Abstract
To evaluate the effect of patient blood management (PBM) since its introduction, we analyzed the need for transfusion and the outcomes in patients undergoing abdominal surgery for different types of tumor pre- and post-PBM. Patients undergoing elective gastric, liver, pancreatic, and colorectal surgery between 2017 and 2020 were included. The implementation of the PBM program was completed on May 1, 2018. The patients were grouped as follows: those who underwent surgery before the implementation of the program (pre-PBM) versus after the implementation (post-PBM). A total of 1302 patients were included in the analysis (445 pre-PBM vs. 857 post-PBM). The number of transfused patients per year decreased significantly after the introduction of PBM. A strong tendency for a decreased incidence of transfusion was evident in gastric and pancreatic surgery and a similar decrease was statistically significant in liver surgery. With regard to gastric surgery, a single-unit transfusion scheme was used more frequently in the post-PBM group (7.7% vs. 55% after PBM; p = 0.049); this was similar in liver surgery (17.6% vs. 58.3% after PBM; p = 0.04). Within the subgroup of patients undergoing liver surgery, a significant reduction in the use of blood transfusion (20.5% vs. 6.7%; p = 0.002) and a decrease in the Hb trigger for transfusion (8.5, 8.2–9.5 vs. 8.2, 7.7–8.4 g/dl; p = 0.039) was reported after the PBM introduction. After the implementation of a PBM protocol, a significant reduction in the number of patients receiving blood transfusion was demonstrated, with a strong tendency to minimize the use of blood products for most types of oncologic surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Blood transfusions are commonly used in general, cardiac, and orthopedic surgery [1,2,3].
On a large scale, approximately 30% of patients undergoing major abdominal surgery require blood transfusion, with those undergoing hepatic and pancreatic resections at higher risk of receiving blood products during hospitalization [2].
Transfusion may prevent severe complications during uncontrolled bleeding and is considered a life-saving treatment; however, it has negative impacts on the post-operative morbidity and long-term outcomes in oncologic surgical patients [4,5,6,7].
By analyzing more than 1200 patients who underwent resection for hepatocellular carcinoma (HCC) or colorectal metastases, we demonstrated that blood transfusion had a negative impact on mortality and that the number of units of packed red cells administered is strongly related to early outcomes; patients receiving two or more units of packed red cells demonstrated a significantly increased risk of post-operative morbidity and mortality [8]. Similarly, Gruttadauria et al. [9], analyzing a cohort of liver resections for colorectal metastases, found that patients who received intraoperative blood transfusions were at higher risk to experience major postoperative complications and, consequently, a longer length of hospital stay.
Patient blood management (PBM) has been introduced in the last decade to reduce the use of blood products and improve tolerance to anemia [10,11,12,13,14]. Most data regarding the relationship between the implementation of PBM and the reduction in the consumption of blood products come from North America [14,15,16,17].
To evaluate the effect of PBM since its introduction in our Center in 2018, we analyzed the need for transfusion and the post-operative outcomes in the last 4 years in patients undergoing abdominal surgery for different types of tumor pre- and post-PBM.
Patients and methods
Patients undergoing elective gastric, liver, pancreatic, and colorectal surgery at Morgagni-Pierantoni Hospital of Forlì from 2017 to 2020 were included in the analysis.
The implementation of the PBM program was completed on May 1, 2018. The patients were grouped as follows: those who underwent surgery before the implementation of the program (from January 1, 2017 to April 30, 2018; pre-PBM) versus after the implementation (from May 1, 2018 to December 31, 2020; post-PBM).
For each patient record, sociodemographic characteristics (age and sex) and illness-related variables (American Society of Anesthesiologists (ASA) score, diagnosis of malignancy) were recorded.
Preoperative Hemoglobin (Hb), platelets count, and INR was also recorded. The type of surgery, the approach (open versus minimally invasive) and postoperative complications were also collected.
Length of hospital stay was defined as the interval between the day of surgery and the day of discharge.
The hemoglobin trigger was defined as the value of hemoglobin below which a transfusion was performed.
