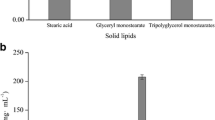
Abstract
Transdermal administration of raloxifene hydrochloride (RLX)-loaded nanostructured lipid carriers (NLCs) has been proposed to circumvent its low oral bioavailability (2%). Preformulation studies were carried out to evaluate drug-excipient compatibility of various adjuvants commonly used for NLC preparation (waxes, cholesterol, compritol, gelucire, span 60, span 80, span 85, tween 80, poloxamer 188, oleic acid, caprylic/capric triglyceride, and castor oil). It was used differential scanning calorimetry (DSC), isothermal stress testing (IST), and solubility studies. The most promising excipients were chosen for NLC obtention, and full characterization was done, including in vitro skin permeation. DSC curves suggested drug-excipient interaction among some compounds, and the IST study showed incompatibility of RLX with waxes, compritol, cholesterol, span 60, and poloxamer 188. Solubility studies helped select gelucire, caprylic/capric triglyceride, span 80, and tween 80 for NLC production. Twelve NLCs were obtained (NLC1 to NLC12), but NLC7 and NLC8 were the most promising ones. In vitro release studies demonstrated that NLC7 and NLC8 were able to control RLX release (14.74 and 9.07% at 24 h, respectively) compared with the unloaded drug (> 90% at 24 h). Unloaded RLX did not permeate the diffusion cells’ receptor medium and showed higher drug skin retention (11-fold) than RLX-loaded NLC. NLC reduced RLX skin retention, favoring drug permeation to deeper skin layers. NLC7 increased drug flux is 2.4-fold. NLC7 is a promising formulation for RLX transdermal drug delivery.
Graphical abstract





Similar content being viewed by others
References
Ravi PR, Aditya N, Kathuria H, et al. Lipid nanoparticles for oral delivery of raloxifene: optimization, stability, in vivo evaluation and uptake mechanism. Eur J Pharm Biopharm. 2014;87:114–24.
Tran TH, Poudel BK, Marasini N, Chi S-C, et al. Preparation and evaluation of raloxifene-loaded solid dispersion nanoparticle by spray-drying technique without an organic solvent. Int J Pharm. 2013;443:50–7.
Kushwaha AK, Vuddanda PR, Karunanidhi P, Singh SK, et al. Development and evaluation of solid lipid nanoparticles of raloxifene hydrochloride for enhanced bioavailability. Biomed Res Int. 2013;2013:584–649.
Nabi-Meibodi M, Vatanara A, Najafabadi AR, Rouini MR, et al. The effective encapsulation of a hydrophobic lipid-insoluble drug in solid lipid nanoparticles using a modified double emulsion solvent evaporation method. Colloid Surface B. 2013;112:408–14.
Mahmood S, Taher M, Mandal UK. Experimental design and optimization of raloxifene hydrochloride loaded nanotransfersomes for transdermal application. Int J Nanomedicine. 2014;9:4331–46.
Thakkar HP, Savsani H, Kumar P. Ethosomal Hydrogel of Raloxifene HCl: Statistical Optimization & Ex Vivo Permeability Evaluation Across Microporated Pig Ear Skin. Curr Drug Deliv. 2016;13:1111–22.
Banerjee R. Overcoming the stratum corneum barrier: a nano approach. Drug Deliv and Transl Res. 2013;3:205–8.
Prow TW, Grice JE, Lin LL, Faye R, et al. Nanoparticles and microparticles for skin drug delivery. Adv Drug Deliv Rev. 2011;63:470–91.
Fang DL, Chen Y, Xu B, Ren K, et al. Sevelopment of lipid-shell and polymer core nanoparticles with water-soluble salidroside for anti-cancer therapy. Int J Mol Sci. 2014;15:3373–88.
Muller RH, Radtke M, Wissing SA. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv Drug Deliv Rev. 2002;54:S131–55.
Garcês A, Amaral MH, Sousa Lobo JM, Silva AC. Formulations based on solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) for cutaneous use: a review. Eur J Pharm Sci. 2018;112:159–67.
Gordillo-Galeano A, Elizabeth C, Mora-Huertas CE. Solid lipid nanoparticles and nanostructured lipid carriers: a review emphasizing on particle structure and drug release. Eur J Pharm Biopharm. 2018;133:285–308.
Joshi MD, Müller RH. Lipid nanoparticles for parenteral delivery of actives. Eur J Pharm Biopharm. 2009;71:161–72.
Teixeira MC, Carbonea C, Souto EB. Beyond liposomes: recent advances on lipid based nanostructures for poorly soluble/poorly permeable drug delivery. Prog Lipid Res. 2017;68:1–11.
Perteghella S, Mandracchia D, Torre ML, Tamma R, Ribatti D, Trapani A, Tripodo G. Anti-angiogenic activity of uncoated- and N, O-carboxymethyl-chitosan surface modified-Gelucire® 50/13 based solid lipid nanoparticles for oral delivery of curcumin. J Drug Deliv Sci Technol. 2020;56:101494.
