its previously approved route of administration, it is generally considered a new drug entity by the regulatory bodies. Therefore, preclinical development of inhaled formulations of currently used anti-TB drugs may face the requirement of rigorous safety and toxicity studies in preclinical models, similar to that for new drugs under development.
A decision-making process known as regulatory nonclinical safety evaluations (RNSE) is used by regulators to assess if an inhaled drug product can be safely used in humans based on the available non-clinical data on the drug product [98]. RNSE determines whether the safety margins of the inhaled drug product are acceptable (both the inhaler device and the formulation) for its intended clinical doses and is a complex decision-making process that takes into account the formulation features, the inhaler device characteristics, and the results from inhaled toxicology study in animals.
The research gaps hindering the transition of inhaled rifampicin from preclinical development to clinical investigation
Studies evaluating inhaled formulations in animals require consideration of several factors, including animal species, drug formulation, inhaled delivery technique, the dose of the drug to be tested, and the parameters of interest together with their endpoints based on the intended clinical use. Only a few studies have reported the testing of a high dose of anti-TB drugs in animals, which is an important consideration in designing, developing, and testing inhaled formulations for TB [106]. Similarly, the inhaled doses of rifampicin reported in the animal studies are not determined based on the intended clinical dose for humans, as no allometric conversion of human and animal doses was reported. On the other hand, some of the studies have not reported the technique of inhaled drug administration to animals [107,108,109] or the dose of inhaled drug [110]. Thus, a research gap between the preclinical testing and the clinical evaluation exists for inhaled rifampicin since the majority of the formulation and animal studies reported in the literature have not considered the requirement of the high dose and the importance of animal dose selection. It is important to plan and conduct in vivo studies on inhaled rifampicin in such a way that the findings in animals can be utilized as the basis for further clinical evaluation.
Apart from the case of inhaled rifampicin, the gap between the large number of studies related to formulation development and the much smaller number of clinical studies involving inhaled anti-TB drugs has left several questions unanswered for inhaled anti-TB therapy. One reason for the few clinical studies on inhaled anti-TB formulations might be the lack of enough in vivo preclinical data on the safety, pharmacokinetics, and efficacy of inhaled anti-TB formulations. Nevertheless, a number of antibacterial drugs, such as ciprofloxacin, tobramycin, and colistimethate have been found to be safe and effective in treating respiratory infections in patients when delivered by inhalation [111]. Such evidence from clinical studies provides confidence for further preclinical and clinical evaluations of inhaled anti-TB drugs in the future. Preclinical evaluations of inhaled anti-TB formulations are necessary to answer key questions regarding the safety, pharmacokinetics, and efficacy of anti-TB drugs delivered via the inhalation route. The findings from such studies can inform clinical studies on the expected dose, the expected tissue and blood concentration of the drugs after inhaled administration, and clinical efficacy compared to the current anti-TB regimen.
Conclusions
Among the anti-TB drugs investigated for their potential inhaled delivery, rifampicin has been extensively investigated as micro-particles, liposomes, solid lipid nanoparticles, polymeric nanoparticles, porous particles, nanoaggregates, and nanocomposites for delivery via the inhalation route. Some of these formulations of rifampicin were also studied in animals to evaluate in vivo properties of the drug after inhaled delivery. However, none of the formulations have progressed to clinical trials.
A lack of well-designed preclinical studies has created a gap between formulation studies and clinical trials. A comparison of inhaled versus oral or intravenously administered drugs needs to be performed in preclinical models at a dose relevant to the intended future human dose. And there must be a careful evaluation of local lung toxicity and pharmacokinetics in comparison to the conventional route of administration for the drug. If this bottleneck in the drug pipeline can be addressed, the promise of inhaled drug formulations for anti-TB therapy may be fulfilled.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Materials availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
WHO. Global tuberculosis report. World Heal Organ. 2021.
WHO. Guidelines for treatment of drug-susceptible tuberculosis and patient care. World Heal Organ. 2017.
WHO. WHO consolidated guidelines on drug-resistant tuberculosis treatment. World Heal Organ. 2019.
Grüber G. Introduction: Novel insights into TB research and drug discovery. Prog Biophys Mol Biol Elsevier. 2020;152:2–5.
Dartois V. The path of anti-tuberculosis drugs: from blood to lesions to mycobacterial cells. Nat Rev Microbiol Nature Publishing Group. 2014;12:159–67.
Pandey R, Khuller GK. Antitubercular inhaled therapy: opportunities, progress and challenges. J Antimicrob Chemother Oxford University Press. 2005;55:430–5.
Seung KJ, Keshavjee S, Rich ML. Multidrug-resistant tuberculosis and extensively drug-resistant tuberculosis. Cold Spring Harb Perspect Med. Cold Spring Harbor Laboratory Press. 2015;5:a017863
Muttil P, Wang C, Hickey AJ. Inhaled drug delivery for tuberculosis therapy. Pharm Res Springer. 2009;26:2401–16.
Libardo MDJ, Boshoff HIM, Barry CE III. The present state of the tuberculosis drug development pipeline. Curr Opin Pharmacol Elsevier. 2018;42:81–94.
Tiberi S, du Plessis N, Walzl G, Vjecha MJ, Rao M, Ntoumi F, et al. Tuberculosis: progress and advances in development of new drugs, treatment regimens, and host-directed therapies. Lancet Infect Dis Elsevier. 2018;18:e183–98.
Braunstein M, Hickey AJ, Ekins S. Why wait? The case for treating tuberculosis with inhaled drugs. Pharm Res Springer. 2019;36:1–6.
Hickey AJ, Misra A, Fourie PB. Dry powder antibiotic aerosol product development: inhaled therapy for tuberculosis. J Pharm Sci Elsevier. 2013;102:3900–7.
Brunaugh AD, Smyth HDC, Williams III RO. Pulmonary drug delivery. Essent Pharm. Springer. 2019;163–81.