Blood management protocol
In 2017, a multidisciplinary working group was established to promote the most appropriate transfusion practices in accordance with the PBM approaches. Surgeons, anesthesiologists, and specialists in transfusion medicine were involved in this project. A team coordinator was also identified and formally commissioned by hospital management.
The constructed protocol was based on the three pillars of PBM [10, 18] and the blood component transfusion policy was defined in compliance with the current transfusion guidelines for red blood cells, platelets, and plasma.
As shown in Table 1, the protocol included pre-operative, intra-operative, and post-operative precautions to prepare the patient to the procedure and to prevent and manage potential blood losses.
The restrictive trigger and single-unit blood transfusion practice were part of the protocol to correct peri-operative anemia. Different triggers were modulated and used according to patients’ conditions and comorbidities (Fig. 1). All patients were reassessed before a second unit was requested.
Educational programs and group discussions with surgeons and anesthesiologists were conducted, as were periodic audits of transfusion practices.
Statistical analysis
Continuous data were shown as median and interquartile range (IQR) and comparisons were performed using the Mann–Whitney U test. Fisher exact test was used to compare categorical variables which were presented as frequencies and percentages. Analyses were performed with statistical software for biomedical research (MedCalc® for Windows®, version 10.2.0.0; MedCalc Software, Ostend, Belgium).
Results
A total of 1302 patients were included in the analysis. In particular, 445 patients were in the pre-PBM group while 857 were in the post-PBM group.
As shown in Table 2, all baseline patients’ characteristics were similar between the groups with the exception of the rate of minimally invasive approaches which was higher in the post-PBM group (140, 31.5% before PBM versus 326, 38% after PBM introduction; p = 0.025).
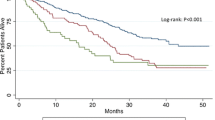
The median preoperative Hb was higher in the patients who did not have postoperative transfusions (13.2, 11.9–14.3 versus 10.4, 9.4–11.7; p < 0.0001) and this was seen also within the pre-PBM era (13.2, 12–14.2 versus 10.3, 9.4–11.9; p < 0.0001) and in the post-PBM groups (13.1, 11.9–14.3 versus 10.4, 9.4–11.6; p < 0.001). The rate of transfused patients according to preoperative Hb value (less or more than 13 gr/dl) is reported in Fig. 2
The number of transfused patients per year decreased significantly after the introduction of PBM, as shown in Fig. 3a. The rate of transfusion according to the type of surgery in each group is shown in Fig. 3b. As shown in Fig. 3b and Table 3, a strong tendency for a decreased incidence of transfusion was evident in gastric and pancreatic surgery and a similar decrease was statistically significant in liver surgery; there was no difference between the two groups of patients requiring colorectal surgery.
The post-operative outcomes are shown in Table 3. The post-operative complications according to surgery type were similar between the groups.
With regard to gastric surgery, a single-unit transfusion scheme was used more frequently in the post-PBM group (1, 7.7% vs. 9, 55% after PBM; p = 0.049); this was similar in liver surgery (3, 17.6% vs. 7, 58.3% after PBM; p = 0.04). A tendency was observed even for pancreatic surgery, although it did not reach statistical significance. The trend in the rate of adoption of the single-transfusion scheme is shown in Fig. 4.
Within the subgroup of patients undergoing liver surgery, a significant reduction in the use of blood transfusion (20.5% vs. 6.7%) and a decrease in the Hb trigger for transfusion (8.5, 8.2–9.5 vs. 8.2, 7.7–8.4 g/dl; p = 0.039) was reported after the implementation of the PBM protocol; similarly, a significant decrease in post-operative stay was reported.
Discussion
Blood transfusion is commonly used as a life-saving treatment in surgical patients, although several negative effects on the early and long-term outcomes have been demonstrated [6, 8, 19].
In the last 10 years, several guidelines and recommendations have been reported to improve the pre-, intra-, and post-operative management of patients with the aim of reducing the use of blood products [12, 14, 20].
In 2015 the Italian Ministry of Health published a recommendation to apply the PBM protocol in all Italian hospitals [21].
