Abstract
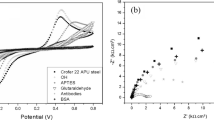
The development of a portable and inexpensive surface plasmon resonance (SPR) measurement device with the integrated biosensor for the detection of snake venom protein is presented in this paper. For the construction of the sensing element, amine coupling chemistry is used to bio-functionalize silver coated glass slide with antibodies like immunoglobulin (IgG). The immobilization of the antibody is confirmed by spectroscopic measurements like ultraviolet-visible spectroscopy (UV-Vis) and Fourier-transforms infrared spectroscopy (FTIR). The device is calibrated with the standard solution of sodium chloride and ethanol before testing venom protein samples. To investigate the bio-molecular interactions, crude venom of Indian cobra (concentration range: 0.1 mg/ml‒1.0 mg/ml) in the phosphate buffer solution (PBS) are exposed to the biosensor. The experimentally measured data indicate the shift in the plasmon resonance angle from its initial value (52°) to 54° for 0.1 mg/ml and 60° for 1.0 mg/ml protein solution.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
P. R. Edwards, A. Gill, D. V. Pollard-Knight, M. Hoare, P. E. Buckle, P. A. Lowe, et al., “Kinetics of protein-protein interactions at the surface of an optical biosensor,” Analytical Biochemistry, 1995, 231(1): 210–217.
T. Endo, K. Kerman, N. Nagatani, Y. Takamura, and E. Tamiya, “Label-free detection of peptide nucleic acid-DNA hybridization using localized surface plasmon resonance based optical biosensor,” Analytical Chemistry, 2005,77(21): 6976–6984.
J. Homola, “Present and future of surface plasmon resonance biosensors,” Analytical and Bioanalytical Chemistry, 2003, 377(3), 528–539.
H. Lijie, P. Quentin, L. Iban, Y. S. Aritz, P. Amaia, Z. Amaia, et al., “Label-free femtomolar cancer biomarker detection in human serum using grapheme coated surface plasmon resonance chip,” Biosensors and Bioelectronics, 2017,89(1): 606–611.
E. Mauriz, A. Calle, J. J. Manclús, A. Montoya, and L. M. Lechuga. “Multi-analyte SPR immunoassays for environmental biosensing of pesticides,” Analytical & Bioanalytical Chemistry, 2007, 387(4): 1449–1458.
Y. Li, X. Liu, and Z. Lin, “Recent developments and applications of surface plasmon resonance biosensors for the detection of mycotoxins in foodstuffs,” Food Chemistry, 2012,132(3): 1549–1554.
H. R. Sim, A. W. Wark, and H. J. Lee, “Attomolar detection of protein biomarkers using biofunctionalized gold nanorods with surface plasmon resonance,” Analyst, 2010, 135(10): 2528–2532.
Y. Wang, W. Knoll, and J. Dostalek, “Bacterial pathogen surface plasmon resonance biosensor advanced by long range surface plasmons and magnetic nanoparticle assays,” Analytical Chemistry, 2012,84(19): 8345–8350.
J. Homola, “Surface plasmon resonance sensors for detection of chemical and biological species,” Chemical Reviews, 2008, 108: 462–493.
K. Yokoyama, M. Oishi, and M. Oshima, “Development of an enhanced surface plasmon resonance sensor substrate by investigating a periodic nanohole array configuration,” Journal of Applied Physics, 2015, 118(2): 023101-1‒023101-7.
R. Boruah, D. Mohanta, A Choudhury, P. Nath, and G. A. Ahmed, “Surface plasmon resonance-based protein bio-sening using a Krestschmann configured double prism arrangement,” IEEE Sensors Journal, 2015, 15: 6791–6796.
Y. W. Fen, W. M. M. Yunus, Z. A. Talib, and N. A. Yusof, “Development of surface plasmon resonance sensor for determining zinc ion using novel active nanolayers as probe,” Spectrochemica Acta Part A: Molecular and Biomolecular Spectroscopy, 2015, 134: 48–52.
D. G. Hong, T. W. Kim, K. B. Kim, J. S. Yuk, and K. S. Ha, “Development of an immunosensor with angular interrogation-based SPR spectroscopy,” Measurement Science & Technology, 2007, 18(5): 1367–1371.
E. Kretschmann and H. Reather, “Radiative decay of non-radiative surface plasmons excited by light,” Zeitschrift für Naturforschung, 1968, 23: 2135–2136.
A. Otto, “Excitation of nonradiative surface plasma waves in silver by the method of frustrated total reflection,” Zeitschrift Für Physik, 1968,216(4): 398–410.
