Abstract
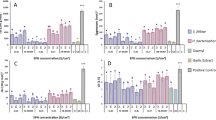
Trichoderma strains were tested for their ability to reduce the severity of root-knot nematode (RKN), Meloidogyne hapla galling on tomato roots. In initial glasshouse trials, under low levels of RKN infestation (1.1 eggs mL soil−1), Trichoderma strains applied as a conidial suspension reduced RKN-induced root galling by 42–88 %. When the nematode infestation level was increased (6.2 eggs mL soil−1), these isolates were only able to suppress galling by 21–32 %. At the highest infestation rate trialled (30 eggs mL soil−1), Trichoderma strains were unable to suppress galling. The chemical control oxamyl (Vydate®) (only evaluated at the highest infestation rate) was 98 % effective. The Trichoderma strains were not effective against RKN when treatments of both organisms were applied pre-planting. However, Trichoderma strains were able to reduce symptoms of RKN galling when applied either at seeding prior to nematode inoculation or post seedling emergence together with the RKN inoculation. These results demonstrate that selected Trichoderma strains have biocontrol potential against RKN at low-medium infestation but fail when the infestation level is rated high.
Similar content being viewed by others
References
Affokpon A, Coyne DL, Htay CC, Agbede RD, Lawouin L, Coosemans J (2011) Biocontrol potential of native Trichoderma isolates against root-knot nematodes in West African vegetable production systems. Soil Biol Biochem 43:600–608. doi:10.1016/j.soilbio.2010.11.029
Barker K, Koenning S (1998) Developing sustainable systems for nematode management. Annu Rev Phytopathol 36:165–205
Barker KR, Shoemaker PB, Nelson LA (1976) Relationships of initial population densities of Meloidogyne incognita and M. hapla to yield of tomato. J Nematol 8:232–239
Bridge J, Page SLJ (1980) Estimation of root-knot nematode infestation levels on roots using a rating chart. Trop Pest Manage 26:296–298
Brodie BB, Evans K, Franco J (1993) Nematode parasites of potatoes. In: Evans K, Trudgill DL, Webster JM (eds) Plant Parasitic Nematodes in Temperate Agriculture. Wallingford, UK, p. 648
Dababat A, Sikora RA (2007) Use of Trichoderma harzianum and Trichoderma viride for the biological control of Meloidogyne incognita on tomato. Jordan J Agric Sci 3:297–309
Dodd-Wilson SL (2006) Biochemical and molecular characterisation of Trichoderma species. University of Auckland, Auckland
Fravel DR (2005) Commercialisation and implementation of biocontrol. Annu Rev Phytopathol 43:337–359
Harman G (2006) Overview of mechanisms and uses of Trichoderma spp. Phytopathology 96:190–194
Harman G, Jin X, Stasz T, Peruzzotti G, Leopold A, Taylor A (1991) Production of conidial biomass of Trichoderma harzianum for biological control. Biol Control 1:23–28
Harman G, Howell C, Viterbo A, Chet I, Lorito M (2004) Trichoderma species - opportunisitc, avirulent plant symbionts. Nat Rev Microbiol 2:43–56
Hill RA, Ambrose A, Sajali NA, Yatim M, Valdez RB, Adbayani F, Bungang J, Minchin R, Stewart A (2010) Bioprotection of Acacia mangium using Trichoderma in Malaysia. In: Zydenbos SM, Jackson TA (eds) Microbial Products: Exploiting Microbial Diversity for Sustainable Plant Protection. Proceedings of an International Symposium held 10 August 2009, Dunedin, New Zealand. New Zealand Plant Protection Society, Lincoln, pp. 51–56
Howell CR (2003) Mechanisms employed by Trichoderma species in the biological control of plant diseases: the history and evolution of current concepts. Plant Dis 87:4–10
Hussey R, Barker K (1973) A comparison of methods of collecting inocula of Meloidogyne spp., Including a new technique. Plant Dis Rep 57:1025–1028
Kerry B (2000) Rhizosphere interactions and the exploitation of microbial agents for the biological control of plant parasitic nematodes. Annu Rev Phytopathol 38:423–441
Kleifeld O, Chet I (1992) Trichoderma harzianum - interaction with plants and effect on growth response. Plant Soil 144:267–272