Silva LAD, Teixeira FV, Serpa RC, Esteves NL, et al. Evaluation of carvedilol compatibility with lipid excipients for the development of lipid-based drug delivery systems. J Therm Anal Calorim. 2016;123:2337–44.
Teixeira F, Alves G, Ferreira M, Taveira SF, et al. Preformulation studies to guide the development of raloxifene lipid-based delivery systems. J Therm Anal Calorim. 2018;132:365–71.
Verma RK, Garg S. Selection of excipients for extended release formulations of glipizide through drug-excipient compatibility testing. Adv Drug Deliv Rev. 2005;38:633–44.
Chadha R, Bhandari S. Drug-excipient compatibility screening–role of thermoanalytical and spectroscopic techniques. J Pharmaceut Biomed. 2014;87:82–97.
Pani NR, Nath LK, Acharya S. Compatibility studies of nateglinide with excipients in immediate release tablets. Acta Pharmaceut. 2011;61:237–47.
Kaur A, Goindi S, Katare OP. Thermal analysis and quantitative characterization of compatibility between diflunisal and lipid excipients as raw materials for development of solid lipid nanoparticles. Thermochim Acta. 2016;643:23–32.
Kaur R, Sinha VR. Use of thermal and non thermal techniques for assessing compatibility between mirtazapine and solid lipids. J Pharm Biomed Anal. 2018;161:144–58.
Kumar N, Goindi S, Saini B, et al. Thermal characterization and compatibility studies of itraconazole and excipients for development of solid lipid nanoparticles. J Therm Anal Calorim. 2014;115:2375–83.
Khosa A, Reddi S, Saha RN. Nanostructured lipid carriers for site-specific drug delivery. Biomed Pharmacother. 2018;103:598–613.
Silva LAD, Cintra ER, Alonso ECP, et al. Selection of excipients for the development of carvedilol loaded lipid-based drug delivery systems. J Therm Anal Calorim. 2017;130:1593–604.
USP. United States Pharmacopeia and National Formulary. 38th ed. Rockville: United States Pharmacopeia Convention; 2015.
Patel K, Padhye S, Nagarsenker M. Duloxetine HCl lipid nanoparticles: preparation, characterization, and dosage form design. AAPS PharmSciTech. 2011;13:125–33.
Setthacheewakul S, Mahattanadul S, Phadoongsombut N, et al. Development and evaluation of self-microemulsifying liquid and pellet formulations of curcumin, and absorption studies in rats. Eur J Pharm Biopharm. 2010;76:475–85.
Patil PH, Belgamwar VS, Patil PR, et al. Enhancement of solubility and dissolution rate of poorly water soluble raloxifene using microwave induced fusion method. Braz J Pharm Sci. 2013;49:571–8.
Teixeira FV, Alves G, Ferreira M, et al. Preformulation studies to guide the development of raloxifene lipid-based delivery systems. J Therm Anal Calorim. 2018;132:365–71.
FDA. Guidance for industry: SUPAC-SS nonsterile semisolid dosage forms. Scale-up and pos approval changes: chemistry, manufacturing and controls: in vitro release testing and in vivo bioequivalence documentation. In: Research CfDEa, editor. United States 1997.
OECD. Guideline 428: skin absorption: in vitro methods. In: Development OfEC-oa, editor. Paris 2004.
Dash S, Murthy PN, Nath L, et al. Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol Pharm. 2010;67:217–23.
Tiţa B, Fulias A, Bandur G, et al. Compatibility study between ketoprofen and pharmaceutical excipients used in solid dosage forms. J Pharm Biomed. 2011;56:221–7.
Weiss-Angeli V, Poletto FS, de Marco SL, et al. Sustained antioxidant activity of quercetin-loaded lipid-core nanocapsules. J Nanosci Nanotechnol. 2012;12:2874–80.
Singh VK, Pramanik K, Ray SS, et al. Development and characterization of sorbitan monostearate and sesame oil-based organogels for topical delivery of antimicrobials. AAPS PharmSciTech. 2015;16:293–305.
Kishore RS, Pappenberger A, Dauphin IB, et al. Degradation of polysorbates 20 and 80: studies on thermal autoxidation and hydrolysis. J Pharm Sci. 2011;100:721–31.
Shah NV, Seth AK, Balaraman R, et al. Nanostructured lipid carriers for oral bioavailability enhancement of raloxifene: design and in vivo study. J Adv Res. 2016;7:423–34.
Lanigan RS, Yamarik TA. Final report on the safety assessment of sorbitan caprylate, sorbitan cocoate, sorbitan diisostearate, sorbitan dioleate, sorbitan distearate, sorbitan isostearate, sorbitan olivate, sorbitan sesquiisostearate, sorbitan sesquistearate, and sorbitan triisostearate. Int J Toxicol. 2002;21:93–112.
