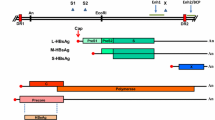
Abstract
Mutations in several regions of HCV genome are shown to correlate with response to interferon (IFN) treatment. Persistence of HCV infection and poor susceptibility to treatment might be contributed by mutations arising within HCV genome which enable the virus to escape from host immune response/IFN treatment. This study investigated mutations in core and NS5A genes of HCV from non-responder and relapser patients after treatment with Peg-IFN-α and ribavirin. Viral RNA was extracted from patient sera and core and NS5A genes were amplified by RT-PCR. Nucleotide sequences of the core and NS5A genes were determined by direct sequencing, and converted to amino acid sequences. Nucleotide and amino acid sequences in the core region, ISDR, PKRBD, and V3 regions within NS5A after treatment were highly conserved when comparing to their corresponding sequences obtained before treatment. Interestingly, when comparing the virus from relapsers to those from non-responders, the number of mutations after treatment in N-terminal region of NS5A of virus from relapsers was significantly higher than those from non-responders (P < 0.05). Amino acid mutations at the N-terminus of NS5A of the virus in relapsers might help the virus to survive and somehow relapse after the cessation of the treatment.


Similar content being viewed by others
References
Akuta N, Suzuki F, Sezaki H, et al. Association of amino acid substitution pattern in core protein of hepatitis C virus genotype 1b high viral load and non-virological response to interferon-ribavirin combination therapy. Intervirology. 2005;48(6):372–80.
Castillo I, Rodriguez-Inigo E, Bartolome J, et al. Hepatitis C virus replicates in peripheral blood mononuclear cells of patients with occult hepatitis C virus infection. Gut. 2005;54(5):682–5.
Cuevas JM, Torres-Puente M, Jimenez-Hernandez N, et al. Refined analysis of genetic variability parameters in hepatitis C virus and the ability to predict antiviral treatment response. J Viral Hepat. 2008;15(8):578–90.
Enomoto N, Sakuma I, Asahina Y, et al. Mutations in the nonstructural protein 5A gene and response to interferon in patients with chronic hepatitis C virus 1b infection. N Engl J Med. 1996;334(2):77–81.
Escudero A, Rodriguez F, Serra MA, Del Olmo JA, Montes F, Rodrigo JM. Pegylated alpha-interferon-2a plus ribavirin compared with pegylated alpha-interferon-2b plus ribavirin for initial treatment of chronic hepatitis C virus: prospective, non-randomized study. J Gastroenterol Hepatol. 2008;23(6):861–6.
Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med. 2002;347(13):975–82.
Gale MJ Jr, Korth MJ, Tang NM, et al. Evidence that hepatitis C virus resistance to interferon is mediated through repression of the PKR protein kinase by the nonstructural 5A protein. Virology. 1997;230(2):217–27.
Gale MJ Jr, Korth MJ, Katze MG. Repression of the PKR protein kinase by the hepatitis C virus NS5A protein: a potential mechanism of interferon resistance. Clin Diagn Virol. 1998;10(2–3):157–62.
Gerotto M, Dal Pero F, Sullivan DG, et al. Evidence for sequence selection within the non-structural 5A gene of hepatitis C virus type 1b during unsuccessful treatment with interferon-alpha. J Viral Hepat. 1999;6(5):367–72.
Gong GZ, Lai LY, Jiang YF, He Y, Su XS. HCV replication in PBMC and its influence on interferon therapy. World J Gastroenterol. 2003;9(2):291–4.
Hayashi K, Katano Y, Ishigami M, et al. Mutations in the core and NS5A region of hepatitis C virus genotype 1b and correlation with response to pegylated-interferon-alpha 2b and ribavirin combination therapy. J Viral Hepat. 2011;18(4):280–6.
Hofmann WP, Zeuzem S, Sarrazin C. Hepatitis C virus-related resistance mechanisms to interferon alpha-based antiviral therapy. J Clin Virol. 2005;32(2):86–91.
Januszkiewicz-Lewandowska D, Wysocki J, Pernak M, et al. Presence of hepatitis C virus (HCV)-RNA in peripheral blood mononuclear cells in HCV serum negative patients during interferon and ribavirin therapy. Jpn J Infect Dis. 2007;60(1):29–32.
