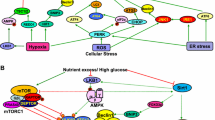
Abstract
Diabetic nephropathy, which is characterized by increased albuminuria, has been the most common cause of end-stage kidney disease for many years in Japan and many other countries. Although the renal prognosis of the disease has been improving in recent years because of the clinical implementation of strict glucose, blood pressure, and lipid controls, some diabetes patients continue to exhibit treatment-resistant macroalbuminuria leading to end-stage kidney disease. Furthermore, renal function decline without macroalbuminuria in diabetes is an emerging issue in Japan, which might be partly due to aging. Thus, a novel therapeutic strategy is needed to further improve renal outcome in diabetes patients. We have recently reported the involvement of dysregulation of intracellular nutrient-sensing signals and the related cellular process, autophagy, in the pathogenesis of diabetic nephropathy and abnormal insulin secretion pattern in type 2 diabetes. This review discusses potential roles of intracellular nutrient-sensing signals and autophagy as novel therapeutic targets for type 2 diabetes and diabetic nephropathy.


Similar content being viewed by others
References
Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54:1615–25.
Viberti GC, Hill RD, Jarrett RJ, Argyropoulos A, Mahmud U, Keen H. Microalbuminuria as a predictor of clinical nephropathy in insulin-dependent diabetes mellitus. Lancet. 1982;1:1430–2.
Pagtalunan ME, Miller PL, Jumping-Eagle S, Nelson RG, Myers BD, Rennke HG, Coplon NS, Sun L, Meyer TW. Podocyte loss and progressive glomerular injury in type II diabetes. J Clin Invest. 1997;99:342–8.
Abbate M, Zoja C, Remuzzi G. How does proteinuria cause progressive renal damage? J Am Soc Nephrol. 2006;17:2974–84.
Afkarian M, Zelnick LR, Hall YN, Heagerty PJ, Tuttle K, Weiss NS, de Boer IH. Clinical manifestations of kidney disease among US adults with diabetes, 1988–2014. JAMA. 2016;316:602–10.
Kume S, Araki SI, Ugi S, Morino K, Koya D, Nishio Y, Haneda M, Kashiwagi A, Maegawa H. Secular changes in clinical manifestations of kidney disease among Japanese adults with type 2 diabetes from 1996 to 2014. J Diabetes Investig. 2018;10:1032–40.
Sohal RS, Weindruch R. Oxidative stress, caloric restriction, and aging. Science. 1996;273:59–63.
Omodei D, Fontana L. Calorie restriction and prevention of age-associated chronic disease. FEBS Lett. 2011;585:1537–42.
Mattison JA, Colman RJ, Beasley TM, Allison DB, Kemnitz JW, Roth GS, Ingram DK, Weindruch R, de Cabo R, Anderson RM. Caloric restriction improves health and survival of rhesus monkeys. Nat Commun. 2017;8:14063.
Efeyan A, Comb WC, Sabatini DM. Nutrient-sensing mechanisms and pathways. Nature. 2015;517:302–10.
Kume S, Thomas MC, Koya D. Nutrient sensing, autophagy, and diabetic nephropathy. Diabetes. 2012;61:23–9.
Zhou XJ, Rakheja D, Yu X, Saxena R, Vaziri ND, Silva FG. The aging kidney. Kidney Int. 2008;74:710–20.
Tanaka T, Kato H, Kojima I, Ohse T, Son D, Tawakami T, Yatagawa T, Inagi R, Fujita T, Nangaku M. Hypoxia and expression of hypoxia-inducible factor in the aging kidney. J Gerontol A Biol Sci Med Sci. 2006;61(8):795–805.
Kume S, Uzu T, Horiike K, Chin-Kanasaki M, Isshiki K, Araki S, Sugimoto T, Haneda M, Kashiwagi A, Koya D. Calorie restriction enhances cell adaptation to hypoxia through Sirt1-dependent mitochondrial autophagy in mouse aged kidney. J Clin Invest. 2010;120:1043–55.
Haigis MC, Guarente LP. Mammalian sirtuins–emerging roles in physiology, aging, and calorie restriction. Genes Dev. 2006;20(21):2913–21.
Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell. 2008;132:27–422.
Kroemer G, Mariño G, Levine B. Autophagy and the integrated stress response. Mol Cell. 2010;40:280–93.
Mundel P, Shankland SJ. Podocyte biology and response to injury. J Am Soc Nephrol. 2002;13(12):3005–15.
