Abstract
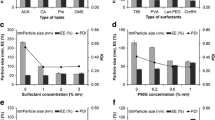
Nanostructured lipid carrier (NLC)-based gel was developed as a potential topical system for clobetasol propionate (CP) topical delivery for the treatment of eczema. The characterizations of the prepared NLC formulation for topical application on the skin were assessed by means of morphology (SEM), particle size distribution, zeta potential analysis, drug entrapment efficiency, and in vitro drug release studies to select the optimized NLC formulation. The optimized NLC formulation encompasses particle size of 137.9 nm with −20.5 mV zeta potential and 0.224 polydispersity index which indicates good stability of NLC dispersion. NLC formulation showed a good entrapment efficiency of 78.5 % ± 0.03 with cumulative in vitro release 85.42 % up to 24 h. The optimized NLC formulation was suitably gelled and characterized for rheology, drug content, ex vivo drug permeation studies, and drug release kinetics studies. The permeation study revealed that the permeability parameters like steady-state flux (Jss), permeability coefficient (Kp), and enhancement ratio were significantly higher for NLC-based gel formulation as compared to marketed formulation of clobetasol propionate. The value of r 2 (Korsmeyer–Peppas equation) indicated good linearity showing anomalous (non-Fickian) diffusion viz. drug release is controlled by more than one process, i.e., superposition of both phenomenon, the diffusion controlled as well as swelling controlled release. The anti-inflammatory activity of NLC gel via paw oedema technique showed a rapid onset of action, as well as a prolonged duration of action as compared with the marketed gel.







Similar content being viewed by others
References
Pardeike J, Hommoss A, Müller RH. Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. Int J Pharm. 2009;366:170–84.
Mehnert W, Mader K. Solid lipid nanoparticles: production, characterization and applications. Adv Drug Del Rev. 2001;47:165–96.
Müller RH, Petersen RD, Hommoss A, Pardeike J. Nanostructured lipid carriers (NLC) in cosmetic dermal products. Adv Drug Del Rev. 2007;59:522–30.
Proksch E, Fölster-Holst R, Jensen JM. Skin barrier function, epidermal proliferation and differentiation in eczema. J Dermatol Sci. 2006;43:159–69.
Chowdhury MMU. Dermatological pharmacology: topical agents. Medicine. 2008;37:232–4.
Simpson EL. Atopic dermatitis: a review of topical treatment options. Curr Med Res Opin. 2010;26(3):633–40.
Keshri L, Pathak K. Development of thermodynamically stable nanostructured lipid carrier system using central composite design for zero order permeation of econazole nitrate through epidermis. Pharm Dev Technol. 2013;18(3):634–44.
Nanjwade BK, Kadam VT, Manvi FV. Formulation and characterization of nanostructured lipid carrier of ubiquinone (Coenzyme Q10). J Biomed Nanotechnol. 2013;9(3):450–60.
Seyfoddin A, Al-Kassas R. Development of solid lipid nanoparticles and nanostructured lipid carriers for improving ocular delivery of acyclovir. Drug Dev Ind Pharm. 2013;39(4):508–19.
Shah M, Agrawal Y. Development of ciprofloxacin HCl-based solid lipid nanoparticles using ouzo effect: an experimental optimization and comparative study. J Dispers Sci Technol. 2013;34(1):37–46.
Li B, Ge ZQ. Nanostructured lipid carriers improve skin permeation and chemical stability of idebenone. AAPS PharmSciTech. 2012;13(1):276–83.
Khurana S, Bedi PMS, Jain NK. Development of nanostructured lipid carriers for controlled delivery of mefenamic acid. Int J Biomed Nanosci Nanotechnol. 2012;2(3/4):232–50.
Thatipamula RP, Palem CR, Gannu R, Mudragada S, Yamsani MR. Formulation and in vitro characterization of domperidone loaded solid lipid nanoparticles and nanostructured lipid carriers. DARU. 2011;19(1):23–32.
Bhalekar MR, Pokharkar V, Madgulkar A, Patil N, Patil N. Preparation and evaluation of miconazole nitrate-loaded solid lipid nanoparticles for topical delivery. AAPS PharmSciTech. 2009;10:289–96.
Mira EG, Egeaa MA, Garciaa ML, Souto EB. Design and ocular tolerance of flurbiprofen loaded ultrasound-engineered NLC. Colloids Surf B: Biointerfaces. 2010;81:412–21.
Sanad AR, Abdelmalak NS, Elbayoomy TS, Badawi AA. Formulation of a novel oxybenzone-loaded nanostructured lipid carrier (NLCs). AAPS PharmSciTech. 2010;11:1684–94.
Misal G, Dixit G, Gulkari V. Formulation and evaluation of herbal gel. Indian J Nat Prod Resour. 2012;3(4):501–5.
Das MK, Ahmed AB. Formulation and ex-vivo evaluation of rofecoxib gel for topical application. Acta Pol Pharma Drug Res. 2007;63(5):461–7.
Pople PV, Singh KK. Development and evaluation of colloidal modified nanolipid carrier: application to topical delivery of tacrolimus. Eur J Pharm Biopharm. 2011;79:82–94.
Araújo J, Nikolic S, Egea MA, Souto EB, Garcia ML. Nanostructured lipid carriers for triamcinolone acetonide delivery to the posterior segment of the eye. Colloids Surf B: Biointerfaces. 2011;88:150–7.
Sharma P, Dube B, Sawant K. Development and evaluation of nanostructured lipid carriers of cytarabine for treatment of meningeal leukemia. J Nanosci Nanotechnol. 2011;11(8):6676–82.
Chawla V, Saraf SA. Glyceryl behenate and its suitability for production of aceclofenac solid lipid nanoparticles. J Am Oil Chem Soc. 2011;88:119–26.
Jain NK, Ram A. Development and characterization of nanostructured lipid carriers of oral hypoglycemic agents: selection of surfactants. Int J Pharm Sci Rev Res. 2011;7(2):125–30.
Shi F, Yang G, Ren J, Guo T, Du T, Feng N. Formulation design, preparation and in vitro and in vivo characterization of ß-Elemene loaded Nanostructured lipid carriers. Int J Nanomed 2013; 8:2533–2541
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was reviewed and approved by the Institutional Animal Ethical Committee with registration number 837/ac/O4/CPCSEA.
Conflict of interest
The authors declare that they have no conflict of interest.
Animal studies
All institutional and national guidelines for the care and use of laboratory animals were followed.
Rights and permissions
About this article
Cite this article
Nagaich, U., Gulati, N. Nanostructured lipid carriers (NLC) based controlled release topical gel of clobetasol propionate: design and in vivo characterization. Drug Deliv. and Transl. Res. 6, 289–298 (2016). https://doi.org/10.1007/s13346-016-0291-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-016-0291-1