Palmer LB, Smaldone GC. Reduction of bacterial resistance with inhaled antibiotics in the intensive care unit. Am J Respir Crit Care Med Am Thoracic Soc. 2014;189:1225–33.
Palmer LB, Smaldone GC. Inhaled antibiotics, pneumonia and bacterial resistance in the ICU. Am J Respir Crit Care Med Eur Respiratory Soc. 2018;189:1225–33.
Wang X, Xie Z, Zhao J, Zhu Z, Yang C, Liu Y. Prospects of inhaled phage therapy for combatting pulmonary infections. Front Cell Infect Microbiol. 2021;1028.
Singh R, Dwivedi SP, Gaharwar US, Meena R, Rajamani P, Prasad T. Recent updates on drug resistance in mycobacterium tuberculosis. J Appl Microbiol Wiley Online Library. 2020;128:1547–67.
Rossi I, Bettini R, Buttini F. Resistant tuberculosis: the latest advancements of second-line antibiotic inhalation products. Curr Pharm Des Bentham Science Publishers. 2021;27:1436–52.
Sharma A, Vaghasiya K, Gupta P, Gupta UD, Verma RK. Reclaiming hijacked phagosomes: hybrid nano-in-micro encapsulated MIAP peptide ensures host directed therapy by specifically augmenting phagosome-maturation and apoptosis in TB infected macrophage cells. Int J Pharm Elsevier. 2018;536:50–62.
Das S, Tucker I, Stewart P. Inhaled dry powder formulations for treating tuberculosis. Curr Drug Deliv Bentham Science Publishers. 2015;12:26–39.
Crompton GK. Dry powder inhalers: advantages and limitations. J Aerosol Med. 199;4:151–6.
Ashurst I, Malton A, Prime D, Sumby B. Latest advances in the development of dry powder inhalers. Pharm Sci Technolo Today Elsevier. 2000;3:246–56.
Pham D-D, Fattal E, Tsapis N. Pulmonary drug delivery systems for tuberculosis treatment. Int J Pharm Elsevier. 2015;478:517–29.
Srichana T, Ratanajamit C, Juthong S, Suwandecha T, Laohapojanart N, Pungrassami P, et al. Evaluation of proinflammatory cytokines and adverse events in healthy volunteers upon inhalation of antituberculosis drugs. Biol Pharm Bull The Pharm Soc Japan. 2016;39:1815–22.
Parikh R, Patel L, Dalwadi S. Microparticles of rifampicin: comparison of pulmonary route with oral route for drug uptake by alveolar macrophages, phagocytosis activity and toxicity study in albino rats. Drug Deliv Taylor & Francis. 2014;21:406–11.
Parikh R, Dalwadi S, Aboti P, Patel L. Inhaled microparticles of antitubercular antibiotic for in vitro and in vivo alveolar macrophage targeting and activation of phagocytosis. J Antibiot (Tokyo). Nature Publishing Group. 2014;67:387–94.
Suarez S, O’Hara P, Kazantseva M, Newcomer CE, Hopfer R, McMurray DN, et al. Respirable PLGA microspheres containing rifampicin for the treatment of tuberculosis: screening in an infectious disease model. Pharm Res Springer. 2001;18:1315–9.
Pham D-D, Grégoire N, Couet W, Gueutin C, Fattal E, Tsapis N. Pulmonary delivery of pyrazinamide-loaded large porous particles. Eur J Pharm Biopharm Elsevier. 2015;94:241–50.
Patil-Gadhe AA, Kyadarkunte AY, Pereira M, Jejurikar G, Patole MS, Risbud A, et al. Rifapentine-proliposomes for inhalation: in vitro and in vivo toxicity. Toxicol Int. Wolters Kluwer--Medknow Publications. 2014;21:275.
Schoubben A, Blasi P, Marenzoni ML, Barberini L, Giovagnoli S, Cirotto C, et al. Capreomycin supergenerics for pulmonary tuberculosis treatment: preparation, in vitro, and in vivo characterization. Eur J Pharm Biopharm Elsevier. 2013;83:388–95.
Hwang SM, Kim DD, Chung SJ, Shim CK. Delivery of ofloxacin to the lung and alveolar macrophages via hyaluronan microspheres for the treatment of tuberculosis. J Control Release Elsevier. 2008;129:100–6.
Sung JC, Garcia-Contreras L, VerBerkmoes JL, Peloquin CA, Elbert KJ, Hickey AJ, et al. Dry powder nitroimidazopyran antibiotic PA-824 aerosol for inhalation. Antimicrob Agents Chemother Am Soc Microbiol. 2009;53:1338–43.
Garcia-Contreras L, Sung JC, Muttil P, Padilla D, Telko M, VerBerkmoes JL, et al. Dry powder PA-824 aerosols for treatment of tuberculosis in guinea pigs. Antimicrob Agents Chemother Am Soc Microbiol. 2010;54:1436–42.
Stewart IE, Lukka PB, Liu J, Meibohm B, Gonzalez-Juarrero M, Braunstein MS, et al. Development and characterization of a dry powder formulation for anti-tuberculosis drug spectinamide 1599. Pharm Res Springer. 2019;36:1–13.
O’Connor G, Krishnan N, Fagan-Murphy A, Cassidy J, O’Leary S, Robertson BD, et al. Inhalable poly (lactic-co-glycolic acid)(PLGA) microparticles encapsulating all-trans-Retinoic acid (ATRA) as a host-directed, adjunctive treatment for Mycobacterium tuberculosis infection. Eur J Pharm Biopharm Elsevier. 2019;134:153–65.
Tsapis N, Bennett D, O’driscoll K, Shea K, Lipp MM, Fu K, et al. Direct lung delivery of para-aminosalicylic acid by aerosol particles. Tuberculosis. Elsevier. 2003;83:379–85.