Kumthip K, Pantip C, Chusri P, et al. Correlation between mutations in the core and NS5A genes of hepatitis C virus genotypes 1a, 1b, 3a, 3b, 6f and the response to pegylated interferon and ribavirin combination therapy. J Viral Hepat. 2011;18(4):e117–25.
Kumthip K, Chusri P, Pantip C, et al. Hepatitis C virus genotypes circulating in patients with chronic hepatitis C in Thailand and their responses to combined PEG-IFN and RBV therapy. J Med Virol. 2014;86(8):1360–5.
Marrone A, Sallie R. Genetic heterogeneity of hepatitis C virus. The clinical significance of genotypes and quasispecies behavior. Clin Lab Med. 1996;16(2):429–49.
Martell M, Esteban JI, Quer J, et al. Hepatitis C virus (HCV) circulates as a population of different but closely related genomes: quasispecies nature of HCV genome distribution. J Virol. 1992;66(5):3225–9.
Munoz de Rueda P, Casado J, Paton R, et al. Mutations in E2-PePHD, NS5A-PKRBD, NS5A-ISDR, and NS5A-V3 of hepatitis C virus genotype 1 and their relationships to pegylated interferon-ribavirin treatment responses. J Virol. 2008;82(13):6644–53.
Nousbaum J, Polyak SJ, Ray SC, et al. Prospective characterization of full-length hepatitis C virus NS5A quasispecies during induction and combination antiviral therapy. J Virol. 2000;74(19):9028–38.
Okanoue T, Itoh Y, Hashimoto H, et al. Predictive values of amino acid sequences of the core and NS5A regions in antiviral therapy for hepatitis C: a Japanese multi-center study. J Gastroenterol. 2009;44(9):952–63.
Pawlotsky JM, Germanidis G, Neumann AU, Pellerin M, Frainais PO, Dhumeaux D. Interferon resistance of hepatitis C virus genotype 1b: relationship to nonstructural 5A gene quasispecies mutations. J Virol. 1998;2(4):2795–805.
Reed KE, Rice CM. Overview of hepatitis C virus genome structure, polyprotein processing, and protein properties. Curr Top Microbiol Immunol. 2000;242:55–84.
Tai AW, Chung RT. Treatment failure in hepatitis C: mechanisms of non-response. J Hepatol. 2009;50(2):412–20.
Taliani G, Badolato C, Lecce R, et al. Hepatitis C virus RNA in peripheral blood mononuclear cells: relation with response to interferon treatment. J Med Virol. 1995;47(1):16–22.
Tellinghuisen TL, Marcotrigiano J, Rice CM. Structure of the zinc-binding domain of an essential component of the hepatitis C virus replicase. Nature. 2005;435(7040):374–9.
Tellinghuisen TL, Foss KL, Treadaway J. Regulation of hepatitis C virion production via phosphorylation of the NS5A protein. PLoS Pathog. 2008;4(3):e1000032.
Tong CY, Gilmore IT, Hart CA. HCV-associated liver cancer. Lancet. 1995;345(8956):1058–9.
Torres-Puente M, Cuevas JM, Jimenez-Hernandez N, et al. Genetic variability in hepatitis C virus and its role in antiviral treatment response. J Viral Hepat. 2008;15(3):188–99.
Wang JT, Sheu JC, Lin JT, Wang TH, Chen DS. Detection of replicative form of hepatitis C virus RNA in peripheral blood mononuclear cells. J Infect Dis. 1992;166(5):1167–9.
Wohnsland A, Hofmann WP, Sarrazin C. Viral determinants of resistance to treatment in patients with hepatitis C. Clin Microbiol Rev. 2007;20(1):23–38.
Acknowledgments
This study was supported by the Thailand Research Fund (TRF), the National Research Council of Thailand (NRCT), and the Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumthip, K., Chusri, P., Pantip, C. et al. Analysis of mutations in the core and NS5A genes of hepatitis C virus in non-responder and relapser patients after treatment with Peg-IFN-α and ribavirin. VirusDis. 27, 55–62 (2016). https://doi.org/10.1007/s13337-015-0300-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13337-015-0300-x