Abstract
Background and Objectives
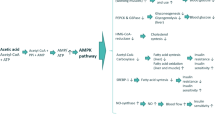
Green tea catechins were recently reported to inhibit drug transporters such as organic anion-transporting polypeptides (OATPs) and metabolic enzymes, affecting the bioavailability of many drugs. This study aimed to evaluate the clinical significance of the effects of different doses of green tea extract on the pharmacokinetic parameters of atorvastatin and to rationalize the associated interaction mechanism.
Methods
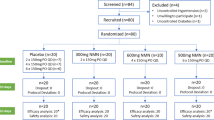
A randomized, double-blind, three-phase crossover study involving 12 healthy volunteers was performed. Participants received a single dose of atorvastatin 40 mg alone (control group), atorvastatin 40 mg plus a capsule containing 300 mg of dry green tea extract, or atorvastatin 40 mg plus a capsule containing 600 mg of dry green tea extract. Plasma samples taken from the volunteers were analyzed for atorvastatin using liquid chromatography–tandom mass spectrometry (LC/MS/MS).
Results
Compared to atorvastatin alone, the administration of 300 mg or 600 mg of the green tea extract along with atorvastatin decreased the peak plasma concentration (Cmax) of atorvastatin by 25% and 24%, respectively (P < 0.05), and the area under the plasma concentration–time curve (AUC0−∞) of atorvastatin by 24% and 22%, respectively (P < 0.05). Additionally, administration of 300 mg or 600 mg of the green tea extract increased the apparent oral clearance (CL/F) of atorvastatin by 31% and 29%, respectively. The time to Cmax (Tmax) and the elimination half-life (t1/2) of atorvastatin did not differ among the three phases. The effects of 600 mg of the green tea extract on the pharmacokinetic parameters of atorvastatin were not significantly different from the effects of 300 mg of the green tea extract.
Conclusion
Green tea extract decreases the absorption but not the elimination of atorvastatin, possibly by inhibiting OATP, albeit not in a dose-dependent manner. Coadministration of green tea extract with atorvastatin may necessitate the monitoring of the plasma concentration of atorvastatin in clinical practice.




Similar content being viewed by others
References
Lloyd-Jones D, Adams RJ, Brown TM, Carnethon M, Dai S, De Simone G, Ferguson TB, Ford E, Furie K, Gillespie C, Go A, Greenlund K, Haase N, Hailpern S, Ho PM, Howard V, Kissela B, Kittner S, Lackland D, Lisabeth L, Marelli A, McDermott MM, Meigs J, Mozaffarian D, Mussolino M, Nichol G, Roger VL, Rosamond W, Sacco R, Sorlie P, Roger VL, Thom T, Wasserthiel-Smoller S, Wong ND, Wylie-Rosett J. Heart disease and stroke statistics—2010 update: a report from the American Heart Association. Circulation. 2010;121:e46–e215.
Jain KS, Kathiravan MK, Somani RS, Shishoo CJ. The biology and chemistry of hyperlipidemia. Bioorg Med Chem. 2007;15:4674–99.
Abdelkawy K, Ibrahim M, Elashmawy N, Elsis A, Habib A. Cardiovascular risk factors among Egyptian university students with relation to residence. Int J Clin Pract. 2016;70(1):91–3.
Benjamin EJ, Virani SS, Callaway CW, Chang AR, Cheng S, Chiuve SE, Cushman M, Delling FN, Deo R, de Ferranti SD, Ferguson JF, Fornage M, Gillespie C, Isasi CR, Jimenez MC, Jordan LC, Judd SE, Lackland D, Lichtman JH, Lisabeth L, Liu S, Longenecker CT, Lutsey PL, Matchar DB, Matsushita K, Mussolino ME, Nasir K, O’Flaherty M, Palaniappan LP, Pandey DK, Reeves MJ, Ritchey MD, Rodriguez CJ, Roth GA, Rosamond WD, Sampson UKA, Satou GM, Shah SH, Spartano NL, Tirschwell DL, Tsao CW, Voeks JH, Willey JZ, Wilkins JT, Wu JHY, Alger HM, Wong SS, Muntner P; on behalf of the American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2018 update: a report from the American Heart Association. Circulation 2018;137(12):e67–e492.