Dharmadhikari AS, Kabadi M, Gerety B, Hickey AJ, Fourie PB, Nardell E. Phase I, single-dose, dose-escalating study of inhaled dry powder capreomycin: a new approach to therapy of drug-resistant tuberculosis. Antimicrob Agents Chemother Am Soc Microbiol. 2013;57:2613–9.
Aronson JK. Rifamycins. Meyler’s Side Eff Drugs Sixteenth Ed. 2016;132–70.
Scholar E. Rifampin. In: Enna SJ, Bylund DBBTTCPR, editors. xPharm Compr Pharmacol Ref [Internet]. New York: Elsevier. 2007;1–8. Available from: https://www.sciencedirect.com/science/article/pii/B9780080552323625346.
Van Ingen J, Aarnoutse RE, Donald PR, Diacon AH, Dawson R, Plemper van Balen G, et al. Why do we use 600 mg of rifampicin in tuberculosis treatment? Clin Infect Dis. Oxford University Press. 2011;52:e194–9.
Seijger C, Hoefsloot W, Bergsma-de Guchteneire I, Te Brake L, van Ingen J, Kuipers S, et al. High-dose rifampicin in tuberculosis: experiences from a Dutch tuberculosis centre. PLoS One. Public Library of Science San Francisco, CA USA. 2019;14:e0213718.
Boeree MJ, Diacon AH, Dawson R, Narunsky K, Du Bois J, Venter A, et al. A dose-ranging trial to optimize the dose of rifampin in the treatment of tuberculosis. Am J Respir Crit Care Med Am Thor Soc. 2015;191:1058–65.
Aarnoutse RE, Kibiki GS, Reither K, Semvua HH, Haraka F, Mtabho CM, et al. Pharmacokinetics, tolerability, and bacteriological response of rifampin administered at 600, 900, and 1,200 milligrams daily in patients with pulmonary tuberculosis. Antimicrob Agents Chemother Am Soc Microbiol. 2017;61:e01054-e1117.
Ruslami R, Nijland HMJ, Alisjahbana B, Parwati I, van Crevel R, Aarnoutse RE. Pharmacokinetics and tolerability of a higher rifampin dose versus the standard dose in pulmonary tuberculosis patients. Antimicrob Agents Chemother Am Soc Microbiol. 2007;51:2546–51.
Peloquin CA, Velásquez GE, Lecca L, Calderón RI, Coit J, Milstein M, et al. Pharmacokinetic evidence from the HIRIF trial to support increased doses of rifampin for tuberculosis. Antimicrob Agents Chemother Am Soc Microbiol. 2017;61:e00038-e117.
Grobbelaar M, Louw GE, Sampson SL, van Helden PD, Donald PR, Warren RM. Evolution of rifampicin treatment for tuberculosis. Infect Genet Evol. 2019;74:103937. Available from: https://www.sciencedirect.com/science/article/pii/S1567134819301583.
Khadka P, Sinha S, Tucker IG, Dummer J, Hill PC, Katare R, et al. Pharmacokinetics of rifampicin after repeated intra-tracheal administration of amorphous and crystalline powder formulations to Sprague Dawley rats. Eur J Pharm Biopharm Elsevier. 2021;162:1–11.
Garcia Contreras L, Sung J, Ibrahim M, Elbert K, Edwards D, Hickey A. Pharmacokinetics of inhaled rifampicin porous particles for tuberculosis treatment: insight into rifampicin absorption from the lungs of guinea pigs. Mol Pharm ACS Publications. 2015;12:2642–50.
Nainwal N, Sharma Y, Jakhmola V. Dry powder inhalers of antitubercular drugs. Tuberculosis. 2022;135:102228. Available from: https://www.sciencedirect.com/science/article/pii/S1472979222000658.
Parumasivam T, Chang RYK, Abdelghany S, Ye TT, Britton WJ, Chan H-K. Dry powder inhalable formulations for anti-tubercular therapy. Adv Drug Deliv Rev Elsevier. 2016;102:83–101.
Mehanna MM, Mohyeldin SM, Elgindy NA. Respirable nanocarriers as a promising strategy for antitubercular drug delivery. J Control Release. 2014;187:183–97. Available from: https://www.sciencedirect.com/science/article/pii/S0168365914003411.
Chae J, Choi Y, Tanaka M, Choi J. Inhalable nanoparticles delivery targeting alveolar macrophages for the treatment of pulmonary tuberculosis. J Biosci Bioeng. 2021;132:543–51. Available from: https://www.sciencedirect.com/science/article/pii/S1389172321002127.
Sethuraman VV, Hickey AJ. Powder properties and their influence on dry powder inhaler delivery of an antitubercular drug. AAPS PharmSciTech Springer. 2002;3:7–16.
Rawal T, Kremer L, Halloum I, Butani S. Dry-powder inhaler formulation of rifampicin: an improved targeted delivery system for alveolar tuberculosis. J Aerosol Med Pulm Drug Deliv. Mary Ann Liebert, Inc. 140 Huguenot Street, 3rd Floor New Rochelle, NY 10801 USA. 2017;30:388–98.
Grotz E, Tateosian NL, Salgueiro J, Bernabeu E, Gonzalez L, Manca ML, et al. Pulmonary delivery of rifampicin-loaded soluplus micelles against mycobacterium tuberculosis. J Drug Deliv Sci Technol. 2019;53:101170. Available from: https://www.sciencedirect.com/science/article/pii/S1773224719305842.
Galdopórpora JM, Martinena C, Bernabeu E, Riedel J, Palmas L, Castangia I, et al. Inhalable mannosylated rifampicin-curcumin co-loaded nanomicelles with enhanced in vitro antimicrobial efficacy for an optimized pulmonary tuberculosis therapy. Pharmaceutics. 2022.
El-Sherbiny IM, El-Baz NM, Yacoub MH. Inhaled nano- and microparticles for drug delivery. Glob Cardiol Sci Pract. 2015;2015:2.