In our City Hospital, which recently became an HPB hub [22] for the region, a PBM program was introduced in 2018. We have demonstrated a progressive and significant reduction in blood transfusion since the implementation of this protocol, including a more restrictive use of peri-operative transfusion and the use of single units of packed red cells. In particular, we reported a relevant reduction in the use of packed red cells in gastric, pancreatic, and liver surgery. In contrast, we did not identify any difference in transfused patients treated for colorectal tumors between the two periods. However, the incidence of transfused patients after colorectal surgery was low, even in the first period (only 9%); therefore, demonstration of a possible impact of the new policy was difficult. In colorectal surgery, only 10–15% of patients required blood products [23, 24]; however, in a large series such as in our experience, PBM appeared not to reduce this percentage significantly.
In a large series from the national database, 30% of patients requiring hepatic and pancreatic surgery were transfused [1, 2]. However, in North America, several multicenter studies have demonstrated the possibility of reducing the use of blood products after different types of surgery, even in hepatobiliary and pancreatic surgery [15, 20, 25, 26]. We have demonstrated that in both gastric and liver surgery, only 13.5% and 6.7% of patients, respectively, required intra- or post-operative blood transfusion with the precise application of PBM. Even in pancreatic surgery, after PBM implementation, only 19% of resected patients required transfusion. Similar results have been reported in a recent large study using the US nationwide database; even after liver resection, 30% of patients received blood transfusion in the most recent period [17].
We noted a significant increase in post-operative complications in patients receiving two or more transfusions compared to those requiring only one packed red cell unit [8]. Other authors have reported similar data [19]. Thus, in the last 5 years, a strong recommendation to restrict the use of transfusion has been reported. The strategy of transfusing only one unit of packed red cells increased in all types of surgery without any adverse effects on the post-operative outcomes; this tendency reached a significant difference, particularly in patients requiring gastric and liver surgery.
Recently, a tendency to restrict the use of blood transfusion with a trigger point of hemoglobin of only 7 g/dl has been reported [12, 15]. However, concerns exist that an overly restrictive transfusion strategy may impact post-operative outcomes and increase cardiac complications [27, 28]. Thus, one of the most recent recommendations is to use 8 g/dl as a trigger cut-off for blood transfusion [29]; this has also been included in our protocol in common clinical conditions (e.g., coronary artery disease…) since 2018. In this series, we have reported a median trigger point for blood transfusion of approximately 8.5 g/dl; however, we demonstrated a significant reduction in the trigger point for transfusion after PBM implementation in patients requiring liver surgery (from 8.5 to 8.1 g/dl from the first to the second period).
In the last 20 years, minimally invasive surgery has increasingly been used. Both laparoscopic and robotic surgery have demonstrated several advantages over “open” surgery; among these, the reduction of the intraoperative blood loss appears consistent [30,31,32,33,34]. In the present series, a significant increase in the use of the minimally invasive approach was evident, from 31 to 38% in the first and second periods, respectively. This strategy, which is also recommended in the PBM protocol, may be useful in reducing the use of transfusions.
This study has a few limitations mainly linked to its retrospective design. As such, few variables about specific comorbidities and detailed postoperative complications which could have potentially helped in analyzing the influence of the PBM protocol could not be retrieved. In addition, the number of patients who were transfused per procedure type was relatively low and this might have affected the statistical analysis in the attempt to highlight the impact of the use of the PBM protocol. To overcome these limitations, we believe a multicenter prospective trial may be helpful in confirming the utility of the PBM protocol in limiting the use of red blood cells in patients undergoing HPB and gastrointestinal surgery.
In conclusion, after the implementation of a PBM protocol in a City Hospital, a significant reduction in the number of patients receiving blood transfusion was demonstrated, with a strong tendency to minimize the use of blood products for most types of oncologic surgery. Additionally, a tendency to reduce the trigger cut-off for transfusion and to increase the frequency of use of only one packed red cell unit was reported.