El-Sisi AA, Hegazy SK, Salem KA, Abdelkawy KS. Atorvastatin improves erectile dysfunction in patients initially irresponsive to sildenafil by the activation of endothelial nitric oxide synthase. Int J Impot Res. 2013;25(4):143–8.
Koskinas K, Windecker S, Raber L. Regression of coronary atherosclerosis: current evidence and future perspectives. Trends Cardiovasc Med. 2016;26(2):150–61.
Shepherd J, Cobbe SM, Ford I, Isles CG, Lorimer AR, MacFarlane PW, McKillop JH, Packard CJ; on behalf of the West of Scotland Coronary Prevention Study Group. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. N Engl J Med. 1995; 333:1301–7.
Pan HY, DeVault AR, Wang-Iverson D, Ivashkiv E, Swanson BN, Sugerman AA. Comparative pharmacokinetics and pharmacodynamics of pravastatin and lovastatin. J Clin Pharmacol. 1990;30:1128–35.
Radulovic L, Cilla D, Posvar E, Sedman A, Whitfield L. Effect of food on the bioavailability of atorvastatin, an HMG-CoA reductase inhibitor. J Clin Pharmacol. 1995;35:990–4.
Princen HM, van Duyvenvoorde W, Buytenhek R, Blonk C, Tijburg LB, Langius JA, Meinders AE, Pijl H. No effect of consumption of green and black tea on plasma lipid and antioxidant levels and on LDL oxidation in smokers. Arterioscler Thromb Vasc Biol. 1998;18:833–41.
Warwick MJ, Dane AL, Raza A, Schneck DW. Single and multiple-dose pharmacokinetics and safety of the new HMGCoA reductase inhibitor ZD4522. Atherosclerosis. 2000;151(1):39.
Fujino H, Yamada I, Kojima J, Hirano M, Matsumoto H, Yoneda M. Studies on the metabolic fate of NK-104, a new inhibitor of HMG-CoA reductase (5): in vitro metabolism and plasma protein binding in animals and human. Xeno Metab Disp. 1999;14:415–24.
Muscari A, Puddu GM, Puddu P. Lipid-lowering drugs: are adverse effects predictable and reversible? Cardiology. 2002;97:115–21.
Knopp R. Drug treatment of lipid disorders. N Engl J Med. 1999;341:498–511.
Graham HN. Green tea composition, consumption and polyphenol chemistry. Prev Med. 1992;21:369–72.
Kuriyama S, Shimazu T, Ohmori K, Kikuchi N, Nakaya N, Nishino Y, Tsubono Y, Tsuji I. Green tea consumption and mortality due to cardiovascular disease, cancer, and all causes in Japan: the Ohsaki Study. JAMA. 2006;296:1255–65.
Stangl V, Dreger H, Stangl K, Lorenz M. Molecular targets of tea polyphenols in the cardiovascular system. Cardiovasc Res. 2007;73:348–58.
Gruenwalkd T, Brendler T, Jaenicke C. PDR for herbal medicines. J Med Econ. 2000;1:369–72.
Lins RL, Matthys KE, Verpooten GA, Peeters PC, Dratwa M, Stolear JC, Lameire NH. Pharmacokinetics of atorvastatin and its metabolites after single and multiple dosing in hypercholesterolaemic haemodialysis patients. Nephrol Dial Transplant. 2003;18(5):967–76.
Clarke JD, Cherrington NJ. Genetics or environment in drug transport: the case of organic anion transporting polypeptides and adverse drug reactions. Expert Opin Drug Metab Toxicol. 2012;8:349–60.
Roth M, Obaidat A, Hagenbuch B. OATPs, OATs and OCTs: the organic anion and cation transporters of the SLCO and SLC22A gene superfamilies. Br J Pharmacol. 2012;165:1260–87.