Berkenfeld K, McConville JT, Lamprecht A. Inhalable dry powders of rifampicin highlighting potential and drawbacks in formulation development for experimental tuberculosis aerosol therapy. Expert Opin Drug Deliv. Taylor & Francis 2020;17:305–22. Available from: https://doi.org/10.1080/17425247.2020.1720644.
Joshi M, Prabhakar B. Development of respirable rifampicin loaded bovine serum albumin formulation for the treatment of pulmonary tuberculosis. J Drug Deliv Sci Technol. 2021;61:102197. Available from: https://www.sciencedirect.com/science/article/pii/S1773224720314866.
Mehanna MM, Mohyeldin SM, Elgindy NA. Rifampicin-carbohydrate spray-dried nanocomposite: a futuristic multiparticulate platform for pulmonary delivery. Int J Nanomedicine. Dove Press. 2019;14:9089.
Manca ML, Sinico C, Maccioni AM, Diez O, Fadda AM, Manconi M. Composition influence on pulmonary delivery of rifampicin liposomes. Pharmaceutics MDPI. 2012;4:590–606.
Zaru M, Sinico C, De Logu A, Caddeo C, Lai F, Manca ML, et al. Rifampicin-loaded liposomes for the passive targeting to alveolar macrophages: in vitro and in vivo evaluation. J Liposome Res Taylor & Francis. 2009;19:68–76.
Vyas SP, Kannan ME, Jain S, Mishra V, Singh P. Design of liposomal aerosols for improved delivery of rifampicin to alveolar macrophages. Int J Pharm Elsevier. 2004;269:37–49.
Elhissi A. Liposomes for pulmonary drug delivery: the role of formulation and inhalation device design. Curr Pharm Des Bentham Science Publishers. 2017;23:362–72.
Mindaugas R, Mohammad N, Mohamed Albed A, Abdelbary E. Liposome delivery systems for inhalation: a critical review highlighting formulation issues and anticancer applications. Med Princ Pract. 2016;25:60–72.
Liang Z, Ni R, Zhou J, Mao S. Recent advances in controlled pulmonary drug delivery. Drug Discov Today Elsevier. 2015;20:380–9.
Alhajj N, O’Reilly NJ, Cathcart H. Designing enhanced spray dried particles for inhalation: A review of the impact of excipients and processing parameters on particle properties. Powder Technol. 2021;384:313–31. Available from: https://www.sciencedirect.com/science/article/pii/S0032591021001340.
Momin MAM, Tucker IG, Doyle CS, Denman JA, Das SC. Manipulation of spray-drying conditions to develop dry powder particles with surfaces enriched in hydrophobic material to achieve high aerosolization of a hygroscopic drug. Int J Pharm. 2018;543:318–27. Available from: https://www.sciencedirect.com/science/article/pii/S037851731830214X.
Fröhlich E. Toxicity of orally inhaled drug formulations at the alveolar barrier: parameters for initial biological screening. Drug Deliv Taylor & Francis. 2017;24:891–905.
Pai R V, Jain RR, Bannalikar AS, Menon MD. Development and evaluation of chitosan microparticles based dry powder inhalation formulations of rifampicin and rifabutin. J Aerosol Med Pulm Drug Deliv. Mary Ann Liebert, Inc. 140 Huguenot Street, 3rd Floor New Rochelle, NY 10801 USA. 2016;29:179–95.
Garg T, Goyal AK, Rath G, Murthy RSR. Spray-dried particles as pulmonary delivery system of anti-tubercular drugs: design, optimization, in vitro and in vivo evaluation. Pharm Dev Technol Taylor & Francis. 2016;21:951–60.
Pandey R, Sharma A, Zahoor A, Sharma S, Khuller GK, Prasad B. Poly (DL-lactide-co-glycolide) nanoparticle-based inhalable sustained drug delivery system for experimental tuberculosis. J Antimicrob Chemother Oxford University Press. 2003;52:981–6.
Kundawala A, Patel V, Patel H, Choudhary D. Preparation, in vitro characterization, and in vivo pharmacokinetic evaluation of respirable porous microparticles containing rifampicin. Sci Pharm Austrian Pharmaceutical Society. 2014;82:665–82.
Khadka P, Sinha S, Tucker IG, Dummer J, Hill PC, Katare R, et al. Studies on the safety and the tissue distribution of inhaled high-dose amorphous and crystalline rifampicin in a rat model. Int J Pharm. Elsevier. 2021;597:120345.
Guillon A, Sécher T, Dailey LA, Vecellio L, De Monte M, Si-Tahar M, et al. Insights on animal models to investigate inhalation therapy: relevance for biotherapeutics. Int J Pharm Elsevier. 2018;536:116–26.
Misra A, Hickey AJ, Rossi C, Borchard G, Terada H, Makino K, et al. Inhaled drug therapy for treatment of tuberculosis. Tuberculosis Elsevier. 2011;91:71–81.
Sakagami M. In vivo, in vitro and ex vivo models to assess pulmonary absorption and disposition of inhaled therapeutics for systemic delivery. Adv Drug Deliv Rev Elsevier. 2006;58:1030–60.
Price DN, Kunda NK, Muttil P. Challenges associated with the pulmonary delivery of therapeutic dry powders for preclinical testing. KONA Powder Part J. Hosokawa Powder Technol Foundation. 2019;36:129–44.
Hanif SNM, Garcia-Contreras L. Pharmaceutical aerosols for the treatment and prevention of tuberculosis. Front Cell Infect Microbiol. Frontiers Media SA. 2012;2:118.
Parasuraman S, Raveendran R, Kesavan R. Blood sample collection in small laboratory animals. J Pharmacol Pharmacother. Wolters Kluwer--Medknow Publications. 2010;1:87.
NC3Rs. Blood Sampling. [cited 2022 May 8]. Available from: https://www.nc3rs.org.uk/3rs-resources/blood-sampling.
Wolfensohn S, Lloyd M. Conduct of minor procedures. Handb Lab Anim Manag Welfare, 3rd ed; Wolfensohn, S, Lloyd, M, Eds. 2003;150–181.