References
Aquina CT, Blumberg N, Probst CP, Becerra AZ, Hensley BJ, Iannuzzi JC, Gonzalez MG, Deeb AP, Noyes K, Monson JR, Fleming FJ (2015) Significant variation in blood transfusion practice persists following upper GI cancer resection. J Gastrointest Surg 19:1927–1937. https://doi.org/10.1007/s11605-015-2903-3
Ecker BL, Simmons KD, Zaheer S, Poe SL, Bartlett EK, Drebin JA, Fraker DL, Kelz RR, Roses RE, Karakousis GC (2016) Blood transfusion in major abdominal surgery for malignant tumors: a trend analysis using the national surgical quality improvement program. JAMA Surg 151:518–525. https://doi.org/10.1001/jamasurg.2015.5094
Spahn DR (2010) Anemia and patient blood management in hip and knee surgery: a systematic review of the literature. Anesthesiology 113:482–495. https://doi.org/10.1097/ALN.0b013e3181e08e97
Busch OR, Hop WC, Hoynck van Papendrecht MA, Marquet RL, Jeekel J (1993) Blood transfusions and prognosis in colorectal cancer. N Engl J Med 328:1372–1376. https://doi.org/10.1056/nejm199305133281902
Goodnough LT, Maggio P, Hadhazy E, Shieh L, Hernandez-Boussard T, Khari P, Shah N (2014) Restrictive blood transfusion practices are associated with improved patient outcomes. Transfusion 54(10 Pt 2):2753–2759. https://doi.org/10.1111/trf.12723
Kooby DA, Stockman J, Ben-Porat L, Gonen M, Jarnagin WR, Dematteo RP, Tuorto S, Wuest D, Blumgart LH, Fong Y (2003) Influence of transfusions on perioperative and long-term outcome in patients following hepatic resection for colorectal metastases. Ann Surg 237:860–869. https://doi.org/10.1097/01.sla.0000072371.95588.da (discussion 869–870)
Carson JL, Stanworth SJ, Dennis JA, Trivella M, Roubinian N, Fergusson DA, Triulzi D, Dorée C, Hébert PC (2021) Transfusion thresholds for guiding red blood cell transfusion. Cochrane Database Syst Rev 12(12):CD002042. https://doi.org/10.1002/14651858.CD002042.pub5
Ercolani G, Ravaioli M, Grazi GL, Cescon M, Del Gaudio M, Vetrone G, Zanello M, Pinna AD (2008) Use of vascular clamping in hepatic surgery: lessons learned from 1260 liver resections. Arch Surg 143:380–387. https://doi.org/10.1001/archsurg.143.4.380
Gruttadauria S, Saint Georges Chaumet M, Pagano D, Marsh JW, Bartoccelli C, Cintorino D, Arcadipane A, Vizzini G, Spada M, Gridelli B (2011) Impact of blood transfusion on early outcome of liver resection for colorectal hepatic metastases. J Surg Oncol 103:140–147. https://doi.org/10.1002/jso.21796
Clevenger B, Mallett SV, Klein AA, Richards T (2015) Patient blood management to reduce surgical risk. Br J Surg 102:1325–1337. https://doi.org/10.1002/bjs.9898
Meybohm P, Froessler B, Goodnough LT, Klein AA, Muñoz M, Murphy MF, Richards T, Shander A, Spahn DR, Zacharowski K (2017) Simplified international recommendations for the implementation of patient blood management (SIR4PBM). Perioper Med (Lond) 6:5. https://doi.org/10.1186/s13741-017-0061-8
Mueller MM, Van Remoortel H, Meybohm P, Aranko K, Aubron C, Burger R, Carson JL, Cichutek K, De Buck E, Devine D, Fergusson D, Folléa G, French C, Frey KP, Gammon R, Levy JH, Murphy MF, Ozier Y, Pavenski K, So-Osman C, Tiberghien P, Volmink J, Waters JH, Wood EM, Seifried E (2019) Patient blood management: recommendations from the 2018 frankfurt consensus conference. JAMA 321:983–997. https://doi.org/10.1001/jama.2019.0554
Shah A, Palmer AJR, Klein AA (2020) Strategies to minimize intraoperative blood loss during major surgery. Br J Surg 107:e26–e38. https://doi.org/10.1002/bjs.11393
