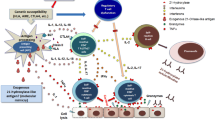
Abstract
A 72-year-old man with type 2 diabetes mellitus with good glucose control for 20 years and maintained on oral hypoglycaemic agents was diagnosed with Hodgkin’s Lymphoma (HL) and started on insulin glargine for glycaemic control. Despite increased doses of insulin, his blood glucose levels went up dramatically. The anti-insulin antibody test proved to be positive, and Scatchard plot analysis showed 2 binding sites with relatively low-affinity constants: K1 = 0.0032, K2 = 0.0002 (108/M); and high binding capacities: R1 = 98.4, R2 = 372 (10−8 M), which were compatible with the features of antibody of insulin autoimmune syndrome (IAS). However, hypoglycaemia was not noted throughout the course of treatment. Since the insulin binding ratio of the antibody decreased from 87.3% to 62% after the termination of insulin treatment, it was suggested that the antibody reacted mainly to exogenously injected insulin. Switching insulin preparations or introducing insulin secretagogues did not improve elevated blood glucose levels. The initiation of brentuximab vedotin (BV), a therapeutic agent for relapsed HL, resulted in a remarkable improvement in glycaemic control despite the absence of insulin therapy and partial remission of HL. This case suggested that HL triggered anti-insulin antibody production, which resulted in poor glycaemic control, and that BV could be a new treatment option for autoimmune diseases associated with HL.

Similar content being viewed by others
References
Berson SA, Yalow RS, Bauman A, et al. Insulin-I131 metabolism in human subjects: demonstration of insulin binding globulin in the circulation of insulin treated subjects. J Clin Invest. 1956;35(2):170–90. https://doi.org/10.1172/JCI103262.
Kaneko K, Satake C, Izumi T, et al. Enhancement of postprandial endogenous insulin secretion rather than exogenous insulin injection ameliorated insulin antibody-induced unstable diabetes: a case report. BMC Endocr Disord. 2019;19(1):5. https://doi.org/10.1186/s12902-018-0326-3.
Hirata Y, Ishizu H, Ouchi N, et al. Insulin autoimmunity in a case with spontaneous hypoglycemia. J Japn Diabetes Soc. 1970;13(4):312–20. https://doi.org/10.11213/tonyobyo1958.13.312 (in Japanese).
Rhie FH, Ganda OP, Bern MM, et al. Insulin resistance and monoclonal gammopathy. Metabolism. 1981;30(1):41–5. https://doi.org/10.1016/0026-0495(81)90216-x.
Redmon B, Pyzdrowski KL, Elson MK, et al. Hypoglycemia due to a monoclonal insulin-binding antibody in multiple myeloma. N Engl J Med. 1992;326(15):994–8. https://doi.org/10.1056/NEJM199204093261505.
Halsall DJ, Mangi M, Soos M, et al. Hypoglycemia due to an insulin binding antibody in a patient with an IgA-kappa myeloma. J Clin Endocrinol Metab. 2007;92(6):2013–6. https://doi.org/10.1210/jc.2007-0075.
Waldron-Lynch F, Inzucchi SE, Menard L, et al. Relapsing and remitting severe hypoglycemia due to a monoclonal anti-insulin antibody heralding a case of multiple myeloma. J Clin Endocrinol Metab. 2012;97(12):4317–23. https://doi.org/10.1210/jc.2012-2388.
Anderson LA, Gadalla S, Morton LM, et al. Population-based study of autoimmune conditions and the risk of specific lymphoid malignancies. Int J Cancer. 2009;125(2):398–405. https://doi.org/10.1002/ijc.24287.
Jachiet V, Mekinian A, Carrat F, et al. Autoimmune manifestations associated with lymphoma: characteristics and outcome in a multicenter retrospective cohort study. Leuk Lymphoma. 2018;59(6):1399–405. https://doi.org/10.1080/10428194.2017.1379075.
Liminet C, Vouillarmet J, Chikh K, Disse E. Antibody-mediated insulin resistance: when insulin and insulin receptor act as autoantigens in humans. Can J Diabetes. 2016;40(5):462–5. https://doi.org/10.1016/j.jcjd.2016.02.007.
Eguchi Y. Scatchard analysis of insulin autoantibodies in the insulin autoimmune syndrome. J Tokyo Women’s Med Coll. 1989;59(12):1286–305 (in Japanese).
Eguchi Y, Uchigata Y, Yao K, et al. Longitudinal changes of serum insulin concentration and insulin antibody features in persistent insulin autoimmune syndrome (Hirata’s disease). Autoimmunity. 1994;19(4):279–84. https://doi.org/10.3109/08916939409071354.
Uchigata Y. Insulin autoimmune syndrome. Nihon Rinsho. 2016;74(Suppl 2):602–5.
Jaeger C, Winter S, Eckhard M, et al. Binding characteristics and crossreactivity of insulin autoantibodies and insulin antibodies directed to three different insulin molecules. Acta Diabetol. 2008;45(3):191–4. https://doi.org/10.1007/s00592-008-0041-z.
Hayashi A, Takano K, Kawai S, Shichiri M. SGLT2 inhibitors provide an effective therapeutic option for diabetes complicated with insulin antibodies. Endocr J. 2016;63(2):187–91. https://doi.org/10.1507/endocrj.EJ15-0523.
