Abstract
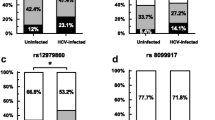
Type III interferon (IFNs) encoded by IFN lambda (IFNL) genes induce antiviral activity. The IFNL clusters include IFNL1/IL29, IFNL2/IL28A, IFNL3/IL28B and IFNL4 genes. The single nucleotide polymorphisms (SNPs, rs12979860 and rs8099917) associated with virological responses against hepatitis C virus (HCV) infections are recently mapped to IFNL4 gene. The IFNL gene polymorphisms also plays role in immune clearance, inflammation and risk of developing hepatocellular carcinoma. There is significant genetic heterogeneity of IFNL4 polymorphisms among ethnic populations that need to be regionally studied for viral infection, treatment response and relapse. The IFNL4 risk allele, genotype and haplotype frequencies across north Indian cohort were determined among chronic hepatitis C (CHC) cases (n = 141) and healthy controls (n = 111) by allele specific real-time PCR. Odds ratio was calculated for HCV exposure and treatment response using dominant and minor allele/genotype as reference. Non-random associations of these two SNP loci were evaluated by linkage disequilibrium plot. The minor allele (T) frequency of rs12979860C/T is 0.241 and 0.229; and minor allele (G) frequency for SNP rs8099917T/G is 0.174 and 0.171 among CHC cases and healthy control respectively. Coefficient of linkage disequilibrium (D′) of these two SNPs is very high (D′ = 0.98, r2 > 0.6) in CHC group than in healthy control (D′ = 0.76, r2 = 0.39) which indicate that both SNPs are strongly linked in CHC population than healthy control. Favorable association of IFNL4 haplotype (C–T), genotype (CC for rs12979860 and TT for rs8099917) with anti HCV therapy were found significant (p = 0.009, 0.021 and 0.001) for SVR. Favorable genotypes are also found to be predominant across the Indian study population.



Similar content being viewed by others
References
Aka PV, Kuniholm MH, Pfeiffer RM, et al. Association of the IFNL4-DeltaG allele with impaired spontaneous clearance of hepatitis C virus. J Infect Dis. 2013;209(3):350–4.
Ank N, Iversen MB, Bartholdy C, et al. An important role for type III interferon (IFN-lambda/IL-28) in TLR-induced antiviral activity. J Immunol. 2008;180(4):2474–85.
Asselah T, Benhamou Y, Marcellin P. Protease and polymerase inhibitors for the treatment of hepatitis C. Liver Int. 2009;29(Suppl 1):57–67.
Asthana M, Sahu SK, Kumar A, et al. Role of Interleukin 28B polymorphisms in response to Interferon based therapy for hepatitis C virus clearance. Curr Drug Metab. 2018;19:215–23.
Bhushan A, Chinnaswamy S. Identifying causal variants at the interferon lambda locus in case-control studies: utilizing non-synonymous variant rs117648444 to probe the role of IFN-lambda4. Gene. 2018;664:168–80.
Blach ZS, Manns M, Altraif I, Duberg AS, Muljono DH, Polaris Observatory HCV Collaborators, et al. Global prevalence and genotype distribution of hepatitis C virus infection in 2015: a modelling study. Lancet Gastroenterol Hepatol. 2017;2(3):161–76.
Chinnaswamy S, Das K, Bairagya BB, et al. Association of IL28B single nucleotide polymorphism rs8099917 with response to treatment in genotype 3 HCV-infected patients from India. Trop Gastroenterol. 2014;35(2):96–102.
EASL. EAftSotL. EASL recommendations on treatment of hepatitis C 2016. J Hepatol. 2017;66(1):153–94.
Eslam M, McLeod D, Kelaeng KS, et al. IFN-lambda3, not IFN-lambda4, likely mediates IFNL3–IFNL4 haplotype-dependent hepatic inflammation and fibrosis. Nat Genet. 2017;49(5):795–800.
Ge D, Fellay J, Thompson AJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature. 2009;461(7262):399–401.
Gonzalez-Aldaco K, Rebello Pinho JR, Roman S, et al. Association with spontaneous hepatitis C viral clearance and genetic differentiation of IL28B/IFNL4 haplotypes in populations from Mexico. PLoS ONE. 2016;11(1):e0146258.
Hadziyannis SJ, Sette H Jr, Morgan TR, et al. Peginterferon-alpha2a and ribavirin combination therapy in chronic hepatitis C: a randomized study of treatment duration and ribavirin dose. Ann Intern Med. 2004;140(5):346–55.