Zhou F, Zheng J, Zhu L, Jodal A, Cui P, Wong M, Gurney H, Church WB, Murray M. Functional analysis of novel polymorphisms in the human SLCO1A2 gene that encodes the transporter OATP1A2. AAPS J. 2013;15:1099–108.
Roth M, Timmermann BN, Hagenbuch B. Interactions of green tea catechins with organic anion-transporting polypeptides. Drug Metab Dispos. 2011;39:920–6.
Abdlekawy K, Donia A, Elbarbry F. Effects of grapefruit and pomegranate juices on the pharmacokinetic properties of dapoxetine and midazolam in healthy subjects. Eur J Drug Metab Pharmacokinet. 2016;42(3):397–405.
Kim TE, Ha N, Kim Y, Kim H, Lee JW, Jeon JY, Kim MG. Effect of epigallocatechin-3-gallate, major ingredient of green tea, on the pharmacokinetics of rosuvastatin in healthy volunteers. Drug Des Devel Ther. 2017;11:1409–16.
Wang J, Luzum JA, Phelps MA, Kitzmiller JP. Liquid chromatography-tandem mass spectrometry assay for the simultaneous quantification of simvastatin, lovastatin, atorvastatin, and their major metabolites in human plasma. J Chromatogr B. 2015;983–984:18–25.
Morse BL, Alberts JJ, Posada MM, Rehmel J, Kolur A, Tham LS, Loghin C, Hillgren KM, Hall SD, Dickinson GL. Physiologically-based pharmacokinetic modeling of atorvastatin incorporating delayed gastric emptying and acid-to-lactone conversion. CPT Pharmacometrics Syst Pharmacol. 2019;8(9):664–75.
Lilja JJ, Kivistö KT, Neuvonen PJ. Grapefruit juice increases serum concentrations of atorvastatin and has no effect on pravastatin. Clin Pharmacol Ther. 1999;66(2):118–27.
Backman JT, Luurila H, Neuvonen M, Neuvonen PJ. Rifampin markedly decreases and gemfibrozil increases the plasma concentrations of atorvastatin and its metabolites. Clin Pharmacol Ther. 2005;78(2):154–67.
Lau YY, Huang Y, Frassetto L, Benet LZ. Effect of OATP1B transporter inhibition on the pharmacokinetics of atorvastatin in healthy volunteers. Clin Pharmacol Ther. 2007;81(2):194–204.
Pasanen MK, Fredrikson H, Neuvonen PJ, Niemi M. Different effects of SLCO1B1 polymorphism on the pharmacokinetics of atorvastatin and rosuvastatin. Clin Pharmacol Ther. 2007;82(6):726–33.
Guo CX, Pei Q, Yin JY, Peng XD, Zhou BT, Zhao YC, Wu LX, Meng XG, Wang G, Li Q, Ouyang DS, Liu ZQ, Zhang W, Zhou HH. Effects of Ginkgo biloba extracts on pharmacokinetics and efficacy of atorvastatin based on plasma indices. Xenobiotica. 2012;42(8):784–90.
Reddy G, Reddy A, Rao G, Kumar M. Pharmacokinetic interaction of garlic and atorvastatin in dyslipidemic rats. Indian J Pharmacol. 2012;44(2):246–52.
Yamazaki T, Desai A, Goldwater R, Han D, Lasseter KC, Howieson C, Akhtar S, Kowalski D, Lademacher C, Rammelsberg D, Townsend R. Pharmacokinetic interactions between isavuconazole and the drug transporter substrates atorvastatin, digoxin, metformin, and methotrexate in healthy subjects. Clin Pharmacol Drug Dev. 2017;6(1):66–75.
Rajput T, Naveed A, Farooqi Z, Khan S. Effects of two functionally important SLCO1B1 gene polymorphisms on pharmacokinetics of atorvastatin. Pak J Pharm Sci. 2017;30(4):1363–70.
Golden E, Lam P, Kardosh A, Gaffney K, Cadenas E, Louie S, Petasis N, Chen T, Schönthal A. Green tea polyphenols block the anticancer effects of bortezomib and other boronic acid-based proteasome inhibitors. Blood. 2009;113(23):5927–37.