Kato T, Iizuka K, Niwa H, Takeda J. Liraglutide improved glycaemic instability in a patient with diabetes with insulin antibodies. BMJ Case Rep. 2016;20(2016):bcr2016216028. https://doi.org/10.1136/bcr-2016-216028.
Censi S, Mian C, Betterle C. Insulin autoimmune syndrome: from diagnosis to clinical management. Ann Transl Med. 2018;6(17):335. https://doi.org/10.21037/atm.2018.07.32.
Li R, Mao J, Yu K, et al. Medical nutrition therapy is effective in the management of hypoglycemia caused by insulin antibodies: a case report and literature review. J Am Coll Nutr. 2016;35(1):86–90. https://doi.org/10.1080/07315724.2014.976673.
Matsuyoshi A, Shimoda S, Tsuruzoe K, et al. A case of slowly progressive type 1 diabetes with unstable glycemic control caused by unusual insulin antibody and successfully treated with steroid therapy. Diabetes Res Clin Pract. 2006;72(3):238–43. https://doi.org/10.1016/j.diabres.2005.10.018.
Fallah M, Liu X, Ji J, et al. Hodgkin lymphoma after autoimmune diseases by age at diagnosis and histological subtype. Ann Oncol. 2014;25(7):1397–404. https://doi.org/10.1093/annonc/mdu144.
Landgren O, Engels EA, Pfeiffer RM, et al. Autoimmunity and susceptibility to Hodgkin lymphoma: a population-based case-control study in Scandinavia. J Natl Cancer Inst. 2006;98(18):1321–30. https://doi.org/10.1093/jnci/djj361.
Grover NS, Dittus CE, Ma AD, Park SI. Hodgkin lymphoma with multiple autoimmune disorders: case report and review of the literature. Clin Lymphoma Myeloma Leuk. 2018;18(9):e365–8. https://doi.org/10.1016/j.clml.2018.06.009.
Váróczy L, Gergely L, Zeher M, et al. Malignant lymphoma-associated autoimmune diseases–a descriptive epidemiological study. Rheumatol Int. 2002;22(6):233–7. https://doi.org/10.1007/s00296-002-0229-4.
Goodnow CC. Multistep pathogenesis of autoimmune disease. Cell. 2007;130(1):25–35. https://doi.org/10.1016/j.cell.2007.06.033.
Deng C, Pan B, O’Connor OA. Brentuximab vedotin. Clin Cancer Res. 2013;19(1):22–7. https://doi.org/10.1158/1078-0432.CCR-12-0290.
Nakazato T, Takanashi S, Hirano M, et al. Brentuximab vedotin is effective for rheumatoid arthritis in a patient with relapsed methotrexate-associated Hodgkin lymphoma. Ann Hematol. 2018;97(8):1489–91. https://doi.org/10.1007/s00277-018-3279-8.
Ichikawa S, Fukuhara N, Saito K, et al. Successful treatment of methotrexate-associated classical Hodgkin lymphoma with brentuximab vedotin-combined chemotherapy: a case series. Int J Hematol. 2020;111(5):667–72. https://doi.org/10.1007/s12185-020-02822-z.
Vachhani P, Bose N, Brodeur JP, et al. Remission of rheumatoid arthritis on brentuximab vedotin. Rheumatology (Oxford). 2014;53(12):2314–5. https://doi.org/10.1093/rheumatology/keu374.
Jassam N, Amin N, Holland P, et al. Analytical and clinical challenges in a patient with concurrent type 1 diabetes, subcutaneous insulin resistance and insulin autoimmune syndrome. Endocrinol Diabetes Metab Case Rep. 2014;2014: 130086. https://doi.org/10.1530/EDM-13-0086.
Saxon DR, McDermott MT, Michels AW. Novel management of insulin autoimmune syndrome with rituximab and continuous glucose monitoring. J Clin Endocrinol Metab. 2016;101(5):1931–4. https://doi.org/10.1210/jc.2016-1097.
Schwab U, Stein H, Gerdes J, et al. Production of a monoclonal antibody specific for Hodgkin and Sternberg-Reed cells of Hodgkin’s disease and a subset of normal lymphoid cells. Nature. 1982;299(5878):65–7. https://doi.org/10.1038/299065a0.
Chiarle R, Podda A, Prolla G, et al. CD30 in normal and neoplastic cells. Clin Immunol. 1999;90(2):157–64. https://doi.org/10.1006/clim.1998.4636.
Romagnani P, Annunziato F, Manetti R, et al. High CD30 ligand expression by epithelial cells and Hassal’s corpuscles in the medulla of human thymus. Blood. 1998;91(9):3323–32.
Acknowledgements
The authors would like to thank Hiroyuki Ito for performing polyethylene glycol insulin assay and Enago (www.enago.jp) for the English language review.
Author information
Authors and Affiliations
Contributions
MI collected the data, interpreted the results, searched the literature, wrote the first draft of most sections of the manuscript, revised the manuscript. MF, FI, and YH supervised the study, interpreted the results, and revised the manuscript. KK, ST, YS, KT, SO, MK and ST revised the manuscript. All authors provided critical feedback and helped shape the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Yasuaki Hayashino received research funding from Ono Pharmaceutical Co. Ltd.
Human and animal rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Informed consent
Informed consent was obtained for the publication of the case report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Ikeda, M., Fujimura, M., Kurosawa, K. et al. Production of insulin antibody associated with relapsed hodgkin’s lymphoma. Diabetol Int 13, 456–460 (2022). https://doi.org/10.1007/s13340-021-00550-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-021-00550-1