Baker L, Park L, Gilbert R, Ahn H, Martel A, Lenet T, Davis A, McIsaac DI, Tinmouth A, Fergusson DA, Martel G (2021) Intraoperative red blood cell transfusion decision-making: a systematic review of guidelines. Ann Surg 274:86–96. https://doi.org/10.1097/sla.0000000000004710
Carson JL, Sieber F, Cook DR, Hoover DR, Noveck H, Chaitman BR, Fleisher L, Beaupre L, Macaulay W, Rhoads GG, Paris B, Zagorin A, Sanders DW, Zakriya KJ, Magaziner J (2015) Liberal versus restrictive blood transfusion strategy: 3-year survival and cause of death results from the FOCUS randomised controlled trial. Lancet 385:1183–1189. https://doi.org/10.1016/s0140-6736(14)62286-8
Verdecchia NM, Wisniewski MK, Waters JH, Triulzi DJ, Alarcon LH, Yazer MH (2016) Changes in blood product utilization in a seven-hospital system after the implementation of a patient blood management program: a 9-year follow-up. Hematology 21:490–499. https://doi.org/10.1080/10245332.2015.1112496
Hamilton CM, Davenport DL, Bernard AC (2021) Demonstration of a US nationwide reduction in transfusion in general surgery and a review of published transfusion reduction methodologies. Transfusion 61(11):3119–3128. https://doi.org/10.1111/trf.16677
Butcher A, Richards T (2018) Cornerstones of patient blood management in surgery. Transfus Med 28:150–157. https://doi.org/10.1111/tme.12476
Ejaz A, Spolverato G, Kim Y, Margonis GA, Gupta R, Amini N, Frank SM, Pawlik TM (2015) Impact of blood transfusions and transfusion practices on long-term outcome following hepatopancreaticobiliary surgery. J Gastrointest Surg 19:887–896. https://doi.org/10.1007/s11605-015-2776-5
Ejaz A, Spolverato G, Kim Y, Frank SM, Pawlik TM (2014) Identifying variations in blood use based on hemoglobin transfusion trigger and target among hepatopancreaticobiliary surgeons. J Am Coll Surg 219(2):217–228. https://doi.org/10.1016/j.jamcollsurg.2014.02.033
Decreto Ministeriale 2 Novembre 2015: Disposizioni relative ai requisiti di qualità e sicurezza del sangue e degli emcomponenti https://www.gazzettaufficiale.it/eli/id/2015/12/28/15A09709/sg
Barba G, Solaini L, Radi G, Mirarchi MT, D’Acapito F, Gardini A, Cucchetti A, Ercolani G (2022) First 100 minimally invasive liver resections in a new tertiary referral centre for liver surgery. J Minim Access Surg 18(1):51–57. https://doi.org/10.4103/jmas.JMAS_310_20
van der Pas MH, Haglind E, Cuesta MA, Fürst A, Lacy AM, Hop WC, Bonjer HJ (2013) Laparoscopic versus open surgery for rectal cancer (COLOR II): short-term outcomes of a randomised, phase 3 trial. Lancet Oncol 14:210–218. https://doi.org/10.1016/s1470-2045(13)70016-0
Veldkamp R, Kuhry E, Hop WC, Jeekel J, Kazemier G, Bonjer HJ, Haglind E, Påhlman L, Cuesta MA, Msika S, Morino M, Lacy AM (2005) Laparoscopic surgery versus open surgery for colon cancer: short-term outcomes of a randomised trial. Lancet Oncol 6(7):477–484. https://doi.org/10.1016/s1470-2045(05)70221-7
Meybohm P, Herrmann E, Steinbicker AU, Wittmann M, Gruenewald M, Fischer D, Baumgarten G, Renner J, Van Aken HK, Weber CF, Mueller MM, Geisen C, Rey J, Bon D, Hintereder G, Choorapoikayil S, Oldenburg J, Brockmann C, Geissler RG, Seifried E, Zacharowski K (2016) Patient blood management is associated with a substantial reduction of red blood cell utilization and safe for patient’s outcome: a prospective, multicenter cohort study with a noninferiority design. Ann Surg 264:203–211. https://doi.org/10.1097/sla.0000000000001747