Garcia-Contreras L. In vivo animal models for controlled-release pulmonary drug delivery. Control Pulm Drug Deliv. Springer. 2011;443–74.
Akhtar A. The flaws and human harms of animal experimentation. Cambridge Q Healthc Ethics. Cambridge University Press. 2015;24:407–19.
Prior H, Baldrick P, de Haan L, Downes N, Jones K, Mortimer-Cassen E, et al. Reviewing the utility of two species in general toxicology related to drug development. Int J Toxicol. SAGE Publications Sage CA: Los Angeles, CA. 2018;37:121–4.
Fok T-F, Al-Essa M, Monkman S, Dolovich M, Girard L, Coates G, et al. Pulmonary deposition of salbutamol aerosol delivered by metered dose inhaler, jet nebulizer, and ultrasonic nebulizer in mechanically ventilated rabbits. Pediatr Res Nature Publishing Group. 1997;42:721–7.
Meisner D, Pringle J, Mezei M. Liposomal pulmonary drug delivery I. In vivo disposition of atropine base in solution and liposomal form following endotracheal instillation to the rabbit lung. J Microencapsul. Taylor & Francis. 1989;6:379–87.
Surendrakumar K, Martyn GP, Hodgers ECM, Jansen M, Blair JA. Sustained release of insulin from sodium hyaluronate based dry powder formulations after pulmonary delivery to beagle dogs. J Control Release Elsevier. 2003;91:385–94.
Kuehl PJ, Barrett EG, McDonald JD, Rudolph K, Vodak D, Dobry D, et al. Formulation development and in vivo evaluation of a new dry powder formulation of albuterol sulphate in beagle dogs. Pharm Res Springer. 2010;27:894–904.
Niven R, Lynch M, Moutvic R, Gibbs S, Briscoe C, Raff H. Safety and toxicology of cyclosporine in propylene glycol after 9-month aerosol exposure to beagle dogs. J Aerosol Med Pulm Drug Deliv. Mary Ann Liebert, Inc. 140 Huguenot Street, 3rd Floor New Rochelle, NY 10801 USA. 2011;24:205–12.
Gagnadoux F, Leblond V, Vecellio L, Hureaux J, Le Pape A, Boisdron-Celle M, et al. Gemcitabine aerosol: in vitro antitumor activity and deposition imaging for preclinical safety assessment in baboons. Cancer Chemother Pharmacol Springer. 2006;58:237–44.
Cryan S-A, Sivadas N, Garcia-Contreras L. In vivo animal models for drug delivery across the lung mucosal barrier. Adv Drug Deliv Rev Elsevier. 2007;59:1133–51.
Fernandes CA, Vanbever R. Preclinical models for pulmonary drug delivery. Expert Opin Drug Deliv England. 2009;6:1231–45.
Crapo JD, Young SL, Fram EK, Pinkerton KE, Barry BE, Crapo RO. Morphometric characteristics of cells in the alveolar region of mammalian lungs. Am Rev Respir Dis. Am Lung Assoc. 1983;128:S42–6.
Phalen RF, Oldham MJ, Wolff RK. The relevance of animal models for aerosol studies. J Aerosol Med Pulm Drug Deliv. Mary Ann Liebert, Inc. 2 Madison Avenue Larchmont, NY 10538 USA. 2008;21:113–24.
Hirota K, Hirai Y, Nakajima T, Goto S, Makino K, Terada H. Uniformity and efficacy of dry powders delivered to the lungs of a mycobacterial-surrogate rat model of tuberculosis. Pharm Res. 2022;39:143–52.
Underwood SL, Raeburn D. Methods of drug administration to the lung in animals. Airways Smooth Muscle Model Asthmatic Response Vivo. Springer. 1996;27–50.
Tepper JS, Kuehl PJ, Cracknell S, Nikula KJ, Pei L, Blanchard JD. Symposium summary: “breathe in, breathe out, its easy: what you need to know about developing inhaled drugs.” Int J Toxicol. SAGE Publications Sage CA: Los Angeles, CA. 2016;35:376–92.
Van Hoecke L, Job ER, Saelens X, Roose K. Bronchoalveolar lavage of murine lungs to analyze inflammatory cell infiltration. JoVE J Vis Exp. 2017;e55398.
Wolff RK. Toxicology studies for inhaled and nasal delivery. Mol Pharm ACS Publications. 2015;12:2688–96.
Clark S, Hall Y, Williams A. Animal models of tuberculosis: guinea pigs. Cold Spring Harb Perspect Med. 2014;5: a018572.
Administration F, FDA D. Guidance for industry estimating the maximum safe starting dose in initial clinical trials for therapeutics in adult healthy volunteers. July 2005. Pharmacol Toxicol. 2020. Available online https//www.fda.gov/media/72309/download. Accessed 3 Oct 2019.
Inhaler FDAMD. And dry powder inhaler (DPI) products—quality considerations guidance for industry. US Dep Heal Hum Serv Food Drug Adm Cent Drug Eval Res. 2018.
European Medicines Agency. Guideline on the pharmaceutical quality of inhalation and nasal. 2016. Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-pharmaceutical-quality-inhalation-nasal-products_en.pdf.
Spray N, Solution I. Suspension, and spray drug products—chemistry, Manufacturing and Controls Documentation. FDA New Hampshire, MD, USA. 2002.
Muttil P, Kaur J, Kumar K, Yadav AB, Sharma R, Misra A. Inhalable microparticles containing large payload of anti-tuberculosis drugs. Eur J Pharm Sci Elsevier. 2007;32:140–50.
Kaur R, Garg T, Das Gupta U, Gupta P, Rath G, Goyal AK. Preparation and characterization of spray-dried inhalable powders containing nanoaggregates for pulmonary delivery of anti-tubercular drugs. Artif cells, nanomedicine, Biotechnol. Taylor & Francis. 2016;44:182–7.