Melakeberhan H (2004) Physiological interactions between nematodes and their plant hosts. In: Chen S, Chen Z, Dickson D (eds) Nematology Advances and Perspectives: Nematode Management and Utilisation, vol 2. C.A.B. International, Beijing, p. 608
Meyer SLF (2003) United states department of agriculture - Agricultural Research Service research programs on microbes for management of plant-parasitic nematodes. Pest Manag Sci 59:665–670. doi:10.1002/ps.708
Olthof TH, Potter JW (1977) Effects of population densities of Meloidogyne hapla on growth and yield of tomato. J Nematol 9:296–300
Ozbay N, Newman S, Brown W (2004) The effect of Trichoderma harzianum strains on the growth of tomato seedlings. In: Vanachter A (ed) Proceedings of the 26th IHC - Managing soil-borne pathogens. Acta Hortic 635:131–135
Potter JW, Olthof JW (1993) Nematode pests of vegetable crops. In: Evans K, Trudgill DL, Webster JM (eds) Plant Parasitic Nematodes in Temperate Agriculture. Wallingford, UK, p. 648
Radwan MA, Farrag SAA, Abu-Elamayem MM, Ahmed NS (2012) Biological control of the root-knot nematode, Meloidogyne incognita on tomato using bioproducts of microbial origin. Appl Soil Ecol 56:58–62. doi:10.1016/j.apsoil.2012.02.008
Reino JL, Guerrero RF, Hernandex-Galan R, Collado IG (2008) Secondary metabolites from species of the biocontrol agent Trichoderma. Phytochem Rev 7:89–123
Sahebani N, Hadavi N (2008) Biological control the root-knot nematode Meloidogyne javanica by Trichoderma harzianum. Soil Biol Biochem 40:2016–2020
Samuels G (2003) Trichoderma in biological control: a taxonomist reports. Biocontrol News Inform, vol 25.
Seinhorst J (1998) The common relation between population density and plant weight in pot and microplot experiments with various nematode plant combinations. Fundam Appl Nematol 21:459–468
Sharon E, Bar-Eyal M, Chet I, Herrera-Estrella A, Kleifeld O, Spiegel Y (2001) Biological control of the root-knot nematode Meloidogyne javanica by Trichoderma harzianum. Phytopathology 91:687–693. doi:10.1094/phyto.2001.91.7.687
Sharon E, Chet I, Viterbo A, Bar-Eyal M, Nagan H, Samuels G, Spiegel Y (2007) Parasitism of Trichoderma on Meloidogyne javanica and the role of the gelatinous matrix. Eur J Plant Pathol 118:247–258
Spiegel Y, Chet I (1998) Evaluation of Trichoderma spp. as a biocontrol agent against soilborne fungi and plant-parasitic nematodes in Israel. Integr Pest Manag Rev 3:169–175. doi:10.1023/A:1009625831128
Stirling G, Kopittke R (2000) Sampling procedures and damage thresholds for root-knot nematode (Meloidogyne javanica) on pineapple. Aust J Exper Agric 40:1003–1010
Verma M, Brar S, Tyagi R, Surampalli R, Valero J (2007) Antagonistic fungi, Trichoderma spp.: Panoply of biological control. Biochem Eng J 37:1–20
VSN International 2013. Genstat for Windows’. 16th edn (VSN International: Hemel Hemstead, UK)
Acknowledgments
This paper is dedicated to the memory and work of Dr. John Marshall (Nematologist) and Annabell Clouston (Research Technician).
Funding from the Ministry of Business, Innovation and Employment (MBIE) programme PROJ- 12529-LNNR-AGR: Microbial Products for International Markets; Development and the Pre-seed Accelerator Fund (PSAF). Dave Saville for statistical advice. Janine Johnson for helpful comments on the manuscript and editorial advice.
Author information
Authors and Affiliations
Corresponding author
Additional information
Annabel Clouston deceased March 2016 and John Marshall deceased January 2014.
Rights and permissions
About this article
Cite this article
Braithwaite, M., Clouston, A., Minchin, R. et al. The density-dependent effect of initial nematode population levels on the efficacy of Trichoderma as a bio-nematicide against Meloidogyne hapla on tomato. Australasian Plant Pathol. 45, 473–479 (2016). https://doi.org/10.1007/s13313-016-0432-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-016-0432-5