Hayes CN, Imamura M, Aikata H, et al. Genetics of IL28B and HCV-response to infection and treatment. Nat Rev Gastroenterol Hepatol. 2012;9(7):406–17.
Hsu CS, Hsu SJ, Liu WL, et al. Association of SCARB1 gene polymorphisms with virological response in chronic hepatitis C patients receiving pegylated interferon plus ribavirin therapy. Sci Rep. 2016;6:32303.
Huang M, Jiang JD, Peng Z. Recent advances in the anti-HCV mechanisms of interferon. Acta Pharm Sin B. 2014;4(4):241–7.
Lang Kuhs KA, Kuniholm MH, Pfeiffer RM, et al. Interferon lambda 4 genotype is not associated with recurrence of oral or genital herpes. PLoS ONE. 2015;10(10):e0138827.
Lee MH, Yang HI, Lu SN, et al. Polymorphisms near the IFNL3 gene associated with HCV RNA spontaneous clearance and hepatocellular carcinoma risk. Sci Rep. 2015;5:17030.
Liao Y, Cai B, Li Y, et al. Association of HLA-DP/DQ, STAT4 and IL-28B variants with HBV viral clearance in Tibetans and Uygurs in China. Liver Int. 2014;35(3):886–96.
Liu MQ, Zhou DJ, Wang X, et al. IFN-lambda3 inhibits HIV infection of macrophages through the JAK-STAT pathway. PLoS ONE. 2012;7(4):e35902.
NCDC. Quarterly newsletter from the National Centre for Disease Control (NCDC). NCDC India. 2014;3(1):1–12.
O’Brien TR, Kottilil S, Pfeiffer RM. IFNL4 genotype is associated with virologic relapse after 8-week treatment with sofosbuvir, velpatasvir, and voxilaprevir. Gastroenterology. 2017;153(1–2):1694–5.
O’brien TR, Prokunina-Olsson L, Donnelly RP. IFN-lambda4: the paradoxical new member of the interferon lambda family. J Interferon Cytokine Res. 2014;34(11):829–38.
Prokunina-Olsson L, Muchmore B, Tang W, et al. A variant upstream of IFNL3 (IL28B) creating a new interferon gene IFNL4 is associated with impaired clearance of hepatitis C virus. Nat Genet. 2013;45(2):164–71.
Rauch A, Kutalik Z, Descombes P, et al. Genetic variation in IL28B is associated with chronic hepatitis C and treatment failure: a genome-wide association study. Gastroenterology. 2010;138(4):1338–45 (45 e1–7).
Sarrazin C. The importance of resistance to direct antiviral drugs in HCV infection in clinical practice. J Hepatol. 2015;64:486–504.
Sheppard P, Kindsvogel W, Xu W, et al. IL-28, IL-29 and their class II cytokine receptor IL-28R. Nat Immunol. 2003;4(1):63–8.
Sivaprasad S, Rao PN, Gupta R, et al. The distribution of genotype and allelic frequency of IL28B gene polymorphism in Andhra Pradesh, India. J Clin Exp Hepatol. 2012;2(2):112–5.
Smith KR, Suppiah V, O’Connor K, et al. Identification of improved IL28B SNPs and haplotypes for prediction of drug response in treatment of hepatitis C using massively parallel sequencing in a cross-sectional European cohort. Genome Med. 2011;3(8):57.
Tanaka Y, Nishida N, Sugiyama M, et al. Genome-wide association of IL28B with response to pegylated interferon-alpha and ribavirin therapy for chronic hepatitis C. Nat Genet. 2009;41(10):1105–9.
Thomas DL, Thio CL, Martin MP, et al. Genetic variation in IL28B and spontaneous clearance of hepatitis C virus. Nature. 2009;461(7265):798–801.
Vasudevan S, Kavimandan A, Kalra N, et al. Demographic profile, host, disease and viral predictive factors of response in patients with chronic hepatitis C virus infection at a tertiary care hospital in north India. Indian J Med Res. 2016;143(3):331–40.
Zang F, Yue M, Yao Y, et al. Influence of IL28B and MxA gene polymorphisms on HCV clearance in Han Chinese population. Epidemiol Infect. 2018;146(3):379–85.
Acknowledgements
This study was supported by AIIMS intramural funding A-147.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Roy, N., Prasad, C., Kumar, A. et al. IFNL4 haplotype, linkage disequilibrium and their influence on virological response to hepatitis C virus infection in Indian population. VirusDis. 30, 344–353 (2019). https://doi.org/10.1007/s13337-019-00535-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13337-019-00535-4