Kaur R, Garg T, Malik B, Gupta UD, Gupta P, Rath G, et al. Development and characterization of spray-dried porous nanoaggregates for pulmonary delivery of anti-tubercular drugs. Drug Deliv Taylor & Francis. 2016;23:872–7.
Bhardwaj A, Kumar L, Narang RK, Murthy RSR. Development and characterization of ligand-appended liposomes for multiple drug therapy for pulmonary tuberculosis. Artif cells, nanomedicine, Biotechnol. Taylor & Francis. 2013;41:52–9.
Bhardwaj A, Mehta S, Yadav S, Singh SK, Grobler A, Goyal AK, et al. Pulmonary delivery of antitubercular drugs using spray-dried lipid–polymer hybrid nanoparticles. Artif Cells, Nanomedicine, Biotechnol. Taylor & Francis. 2016;44:1544–55.
Chorepsima S, Kechagias KS, Kalimeris G, Triarides NA, Falagas ME. Spotlight on inhaled ciprofloxacin and its potential in the treatment of non-cystic fibrosis bronchiectasis. Drug Des Devel Ther. Dove Press. 2018;12:4059.
Vadakkan MV, Annapoorna K, Sivakumar KC, Mundayoor S, Kumar GSV. Dry powder cationic lipopolymeric nanomicelle inhalation for targeted delivery of antitubercular drug to alveolar macrophage. Int J Nanomedicine. Dove Press. 2013;8:2871.
Son Y-J, McConville JT. Preparation of sustained release rifampicin microparticles for inhalation. J Pharm Pharmacol Oxford University Press. 2012;64:1291–302.
Ohashi K, Kabasawa T, Ozeki T, Okada H. One-step preparation of rifampicin/poly (lactic-co-glycolic acid) nanoparticle-containing mannitol microspheres using a four-fluid nozzle spray drier for inhalation therapy of tuberculosis. J Control release Elsevier. 2009;135:19–24.
O’Hara P, Hickey AJ. Respirable PLGA microspheres containing rifampicin for the treatment of tuberculosis: manufacture and characterization. Pharm Res Springer. 2000;17:955–61.
Rojanarat W, Changsan N, Tawithong E, Pinsuwan S, Chan H-K, Srichana T. Isoniazid proliposome powders for inhalation—preparation, characterization and cell culture studies. Int J Mol Sci. Molecular Diversity Preservation International (MDPI). 2011;12:4414–34.
Pham D-D, Fattal E, Ghermani N, Guiblin N, Tsapis N. Formulation of pyrazinamide-loaded large porous particles for the pulmonary route: avoiding crystal growth using excipients. Int J Pharm Elsevier. 2013;454:668–77.
Parumasivam T, Leung SSY, Quan DH, Triccas JA, Britton WJ, Chan H-K. Rifapentine-loaded PLGA microparticles for tuberculosis inhaled therapy: preparation and in vitro aerosol characterization. Eur J Pharm Sci Elsevier. 2016;88:1–11.
Chan JGY, Tyne AS, Pang A, McLachlan AJ, Perera V, Chan JCY, et al. Murine pharmacokinetics of rifapentine delivered as an inhalable dry powder. Int J Antimicrob Agents Elsevier. 2015;45:319–23.
Chan JGY, Duke CC, Ong HX, Chan JCY, Tyne AS, Chan H-K, et al. A novel inhalable form of rifapentine. J Pharm Sci Elsevier. 2014;103:1411–21.
Patil-Gadhe A, Pokharkar V. Single step spray drying method to develop proliposomes for inhalation: a systematic study based on quality by design approach. Pulm Pharmacol Ther Elsevier. 2014;27:197–207.
Ahmad MI, Ungphaiboon S, Srichana T. The development of dimple-shaped chitosan carrier for ethambutol dihydrochloride dry powder inhaler. Drug Dev Ind Pharm. Taylor & Francis. 201541:791–800.
Ahmad MI, Nakpheng T, Srichana T. The safety of ethambutol dihydrochloride dry powder formulations containing chitosan for the possibility of treating lung tuberculosis. Inhal Toxicol Taylor & Francis. 2014;26:908–17.
Schoubben A, Giovagnoli S, Tiralti MC, Blasi P, Ricci M. Capreomycin inhalable powders prepared with an innovative spray-drying technique. Int J Pharm Elsevier. 2014;469:132–9.
Shao Z, Tai W, Qiu Y, Man RCH, Liao Q, Chow MYT, et al. Spray-dried powder formulation of capreomycin designed for inhaled tuberculosis therapy. Pharm. 2021.
Verma RK, Germishuizen WA, Motheo MP, Agrawal AK, Singh AK, Mohan M, et al. Inhaled microparticles containing clofazimine are efficacious in treatment of experimental tuberculosis in mice. Antimicrob Agents Chemother Am Soc Microbiol. 2013;57:1050–2.
Palazzo F, Giovagnoli S, Schoubben A, Blasi P, Rossi C, Ricci M. Development of a spray-drying method for the formulation of respirable microparticles containing ofloxacin–palladium complex. Int J Pharm Elsevier. 2013;440:273–82.
Momin MAM, Sinha S, Tucker IG, Doyle C, Das SC. Dry powder formulation of kanamycin with enhanced aerosolization efficiency for drug-resistant tuberculosis. Int J Pharm Elsevier. 2017;528:107–17.
Ibrahim M, Hatipoglu MK, Garcia-Contreras L. SHetA2 dry powder aerosols for tuberculosis: formulation, design, and optimization using quality by design. Mol Pharm ACS Publications. 2018;15:300–13.
Rossi I, Buttini F, Sonvico F, Affaticati F, Martinelli F, Annunziato G, et al. Sodium hyaluronate nanocomposite respirable microparticles to tackle antibiotic resistance with potential application in treatment of mycobacterial pulmonary infections. Pharmaceutics. MDPI. 2019;11:203.
