Abstract
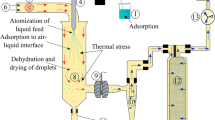
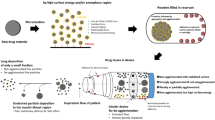
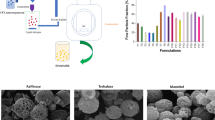
Dry powder inhalers (DPIs) have been proposed as an alternative administration route for protein and peptide drugs. However, DPI particles are easy to aggregate due to the strong interactions between the particles, leading to poor aerosolization performance. In this study, fragmented particles containing octreotide acetate (OA) were prepared by spray drying technique for dry powder inhalation, which were expected to decrease the particle-particle interaction by reducing the contact sites. Mannitol and ammonium carbonate were used as protein stabilizer and fragment-forming agent, respectively. The obtained fragmented particles presented larger particle size, lower density, better dispersibility, and well in vitro aerodynamic behavior (emitted dose > 97%, fine particle fraction ≈ 40%). The circular dichroism spectrum results indicated that OA maintained the stability throughout the spray drying process. The relative bioavailability of dry powder inhalation (DPI) compared with subcutaneous injection of commercial product was up to 88.0%, demonstrating the feasibility of DPI for OA delivery. These results confirmed that the proposed fragmented particles had great potential for pulmonary delivery of protein and peptide drugs in a painless, rapid, and convenient manner.






Similar content being viewed by others
References
Katz MD, Erstad BL. Octreotide, a new somatostatin analogue. Clin Pharm. 1989;8(4):255–73.
Peng JQ, Qi XL, Chen Y, Ma N, Zhang ZW, Xing JX, et al. Octreotide-conjugated PAMAM for targeted delivery to somatostatin receptors over-expressed tumor cells. J Drug Target. 2014;22(5):428–38.
Azkur D, Yoldas T, Toyran M, Kocabas CN. A pediatric case of anaphylaxis due to octreotide. Asian Pac J Allergy Immunol. 2011;29(4):361–3.
Maggio ET, Grasso P. Oral delivery of octreotide acetate in Intravail® improves uptake, half-life, and bioavailability over subcutaneous administration in male Swiss Webster mice. Regul Pept. 2011;167(2):233–8.
Lerner EN, van Zanten EH, Stewart GR. Enhanced delivery of octreotide to the brain via transnasal iontophoretic administration. J Drug Target. 2004;12(5):273–80.
Schuetz YB, Naik A, Guy RH, Kalia YN. Effect of amino acid sequence on transdermal iontophoretic peptide delivery. Eur J Pharm Sci. 2005;26(5):429–37.
Tug T, Kara H, Karaoglu A, Karatas F, Turgut NH, Ayan E, et al. The effect of octreotide, an analog of somatostatin, on bleomycin-induced interstitial pulmonary fibrosis in rats. Drug Chem Toxicol. 2013;36(2):181–6.
Dua K, Hansbro NG, Foster PS, Hansbro PM. MicroRNAs as therapeutics for future drug delivery systems in treatment of lung diseases. Drug Deliv Transl Res. 2017;7(1):168–78.
Wu L, Miao X, Shan Z, Huang Y, Li L, Pan X, et al. Studies on the spray dried lactose as carrier for dry powder inhalation. Asian J Pharm Sci. 2014;9(6):336–41.
Elia A, Cocchi M, Cottini C, Riolo D, Cafiero C, Bosi R, et al. Multivariate data analysis to assess dry powder inhalers performance from powder properties. Powder Technol. 2016;301(Supplement C):830–8.
Salama RO, Young PM, Traini D. Concurrent oral and inhalation drug delivery using a dual formulation system: the use of oral theophylline carrier with combined inhalable budesonide and terbutaline. Drug Deliv Transl Res. 2014;4(3):256–67.
Saboti D, Maver U, Chan HK, Planinsek O. Novel budesonide particles for dry powder inhalation prepared using a microfluidic reactor coupled with ultrasonic spray freeze drying. J Pharm Sci. 2017;106:1881–8.
Muhammad SAFS, Oubani H, Abbas A, Chan H-K, Kwok PCL, Dehghani F. The production of dry powder by the sonocrystallisation for inhalation drug delivery. Powder Technol. 2013;246:337–44.
Chow AH, Tong HH, Chattopadhyay P, Shekunov BY. Particle engineering for pulmonary drug delivery. Pharm Res. 2007;24(3):411–37.
Sung JC, Pulliam BL, Edwards DA. Nanoparticles for drug delivery to the lungs. Trends Biotechnol. 2007;25(12):563–70.
Tewes F, Paluch KJ, Tajber L, Gulati K, Kalantri D, Ehrhardt C, et al. Steroid/mucokinetic hybrid nanoporous microparticles for pulmonary drug delivery. Eur J Pharm Biopharm. 2013;85(3 Pt A):604–13.