R. A. Harrison, A. Hargreaves, S. C. Wagstaff, B. Faragher, and D. G. Lalloo, “Snakebite envenoming: a disease of poverty,” PLoS Neglected Tropical Diseases, 2009, 3(12): 1–6.
J. J. Calvete, P. Juárez, and L. Sanz, “Snake venomics: strategy and applications,” Journal of Mass Spectrometry, 2010, 42(11): 1405–1414.
L. V. Dong, K. H. Eng, L. K. Quyen, and P. Gopalakrishnakone, “Optical immunoassay for snake venom detection,” Biosensors and Bioelectronics, 2004,19(10): 1285–1294.
G. D. Brand, R. Salbo, T. J. Jørgensen, C. Jr. Bloch, E. E. Boeri, C. V. Robinson, et al., “The interaction of the antitoxin DM43 with a snake venom metalloproteinase analyzed by mass spectrometry and surface plasmon resonance,” Journal of Mass Spectrometry, 2012, 47(5): 567–573.
I. Malih, M. R. A. Rusmili, T. Y. Tee, Rachid Saile, Noreddine Ghalim, and Iekhsan Othman, “Proteomic analysis of Moroccan cobra naja haje legionis venom using tandem mass spectrometry,” Journal of Proteomics, 2014,96(2): 240–252.
H. J Simon, D. E Mitchell, and J. G Watson, “Surface plasmons in silver films–a novel undergraduate experiment,” American Journal of Physics, 1975, 43(7): 630–636.
S. Szunerits, X. Castel, and R. Boukherroub, “Surface plasmon resonance investigation of silver and gold films coated with thin indium tin oxide layers: influence on stability and sensitivity,” Journal of Physics Chemistry C, 2008, 112: 15813–15817.
X. C. Yuan, B. H. Ong, Y. G. Tan, D. W. Zhang, R. Irawan, and S. C. Tjin, “Sensitivitystability-optimized surface plasmon resonance sensing with double metal layers,” Journal of Optics A Pure & Applied Optics, 2006,8(11): 959–963.
S. B. Nimse, K. Song, M. D. Sonawane, D. R. Sayyed, and T. Kim, “Immobilization techniques for microarray: challenges and applications,” Sensors, 2014, 14(12): 22208–22229.
F. Rusmini, Z. Zhong, and J. Feijen, “Protein immobilization strategies for protein biochis,” Biomacromolecules, 2007, 8(6): 1775–1789.
J. E. Lee, J. H. Seo, C. S. Kim, Y. Kwon, J. H. Ha, S. S. Choi, et al., “A comparative study on antibody immobilization strategies onto solid surface,” Korean Journal of Chemical Engineering, 2013, 30(10): 1934–1938.
C. D. Hodneland, Y. S. Lee, D. H. Min, and M. Mrksich, “Selective immobilization of proteins to self-assembled monolayers presenting active site-directed capture ligands,” Proceedings of the National Academy of Sciences, 2002, 99(8): 5048–5052.
R. F. Peters, L. Gutierrez-Rivera, S. K. Dew, and M. Stepanova, “Surface enhanced Raman spectroscopy detection of biomolecules using EBL fabricated nanostructured substrates,” Journal of Visualized Experiments Jove, 2015, 2015(97): e52712-1‒e52712-17.
L. F. Hoyt, “New table of the refractive index of pure glycerol at 20,” Industrial & Engineering Chemistry, 1934, 26(3): 329–332.
K. Aslan, C. C. Luhrs, and V. H. Pérez-Luna, “Controlled and reversible aggregation of biotinylated gold nanoparticles with streptavidin,” Journal of Physical Chemistry B, 2004, 108(40): 15631–15639.
G. Weng, J. Li, and J. Zhoa, “Enhanced resonance light scattering of antibody covalently conjugated gold nanoparticles due to antigen-antibody interaction induced aggregation,” Nanoscience and Nanotechnology Letters, 2013, 5(8): 1–7.
B. M. Amoli, S. Gumfekar, A. Hu, Y. N. Zhou, and B. Zhao, “Thiocarboxylate functionalization of silver nanoparticles: effect of chain length on the electrical conductivity of nanoparticles and their polymer composites,” Journal of Materials Chemistry, 2012, 22(37): 20048–20056.
E. Goormaghtigh, J. M. Ruysschaert, and V. Raussens, “Evaluation of the information content in infrared spectra for protein secondary structure determination,” Biophysical Journal, 2006,90(8): 2946–2957.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Choudhury, S.N., Konwar, B., Kaur, S. et al. Study on Snake Venom Protein-Antibody Interaction by Surface Plasmon Resonance Spectroscopy. Photonic Sens 8, 193–202 (2018). https://doi.org/10.1007/s13320-018-0501-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13320-018-0501-1