Kadota K, Senda A, Tagishi H, Ayorinde JO, Tozuka Y. Evaluation of highly branched cyclic dextrin in inhalable particles of combined antibiotics for the pulmonary delivery of anti-tuberculosis drugs. Int J Pharm Elsevier. 2017;517:8–18.
Manca ML, Cassano R, Valenti D, Trombino S, Ferrarelli T, Picci N, et al. Isoniazid-gelatin conjugate microparticles containing rifampicin for the treatment of tuberculosis. J Pharm Pharmacol Oxford University Press. 2013;65:1302–11.
Chan JGY, Chan H-K, Prestidge CA, Denman JA, Young PM, Traini D. A novel dry powder inhalable formulation incorporating three first-line anti-tubercular antibiotics. Eur J Pharm Biopharm Elsevier. 2013;83:285–92.
Cunha L, Rodrigues S, Rosa da Costa AM, Faleiro ML, Buttini F, Grenha A. Inhalable fucoidan microparticles combining two antitubercular drugs with potential application in pulmonary tuberculosis therapy. Polymers (Basel). MDPI. 2018;10:636.
Rodrigues S, Alves AD, Cavaco JS, Pontes JF, Guerreiro F, da Costa AMR, et al. Dual antibiotherapy of tuberculosis mediated by inhalable locust bean gum microparticles. Int J Pharm Elsevier. 2017;529:433–41.
Eedara BB, Rangnekar B, Sinha S, Doyle C, Cavallaro A, Das SC. Development and characterization of high payload combination dry powders of anti-tubercular drugs for treating pulmonary tuberculosis. Eur J Pharm Sci Elsevier. 2018;118:216–26.
Rangnekar B, Momin MAM, Eedara BB, Sinha S, Das SC. Bedaquiline containing triple combination powder for inhalation to treat drug-resistant tuberculosis. Int J Pharm. Elsevier. 2019;570:118689.
Momin MAM, Sinha S, Tucker IG, Das SC. Carrier-free combination dry powder inhaler formulation of ethionamide and moxifloxacin for treating drug-resistant tuberculosis. Drug Dev Ind Pharm. Taylor & Francis. 2019;45:1321–31.
Ranjan R, Srivastava A, Bharti R, Ray L, Singh J, Misra A. Preparation and optimization of a dry powder for inhalation of second-line anti-tuberculosis drugs. Int J Pharm Elsevier. 2018;547:150–7.
Parumasivam T, Chan JGY, Pang A, Quan DH, Triccas JA, Britton WJ, et al. In vitro evaluation of inhalable verapamil-rifapentine particles for tuberculosis therapy. Mol Pharm ACS Publications. 2016;13:979–89.
Parumasivam T, Chan JGY, Pang A, Quan DH, Triccas JA, Britton WJ, et al. In vitro evaluation of novel inhalable dry powders consisting of thioridazine and rifapentine for rapid tuberculosis treatment. Eur J Pharm Biopharm Elsevier. 2016;107:205–14.
Durham PG, Young EF, Braunstein MS, Welch JT, Hickey AJ. A dry powder combination of pyrazinoic acid and its n-propyl ester for aerosol administration to animals. Int J Pharm Elsevier. 2016;514:384–91.
Gupta A, Sharma D, Meena J, Pandya S, Sachan M, Kumar S, et al. Preparation and preclinical evaluation of inhalable particles containing rapamycin and anti-tuberculosis agents for induction of autophagy. Pharm Res Springer. 2016;33:1899–912.
Hoppentocht M, Akkerman OW, Hagedoorn P, Frijlink HW, de Boer AH. The cyclops for pulmonary delivery of aminoglycosides; a new member of the Twincer™ family. Eur J Pharm Biopharm Elsevier. 2015;90:8–15.
Chan JGY, Tyne AS, Pang A, Chan H-K, Young PM, Britton WJ, et al. A rifapentine-containing inhaled triple antibiotic formulation for rapid treatment of tubercular infection. Pharm Res Springer. 2014;31:1239–53.
Maretti E, Costantino L, Rustichelli C, Leo E, Croce MA, Buttini F, et al. Surface engineering of solid lipid nanoparticle assemblies by methyl α-d-mannopyranoside for the active targeting to macrophages in anti-tuberculosis inhalation therapy. Int J Pharm Elsevier. 2017;528:440–51.
Scolari IR, Páez PL, Sánchez-Borzone ME, Granero GE. Promising chitosan-coated alginate-tween 80 nanoparticles as rifampicin coadministered ascorbic acid delivery carrier against mycobacterium tuberculosis. Aaps Pharmscitech Springer. 2019;20:1–21.
Rawal T, Parmar R, Tyagi RK, Butani S. Rifampicin loaded chitosan nanoparticle dry powder presents an improved therapeutic approach for alveolar tuberculosis. Colloids Surfaces B Biointerfaces Elsevier. 2017;154:321–30.
Diab R, Brillault J, Bardy A, Gontijo AVL, Olivier JC. Formulation and in vitro characterization of inhalable polyvinyl alcohol-free rifampicin-loaded PLGA microspheres prepared with sucrose palmitate as stabilizer: efficiency for ex vivo alveolar macrophage targeting. Int J Pharm Elsevier. 2012;436:833–9.
Mukhtar M, Csaba N, Robla S, Varela-Calviño R, Nagy A, Burian K, et al. Dry powder comprised of isoniazid-loaded nanoparticles of hyaluronic acid in conjugation with mannose-anchored chitosan for macrophage-targeted pulmonary administration in tuberculosis. Pharm. 2022.
Lawlor C, O’Sullivan MP, Rice B, Dillon P, Gallagher PJ, O’Leary S, et al. Therapeutic aerosol bioengineering of targeted, inhalable microparticle formulations to treat Mycobacterium tuberculosis (MTb). J Mater Sci Mater Med Springer. 2012;23:89–98.
Nemati E, Mokhtarzadeh A, Panahi-Azar V, Mohammadi A, Hamishehkar H, Mesgari-Abbasi M, et al. Ethambutol-loaded solid lipid nanoparticles as dry powder inhalable formulation for tuberculosis therapy. AAPS PharmSciTech Springer. 2019;20:1–9.