Gani F, Cerullo M, Ejaz A, Gupta PB, Demario VM, Johnston FM, Frank SM, Pawlik TM (2019) Implementation of a blood management program at a tertiary care hospital: effect on transfusion practices and clinical outcomes among patients undergoing surgery. Ann Surg 269:1073–1079. https://doi.org/10.1097/sla.0000000000002585
de Almeida JP, Vincent JL, Galas FR, de Almeida EP, Fukushima JT, Osawa EA, Bergamin F, Park CL, Nakamura RE, Fonseca SM, Cutait G, Alves JI, Bazan M, Vieira S, Sandrini AC, Palomba H, Ribeiro U Jr, Crippa A, Dalloglio M, DizMdel P, KalilFilho R, Auler JO Jr, Rhodes A, Hajjar LA (2015) Transfusion requirements in surgical oncology patients: a prospective, randomized controlled trial. Anesthesiology 122:29–38. https://doi.org/10.1097/aln.0000000000000511
Abdelsattar ZM, Hendren S, Wong SL, Campbell DA Jr, Henke P (2015) Variation in transfusion practices and the effect on outcomes after noncardiac surgery. Ann Surg 262:1–6. https://doi.org/10.1097/sla.0000000000001264
Muñoz M, Acheson AG, Bisbe E, Butcher A, Gómez-Ramírez S, Khalafallah AA, Kehlet H, Kietaibl S, Liumbruno GM, Meybohm P, Rao Baikady R, Shander A, So-Osman C, Spahn DR, Klein AA (2018) An international consensus statement on the management of postoperative anaemia after major surgical procedures. Anaesthesia 73:1418–1431. https://doi.org/10.1111/anae.14358
Cavaliere D, Solaini L, Di Pietrantonio D, D’Acapito F, Tauceri F, Framarini M, Ercolani G (2018) Robotic vs laparoscopic splenectomy for splenomegaly: a retrospective comparative cohort study. Int J Surg 55:1–4. https://doi.org/10.1016/j.ijsu.2018.05.012
Papadopoulou K, Dorovinis P, Kykalos S, Schizas D, Stamopoulos P, Tsourouflis G, Dimitroulis D, Nikiteas N (2022) Short-term outcomes after robotic versus open liver resection: a systematic review and meta-analysis. J Gastrointest Cancer. https://doi.org/10.1007/s12029-022-00810-6
Rompianesi G, Montalti R, Ambrosio L, Troisi RI (2021) Robotic versus laparoscopic surgery for spleen-preserving distal pancreatectomies: systematic review and meta-analysis. J Pers Med. https://doi.org/10.3390/jpm11060552
Solaini L, Avanzolini A, Pacilio CA, Cucchetti A, Cavaliere D, Ercolani G (2020) Robotic surgery for gastric cancer in the west: a systematic review and meta-analyses of short-and long-term outcomes. Int J Surg 83:170–175. https://doi.org/10.1016/j.ijsu.2020.08.055
Solaini L, Bocchino A, Avanzolini A, Annunziata D, Cavaliere D, Ercolani G (2022) Robotic versus laparoscopic left colectomy: a systematic review and meta-analysis. Int J Colorectal Dis 37:1497–1507. https://doi.org/10.1007/s00384-022-04194-8
Funding
Open access funding provided by Alma Mater Studiorum - Università di Bologna within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
GE, VA, CM and LS contributed to the study conception and design. Data collection was performed by CM, CI, CAP and FD’A. Analyses were performed by LS and AC. The first draft of the manuscript was written by GE and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in the study were in accordance with ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The need for an informed consent was waived by the local Institutional Review Board.
Consent for publication
All authors give their consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ercolani, G., Solaini, L., D’Acapito, F. et al. Implementation of a patient blood management in an Italian City Hospital: is it effective in reducing the use of red blood cells?. Updates Surg 75, 245–253 (2023). https://doi.org/10.1007/s13304-022-01409-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-022-01409-z