Abstract
Aims and introduction
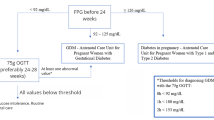
It is helpful for both diabetologists and obstetricians to identify patients with gestational diabetes who require insulin therapy for glycemic control during pregnancy. The aim of the present study was to assess potential predictors of insulin requirement in patients with gestational diabetes.
Materials and methods
One hundred thirteen patients with gestational diabetes [mean age 34.2 ± 4.5 years; pre-gestational body mass index (BMI), 23.6 ± 6.0 kg/m2] were included in this study. The associations between insulin requirement and the following factors were analyzed: clinical maternal characteristics, number of abnormal oral glucose tolerance test (OGTT) values, gestational age at diagnosis, plasma glucose levels measured during the OGTT, glycated hemoglobin (HbA1c) and glycated albumin levels, and serum C-peptide level before breakfast and 1 and 2 h after breakfast.
Results
Thirty-six patients (32 %) required insulin during pregnancy (“insulin group”); the remaining patients were treated with nutrition therapy (“nutrition group”). The insulin group had a higher pre-gestational BMI, higher fasting glucose level, higher area under the curve (AUC) for serum C-peptide level, higher HbA1c level, and a younger gestational age at diagnosis than the nutrition group (p < 0.05, all). Logistic regression analysis showed that the pre-gestational BMI, AUC for serum C-peptide level, and the HbA1c level were independent predictors for the insulin group (p < 0.05, all).
Conclusions
The results suggest that a high insulin resistance and HbA1c level may be associated with insulin requirement in patients with gestational diabetes.

Similar content being viewed by others
References
The HAPO Study Cooperative Research Group. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med. 2008;358:1991–2002.
Hartling L, Dryden DM, Guthrie A, Muise M, Vandermeer B, Donovan L. Benefits and harms of treating gestational diabetes mellitus: a systematic review and meta-analysis for the US Preventive Services Task Force and the National Institutes of Health Office of Medical Applications of Research. Ann Intern Med. 2013;159:123–9.
Landon MB, Spong CY, Thom E, Carpenter MW, Ramin SM, Casey B, Wanper RJ, Varner MW, Rouse DJ, Thorp JM, Sciscione A, Catalano P, Harper M, Saade G, Lain KY, Sorokin Y, Peaceman AM, Tolosa JE, Anderson GB, for the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. A multicenter, randomized trial of treatment for mild gestational diabetes. N Engl J Med. 2009;361:1339–48.
Crowther CA, Hiller JE, Moss JR, McPhee AJ, Jeffries WS, Robinson JS, for the Australian Carbohydrate Intolerance Study in Pregnant Women (ACHOIS) Trial Group. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med. 2005;352:2477–86.
Sugiyama T, Kusaka H, Sagawa S, Toyoda N. Committee report on nutrition and metabolism in gyneco-obstetrics—abnormal glucose metabolism in pregnancy, with special reference to the diagnosis of diabetes mellitus in pregnancy. Nippon Sanka Fujinka Gakkai Zasshi. 1984; 36:2055–2058 (in Japanese).
International Association of Diabetes and Pregnancy Study Groups, Consensus Panel. International Association of Diabetes and Pregnancy Study Groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care. 2010;33:676–82.
Morikawa M, Yamada T, Yamada T, Akaishi R, Nishida R, Cho K, Minakaki H. Change in the number of patients after the adoption of IADPSG criteria for hyperglycemia during pregnancy in Japanese women. Diabetes Res Clin Pract. 2010;90:339–42.
Ito Y, Shibuya M, Hosokawa S, Motoki Y, Nagata R, Konishi H, Miyazaki T, Matsunaga T, Nomura Y, Mihara T, Ito S, Sugiura K, Terauchi Y. Indicators of the need for insulin treatment and the effect of treatment for gestational diabetes on pregnancy outcomes in Japan. Endocr J. 2016;63:231–7.
Yogev Y, Ben-Haroush A, Hod M. Pathogenesis of gestational diabetes mellitus. In: Hod M, Jovanovic L, Di Renzo GC, De Leiva A, Langer O, editors. Textbook of diabetes and pregnancy. London: Informa Healthcare; 2008. p. 71–8.
Saisho Y, Miyakoshi K, Ikenoue S, Kasuga Y, Matsumoto T, Minegishi K, Yoshimura Y, Itoh H. Marked decline in beta cell function during pregnancy leads to the development of glucose intolerance in Japanese. Endocr J. 2013;60:533–9.
Ryan EA, Enns L. Role of gestational hormones in the induction of insulin resistance. J Clin Endocrinol Metab. 1988;67:341–7.
Richardson AC, Carpenter MW. Inflammatory mediators in gestational diabetes mellitus. Obstet Gynecol Clin N Am. 2007;34:213–24.