Brunaugh AD, Jan SU, Ferrati S, Smyth HDC. Excipient-free pulmonary delivery and macrophage targeting of clofazimine via air jet micronization. Mol Pharm ACS Publications. 2017;14:4019–31.
Sibum I, Hagedoorn P, Frijlink HW, Grasmeijer F. Characterization and formulation of isoniazid for high-dose dry powder inhalation. Pharmaceutics. MDPI. 2019;11:233.
Sibum I, Hagedoorn P, Kluitman MPG, Kloezen M, Frijlink HW, Grasmeijer F. Dispersibility and storage stability optimization of high dose isoniazid dry powder inhalation formulations with L-leucine or trileucine. Pharmaceutics. MDPI. 201912:24.
Wang C, Hickey AJ. Isoxyl aerosols for tuberculosis treatment: preparation and characterization of particles. AAPS PharmSciTech Springer. 2010;11:538–49.
Etschmann C, Scherließ R. Formulation of rifampicin softpellets for high dose delivery to the lungs with a novel high dose dry powder inhaler. Int J Pharm. 2022;617:121606. Available from: https://www.sciencedirect.com/science/article/pii/S0378517322001600.
Kujur S, Singh A, Singh C. Inhalation potential of rifampicin-loaded novel metal-organic frameworks for improved lung delivery: physicochemical characterization, in vitro aerosolization and antimycobacterial studies. J Aerosol Med Pulm Drug Deliv. United States. 2022.
Fernández-Paz C, Fernández-Paz E, Salcedo-Abraira P, Rojas S, Barrios-Esteban S, Csaba N, et al. Microencapsulated isoniazid-loaded metal-organic frameworks for pulmonary administration of antituberculosis drugs. Molecules. 2021;26.
Costa-Gouveia J, Pancani E, Jouny S, Machelart A, Delorme V, Salzano G, et al. Combination therapy for tuberculosis treatment: pulmonary administration of ethionamide and booster co-loaded nanoparticles. Sci Rep Nature Publishing Group. 2017;7:1–14.
Gaspar DP, Faria V, Gonçalves LMD, Taboada P, Remuñán-López C, Almeida AJ. Rifabutin-loaded solid lipid nanoparticles for inhaled antitubercular therapy: physicochemical and in vitro studies. Int J Pharm Elsevier. 2016;497:199–209.
Chimote G, Banerjee R. In vitro evaluation of inhalable isoniazid‐loaded surfactant liposomes as an adjunct therapy in pulmonary tuberculosis. J Biomed Mater Res Part B Appl Biomater. Wiley Online Library. 2010;94:1–10.
Chimote G, Banerjee R. Evaluation of antitubercular drug‐loaded surfactants as inhalable drug‐delivery systems for pulmonary tuberculosis. J Biomed Mater Res Part A An Off J Soc Biomater Japanese Soc Biomater Aust Soc Biomater Korean Soc Biomater. Wiley Online Library. 2009;89:281–92.
Cilfone NA, Pienaar E, Thurber GM, Kirschner DE, Linderman JJ. Systems pharmacology approach toward the design of inhaled formulations of rifampicin and isoniazid for treatment of tuberculosis. CPT pharmacometrics Syst Pharmacol. Wiley Online Library. 2015;4:193–203.
Khadka P, Hill PC, Zhang B, Katare R, Dummer J, Das SC. A study on polymorphic forms of rifampicin for inhaled high dose delivery in tuberculosis treatment. Int J Pharm. Elsevier. 2022;587:119602.
Singh C, Koduri LVSK, Singh A, Suresh S. Novel potential for optimization of antitubercular therapy: pulmonary delivery of rifampicin lipospheres. Asian J Pharm Sci. Elsevier. 2015;10:549–62.
Singh C, Koduri LVSK, Dhawale V, Bhatt TD, Kumar R, Grover V, et al. Potential of aerosolized rifampicin lipospheres for modulation of pulmonary pharmacokinetics and bio-distribution. Int J Pharm Elsevier. 2015;495:627–32.
Changsan N, Chan H, Separovic F, Srichana T. Physicochemical characterization and stability of rifampicin liposome dry powder formulations for inhalation. J Pharm Sci Elsevier. 2009;98:628–39.
Maretti E, Rustichelli C, Lassinantti Gualtieri M, Costantino L, Siligardi C, Miselli P, et al. The impact of lipid corona on rifampicin intramacrophagic transport using inhaled solid lipid nanoparticles surface-decorated with a mannosylated surfactant. Pharmaceutics. MDPI. 2019;11:508.
Sharma PR, Dravid AA, Kalapala YC, Gupta VK, Jeyasankar S, Goswami A, et al. Cationic inhalable particles for enhanced drug delivery to M. tuberculosis infected macrophages. Biomater Adv. 2022;133:112612. Available from: https://www.sciencedirect.com/science/article/pii/S0928493121007529.
Sung JC, Padilla DJ, Garcia-Contreras L, VerBerkmoes JL, Durbin D, Peloquin CA, et al. Formulation and pharmacokinetics of self-assembled rifampicin nanoparticle systems for pulmonary delivery. Pharm Res Springer. 2009;26:1847–55.
Larson SG. Scaling of organ weights in Macaca arctoides. Am J Phys Anthropol. Wiley Online Library. 1978;49:95–102.
Acknowledgements
Prakash Khadka would like to acknowledge the University of Otago for the postgraduate publishing bursary support.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. The first draft of the manuscript was written by Prakash Khadka, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Khadka, P., Dummer, J., Hill, P.C. et al. A review of formulations and preclinical studies of inhaled rifampicin for its clinical translation. Drug Deliv. and Transl. Res. 13, 1246–1271 (2023). https://doi.org/10.1007/s13346-022-01238-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-022-01238-y