Rowe R, et al. Hand Book of Pharmaceutical Excipients 2006.
Yeh CK, Peng SL, Hsu IY. Co-surfactant of ethoxylated sorbitan ester and sorbitan monooleate for enhanced flushing of tetrachloroethylene. Chemosphere. 2002;49:421–30.
Elsheikh MA, Elnaggar YSR, Gohar EY, et al. Nanoemulsion liquid preconcentrates for raloxifene hydrochloride: optimization and in vivo appraisal. Int J Nanomedicine. 2012;7:3787–802.
Tran TH, Poudel BK, Marasini N, et al. Preparation and evaluation of raloxifene-loaded solid dispersion nanoparticle by spray-drying technique without an organic solvent. Int J Pharm. 2013;443:50–7.
Patel J, Kevin G, Patel A, et al. Design and development of a self-nanoemulsifying drug delivery system for telmisartan for oral drug delivery. Int J Pharm Investig. 2011;1:112–8.
Rosenblatt KM, Bunjes H. Evaluation of the drug loading capacity of different lipid nanoparticle dispersions by passive drug loading. Eur J Pharm Biopharm. 2017;117:49–59.
Souza LG, Silva EJ, Martins AL, et al. Development of topotecan loaded lipid nanoparticles for chemical stabilization and prolonged release. Eur J Pharm Biopharm. 2011;79:189–96.
Murthy A, Ravi PR, Kathuria H, et al. Oral bioavailability enhancement of raloxifene with nanostructured lipid carriers. Nanomaterials. 2020;10:1–17.
Burra M, Jukanti R, Janga KY, et al. Enhanced intestinal absorption and bioavailability of raloxifene hydrochloride via lyophilized solid lipid nanoparticles. Adv Powder Technol. 2013;24:393–402.
Nekkantia V, Venkateshwarlu V, Pillai R. Preparation, characterization and in vivo evaluation of raloxifene hydrochloride solid lipid nanoparticles. Pharm Nanotechnol. 2013;1:68–77.
Pereira MN, Schulte HL, Duarte N, et al. Solid effervescent formulations as new approach for topical minoxidil delivery. Eur J Pharm Sci. 2017;96:411–9.
Andrade LM, Silva LAD, Krawczyk-Santos AP, et al. Improved tacrolimus skin permeation by co-encapsulation with clobetasol in lipid nanoparticles: study of drug effects in lipid matrix by electron paramagnetic resonance. Eur J Pharm Biopharm. 2017;119:142–9.
Karjiban RA, Basri M, Rahman MB, et al. Molecular dynamics simulation of palmitate ester self-assembly with diclofenac. Int J Mol Sci. 2012;13:9572–83.
Alonso C, Lucas R, Barba C, et al. Skin delivery of antioxidant surfactants based on gallic acid and hydroxytyrosol. J Pharm Pharmacol. 2015;67:900–8.
Morello KC, Wurz GT, DeGregorio MW. Pharmacokinetics of selective estrogen receptor modulators. Clin Pharmacokinet. 2003;42:361–72.
Acknowledgements
The authors acknowledge the Chemical Industry of Goiás State (IQUEGO) to provide the necessary facilities and Dr. Luis Antonio Dantas Silva to contribute to DSC measurements.
Funding
This work was supported by Brazilian agencies CNPq, CAPES, and FAPEG.
Author information
Authors and Affiliations
Contributions
All the authors have read and approved the manuscript. The study concept was performed by Taveira, S. F. as the study design. Alves, G. L., Teixeira, F. V., Da Rocha, P. B. R., Krawczyk-Santos, A. P., and Andrade, L. M. had performed the data acquisition. Andrade, L. M. and Krawczyk-Santos, A. P had performed quality control of data. Cunha-Filho M., Marreto, R. N., and Taveira, S. F. had carried out data analysis and interpretation. Alves, G. L. had performed the statistical analysis. Taveira, S. F., Cunha-Filho M., and Marreto, R. N. had written and edited the manuscript for intellectual content. All the authors were agreed to be accountable for all aspects of work, ensuring integrity and accuracy.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Experimental procedures were performed according to ethical standards, especially as Normative Resolutions of the National Council for the Control of Animal Experimentation—CONCEA. Approval in June of 2017 by the Ethics Committee on the Use of Animals/CEUA-PRPI-UFG (process number 053/17). The consent to participate does not apply to this study.
Consent for publication
The approval for publication, including all formats irrespective of time and language, had been taken from all authors.
Conflict interests
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alves, G.L., Teixeira, F.V., da Rocha, P.B.R. et al. Preformulation and characterization of raloxifene-loaded lipid nanoparticles for transdermal administration. Drug Deliv. and Transl. Res. 12, 526–537 (2022). https://doi.org/10.1007/s13346-021-00949-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-021-00949-y