Pendergrass M, Fazioni E, DeFronzo RA. Non-insulin-dependent diabetes mellitus and gestational diabetes mellitus: same disease, another name? Diabetes Rev. 1995;3:566–83.
Bakiner O, Bozkirli E, Ozsahin K, Sariturk C, Ertorer E. Risk factors that can predict antenatal insulin need in gestational diabetes. J Clin Med Res. 2013;5:381–8.
Wong VW, Jalaludin B. Gestational diabetes mellitus: who requires insulin therapy? Aust N Z J Obstet Gynaecol. 2011;51:432–6.
Pertot T, Molyneaux L, Tan K, Ross GP, Yue DK, Wong J. Can common clinical parameters be used to identify patients who will need insulin treatment in gestational diabetes mellitus? Diabetes Care. 2011;34:2214–6.
Akinchi B, Celtik A, Yener S, Yesil S. Is fasting glucose level during oral glucose tolerance test an indicator of the insulin need in gestational diabetes? Diabetes Res Clin Pract. 2008;82:219–25.
Gonzalez-Quintero VH, Istwan NB, Rhea DJ, Tudela CM, Flick AA, de la Torre L, Stanziano GJ. Antenatal factors predicting subsequent need for insulin treatment in women with gestational diabetes. J Womens Health (Larchmt). 2008;17:1183–7.
Hanna FW, Peter JR. Screening for gestational diabetes; past, present and future. Diabet Med. 2002;19:351–8.
Hiramatsu Y, Shimizi I, Omori Y, Nakabayashi M, for the JGA (Japan Glycated Albumin) Study Group. Determination of reference intervals of glycated albumin and hemoglobin A1c in healthy pregnant Japanese women and analysis of their time courses and influencing factors during pregnancy. Endocr J. 2012;59:145–51.
Borg R, Kuenen JC, Carstensen B, Zheng H, Nathan DM, Heine RJ, Nerup J, Borch-Johnsen K, Witte DR, on behalf of the ADAG Study Group. Associations between features of glucose exposure and A1C. The A1C-derived average glucose (ADAG) study. Diabetes. 2010;59:1585–90.
Sakuma N, Omura M, Oda E, Saito T. Converse contributions of fasting and postprandial glucose to HbA1c and glycated albumin. Diabetol Int. 2011;2:162–71.
Koga M, Matsumoto S, Saito H, Kasayama S. Body mass index negatively influences glycated albumin, but not glycated hemoglobin, in diabetic patients. Endocr J. 2006;53:387–91.
Sapienza AD, Francisco RPV, Trindade TC, Zugaib M. Factors predicting the need for insulin therapy in patients with gestational diabetes mellitus. Diab Res Clin Pract. 2010;88:81–6.
Mitra S, Nayak PK, Sahoo J, Mathew A, Padma A, Kamalanathan S, Agrawal S. Predictors for antenatal insulin requirement in gestational diabetes. Gynecol Endocrinol. 2014;30:565–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H. Sakura received lecture fees from Mitsubishi Tanabe Pharma Corp. and research grants from Astra Zeneca K.K., Chugai Pharma Manufacturing Co., Ltd., Takeda Pharma Ltd., and Ono Pharmaceutical Co., Ltd. Y. Uchigata received lecture fees from Novo Nordisk Pharma Ltd., Sanofi K.K., Takeda Pharma Ltd., Mitsubishi Tanabe Pharma Corp., Ono Pharmaceutical Co., Eli Lilly Japan K.K., MSD K.K., and research grants from Novartis Phama K.K., Astellas Pharma Inc., Pfizer Japan Inc., Chugai Pharma Manufacturing Co., Ltd., Boehringer Ingerheim, Astra Zeneca K.K., Kyowa Hakko Kirin Co., Ltd., Alcon, Otsuka Pharmacuetical Co., Ltd., Nipro, Eli Lilly Japan K.K., Kowa Shinyaku Co., Ltd., Eizai Co., Ltd., Takeda Pharma Ltd., Sanofi K.K., Mitsubishi Tanabe Pharma Corp., MSD K.K., Ono Pharmaceutical Co., Novo Nordisk Phama Ltd., Terumo Corp., Sumitomo Dainippon Pharma Co., Ltd., and Diaiichu Sankyo Co., Ltd. The other authors declare that they have no conflict of interest.
Human rights statement and informed consent
All procedures conducted in this study were in accordance with the ethics committee of Tokyo Women’s Medical University and with the Declaration of Helsinki of 1964 and later revisions. Informed consent or a substitute for it was obtained from all patients before they were included in the study.
About this article
Cite this article
Yanagisawa, K., Muraoka, M., Takagi, K. et al. Assessment of predictors of insulin therapy in patients with gestational diabetes diagnosed according to the IADPSG criteria. Diabetol Int 7, 440–446 (2016). https://doi.org/10.1007/s13340-016-0272-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-016-0272-0