Peng T, Zhang X, Huang Y, Zhao Z, Liao Q, Xu J, et al. Nanoporous mannitol carrier prepared by non-organic solvent spray drying technique to enhance the aerosolization performance for dry powder inhalation. Sci Rep. 2017;7:46517.
Littringer EM, Noisternig MF, Mescher A, Schroettner H, Walzel P, Griesser UJ, et al. The morphology and various densities of spray dried mannitol. Powder Technol. 2013;246:193–200.
Bivas-Benita M, Zwier R, Junginger HE, Borchard G. Non-invasive pulmonary aerosol delivery in mice by the endotracheal route. Eur J Pharm Biopharm. 2005;61(3):214–8.
Gradon L, Sosnowski TR. Formation of particles for dry powder inhalers. Adv Powder Technol. 2014;25(1):43–55.
Traini D, Young PM, Rogueda P, Price R. In vitro investigation of drug particulates interactions and aerosol performance of pressurised metered dose inhalers. Pharm Res. 2007;24(1):125–35.
Healy AM, McDonald BF, Tajber L, Corrigan OI. Characterisation of excipient-free nanoporous microparticles (NPMPs) of bendroflumethiazide. Eur J Pharm and Biopharm. 2008;69(3):1182–6.
Nolan LM, Tajber L, McDonald BF, Barham AS, Corrigan OI, Healy AM. Excipient-free nanoporous microparticles of budesonide for pulmonary delivery. Eur J Pharm Sci. 2009;37(5):593–602.
Littringer EM, Paus R, Mescher A, Schroettner H, Walzel P, Urbanetz NA. The morphology of spray dried mannitol particles—the vital importance of droplet size. Powder Technol. 2013;239:162–74.
Kaialy W, Alhalaweh A, Velaga SP, Nokhodchi A. Influence of lactose carrier particle size on the aerosol performance of budesonide from a dry powder inhaler. Powder Technol. 2012;227(Supplement C):74–85.
Kwok PC, Chan HK. Effect of relative humidity on the electrostatic charge properties of dry powder inhaler aerosols. Pharm Res. 2008;25(2):277–88.
Mishra DK, Dhote V, Bhargava A, Jain DK, Mishra PK. Amorphous solid dispersion technique for improved drug delivery: basics to clinical applications. Drug Deliv Transl Res. 2015;5(6):552–65.
Monckedieck M, Kamplade J, Fakner P, Urbanetz NA, Walzel P, Steckel H, et al. Dry powder inhaler performance of spray dried mannitol with tailored surface morphologies as carrier and salbutamol sulphate. Int J Pharm. 2017;524(1–2):351–63.
Hulse WL, Forbes RT, Bonner MC, Getrost M. Do co-spray dried excipients offer better lysozyme stabilisation than single excipients? Eur J Pharm Sci. 2008;33(3):294–305.
Saint-Lorant G, Leterme P, Gayot A, Flament MP. Influence of carrier on the performance of dry powder inhalers. Int J Pharm. 2007;334(1–2):85–91.
Kwok PC, Grabarek A, Chow MY, Lan Y, Li JC, Casettari L, et al. Inhalable spray-dried formulation of D-LAK antimicrobial peptides targeting tuberculosis. Int J Pharm. 2015;491(1–2):367–74.
Schule S, Schulz-Fademrecht T, Garidel P, Bechtold-Peters K, Frieb W. Stabilization of IgG1 in spray-dried powders for inhalation. Eur J Pharm Biopharm. 2008;69(3):793–807.
Koushik K, Dhanda DS, Cheruvu NP, Kompella UB. Pulmonary delivery of deslorelin: large-porous PLGA particles and HPbetaCD complexes. Pharm Res. 2004;21(7):1119–26.
Zheng M, Yu J. The effect of particle shape and size on cellular uptake. Drug Deliv Transl Res. 2016;6(1):67–72.
Funding
This study received financial support from the National Science Foundation of China (Grant No. 81673375), the Science and Technology Plan Projects of Guangdong Province (Grant No. 2015B020232010), and Basic Scientific Research Business Expense of the University for the Young Teacher Cultivation Project of Sun Yat-sen University (Grant No. 16ykpy23).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical approval
All the animal experiments were approved by the Ethical Committee of Sun Yat-sen University and performed in accordance with the guidelines of the Ethics of the World Medical Association.
Rights and permissions
About this article
Cite this article
Hou, A., Li, L., Huang, Y. et al. Fragmented particles containing octreotide acetate prepared by spray drying technique for dry powder inhalation. Drug Deliv. and Transl. Res. 8, 693–701 (2018). https://doi.org/10.1007/s13346-018-0515-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-018-0515-7