Jang EH, Choi JY, Park CS, Lee SK, Kim CE, Park HJ, Kang JS, Lee JW, Kang JH. Effects of green tea extract administration on the pharmacokinetics of clozapine in rats. J Pharm Pharmacol. 2005;57(3):311–6.
Ge J, Tan BX, Chen Y, Yang L, Peng XC, Li HZ, Lin HJ, Zhao Y, Wei M, Cheng K, Li LH, Dong H, Gao F, He JP, Wu Y, Qiu M, Zhao YL, Su JM, Hou JM, Liu JY. Interaction of green tea polyphenol epigallocatechin-3-gallate with sunitinib: potential risk of diminished sunitinib bioavailability. J Mol Med. 2011;89(6):595–602.
Misaka S, Miyazaki N, Fukushima T, Yamada S, Kimura J. Effects of green tea extract and (–)–epigallocatechin-3-gallate on pharmacokinetics of nadolol in rats. Phytomedicine. 2013;20(14):1247–50.
Knop J, Misaka S, Singer K, Hoier E, Müller F, Glaeser H, König J, Fromm MF. Inhibitory effects of green tea and (−)-epigallocatechin gallate on transport by OATP1B1, OATP1B3, OCT1, OCT2, MATE1, MATE2-K and P-glycoprotein. PLoS ONE. 2015;10(10):1–13.
Kim TE, Shin KH, Park JE, Kim MG, Yun YM, Choi DH, Kwon KJ, Lee J. Effect of green tea catechins on the pharmacokinetics of digoxin in humans. Drug Des Devel Ther. 2018;12:2139–47.
Abe O, Ono T, Sato H, Müller F, Ogata H, Miura I, Shikama Y, Yabe H, Onoue S, Fromm MF, Kimura J, Misaka S. Role of (−)-epigallocatechin gallate in the pharmacokinetic interaction between nadolol and green tea in healthy volunteers. Eur J Clin Pharmacol. 2018;74(6):775–83.
Misaka S, Abe O, Sato H, Ono T, Shikama Y, Onoue S, Yabe H, Kimura J. Lack of pharmacokinetic interaction between fluvastatin and green tea in healthy volunteers. Eur J Clin Pharmacol. 2018;74(5):601–9.
Misaka S, Kawabe K, Onoue S, Werba JP, Giroli M, Watanabe H, Yamada S. Green tea extract affects the cytochrome P450 3A activity and pharmacokinetics of simvastatin in rats. Drug Metab Pharmacokinet. 2013;28(6):514–8.
Abdelkawy K, Donia A, Abdallah M. Pharmacokinetics interaction of dapoxetine with different doses of green tea extract in male healthy volunteers using midazolam as CYP3A4 enzyme probe. J Appl Pharmaceut Sci. 2015;5(12):001–7.
Acknowledgements
We thank the members of the Clinical Pharmacy Department and the Pharmaceutical Technology Research Center in the Faculty of Pharmacy, Tanta University, Egypt.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported and funded by the Pharmaceutical Technology Research Center in the Faculty of Pharmacy, Tanta University, Egypt.
Conflict of interest
Khaled Abdelkawy, Reham Abdelaziz, Ahmed Abdelmageed, Ahmed Donia, and Noha El-Khodary declare that they have no conflict of interest relating to the content of this article.
Ethical approval
The study protocol was approved by the ethics committee of Tanta University in accordance with the Declaration of Helsinki and its amendments.
Informed consent
All subjects provided written informed consent before participation.
Rights and permissions
About this article
Cite this article
Abdelkawy, K.S., Abdelaziz, R.M., Abdelmageed, A.M. et al. Effects of Green Tea Extract on Atorvastatin Pharmacokinetics in Healthy Volunteers. Eur J Drug Metab Pharmacokinet 45, 351–360 (2020). https://doi.org/10.1007/s13318-020-00608-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13318-020-00608-6