Hartleben B, Gödel M, Meyer-Schwesinger C, Liu S, Ulrich T, Köbler S, Wiech T, Grahammer F, Arnold SJ, Lindenmeyer MT, Cohen CD, Pavenstädt H, Kerjaschki D, Mizushima N, Shaw AS, Walz G, Huber TB. Autophagy influences glomerular disease susceptibility and maintains podocyte homeostasis in aging mice. J Clin Invest. 2010;120:1084–96.
Tagawa A, Yasuda M, Kume S, Yamahara K, Nakazawa J, Chin-Kanasaki M, Araki H, Araki S, Koya D, Asanuma K, Kim EH, Haneda M, Kajiwara N, Hayashi K, Ohashi H, Ugi S, Maegawa H, Uzu T. Impaired podocyte autophagy exacerbates proteinuria in diabetic nephropathy. Diabetes. 2016;65:755–67.
Yasuda-Yamahara M, Kume S, Tagawa A, Maegawa H, Uzu T. Emerging role of podocyte autophagy in the progression of diabetic nephropathy. Autophagy. 2015;11:2385–6.
Yamahara K, Kume S, Koya D, Tanaka Y, Morita Y, Chin-Kanasaki M, Araki H, Isshiki K, Araki S, Haneda M, Matsusaka T, Kashiwagi A, Maegawa H, Uzu T. Obesity-mediated autophagy insufficiency exacerbates proteinuria-induced tubulointerstitial lesions. J Am Soc Nephrol. 2013;24:1769–81.
Kitada M, Ogura Y, Suzuki T, Sen S, Lee SM, Kanasaki K, Kume S, Koya D. A very-low-protein diet ameliorates advanced diabetic nephropathy through autophagy induction by suppression of the mTORC1 pathway in Wistar fatty rats, an animal model of type 2 diabetes and obesity. Diabetologia. 2016;59:1307–17.
Takagi A, Kume S, Kondo M, Nakazawa J, Chin-Kanasaki M, Araki H, Araki S, Koya D, Haneda M, Chano T, Matsusaka T, Nagao K, Adachi Y, Chan L, Maegawa H, Uzu T. Mammalian autophagy is essential for hepatic and renal ketogenesis during starvation. Sci Rep. 2016;6:18944.
Takagi A, Kume S, Maegawa H, Uzu T. Emerging role of mammalian autophagy in ketogenesis to overcome starvation. Autophagy. 2016;12:709–10.
Krebs HA, Bennett DA, De Gasquet P, Gasquet P, Gascoyne T, Yoshida T. Renal gluconeogenesis The effect of diet on the gluconeogenic capacity of rat-kidney-cortex slices. Biochem J. 1963;86:22–7.
Hardie DG, Ross FA, Hawley SA. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol. 2012;13:251–62.
Seino S, Shibasaki T, Minami K. Dynamics of insulin secretion and the clinical implications for obesity and diabetes. J Clin Invest. 2011;121:2118–255.
Kume S, Kondo M, Maeda S, Nishio Y, Yanagimachi T, Fujita Y, Haneda M, Kondo K, Sekine A, Araki SI, Araki H, Chin-Kanasaki M, Ugi S, Koya D, Kitahara S, Maeda K, Kashiwagi A, Uzu T, Maegawa H. Hypothalamic AMP-activated protein kinase regulates biphasic insulin secretion from pancreatic β cells during fasting and in type 2 diabetes. EBioMedicine. 2016;13:168–80.
Lundbaek K. Metabolic abnormalities in starvation diabetes. Yale J Biol Med. 1948;20(6):533–44.
Acknowledgements
This review is a summary of my presentation in the Lilly Award Lecture at the 62nd annual meeting of the Japan Diabetes Society, Sendai, Japan. I would like to express sincere gratitude to Professor Hiroshi Maegawa, Professor Daisuke Koya, Professor Masakazu Haneda, Professor Atsunori Kashiwagi, Professor Ryuichi Kikkawa, and all members of department of medicine, Shiga University of Medical Science for their guidance and support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Shinji Kume declares that he has no conflict of interest.
Statement of animal and/or human participants
This article does not contain any studies with human or animal subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Kume, S. Pathophysiological roles of nutrient-sensing mechanisms in diabetes and its complications. Diabetol Int 10, 245–249 (2019). https://doi.org/10.1007/s13340-019-00406-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-019-00406-9