Abstract
Introduction
Type 2 diabetes is a prevalent condition. The change in glucose control and body weight with the use of once-weekly semaglutide was evaluated in individuals with Type 2 diabetes in Colombia.
Methods
This was a real-world, multi-centre, single-arm study involving adults in Colombia with Type 2 diabetes treated with once-weekly subcutaneous semaglutide for approximately 26 weeks. The primary endpoint assessed the change in glycated hemoglobin (HbA1c) from baseline to end of study. Secondary endpoints included changes in body weight from baseline to end of study. The study also explored the proportion of participants achieving predefined HbA1c targets and weight-loss responses at the end of the study.
Results
Data from 225 patients across 11 centers were collected. Most patients were women (65%), and the mean age of the population was 57 years with a median HbA1c of 7.6% and a median body weight of 86 kg. After approximately 26 weeks, semaglutide was associated with a significant reduction in HbA1c of − 0.88 and a body weight reduction of − 4.04kg. The proportion of patients with HbA1c < 7% increased from 32 to 66% at end of study.
Conclusion
Patients treated with once-weekly semaglutide experienced a clinically significant reduction in HbA1c and body weight. These results are in line with previous clinical trials.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? | |
Real-life results of a medication may vary depending on several factors. Real-life results support clinical trial findings and allow different populations to be evaluated in various clinical scenarios | |
Once-weekly semaglutide effectiveness and safety were extensively assessed across various SUSTAIN clinical trial program scenarios. However, Latin American populations are underrepresented in clinical trials and real-life semaglutide studies | |
The COLIBRI study sought to explore the real-world use of once-weekly semaglutide on a diverse type 2 diabetes patient population in Colombia | |
What was learned from the study? | |
After approximately 26 weeks, semaglutide was associated with a significant reduction in glycated hemoglobin (HbA1c), and a high proportion of patients achieved a HbA1c achieved a HbA1c < 7%. Also, the patients treated with semaglutide had a significant weight reduction, with many experiencing a weight loss > 5% | |
The effects of semaglutide were notably more significant in patients with obesity |
Introduction
In Colombia, approximately 3.5 million people live with diabetes, with the majority having Type 2 diabetes (T2D) [1]. Diabetes complications, specifically cardiovascular and renal disease, impose a significant burden on Colombia's healthcare system [2].
Glucose control is essential in preventing microvascular complications in people with T2D [3]. Recent approvals of newer drug classes that reduce blood glucose and mitigate certain risks have prompted adjustments in local and international guidelines. These now recommend therapy tailored to individual characteristics, considering factors such as cardiorenal disease, risk of hypoglycemia, or the need for weight loss. Current evidence supports the use of medications such as glucagon-like peptide one receptor agonists (GLP1-RA) for individuals at high cardiovascular (CV) risk or those with significant weight reduction needs [4].
The efficacy and safety of once-weekly subcutaneous GLP1-RA, semaglutide (OWS) in individuals with T2D were extensively assessed across various clinical scenarios. It was compared to several treatment alternatives, including basal insulin and other GLP1-RA, in the SUSTAIN clinical trial program, demonstrating superior efficacy in glycemic control and weight loss [5]. In the SUSTAIN 6, individuals with T2D at high CV risk treated with OWS significantly reduced the risk of major CV events [6].
The prevalence of diabetes in the Latino population, their response to T2D treatment, and risk of developing T2D-related chronic complications differ from those of other racial and ethnic groups [7]. Cardiovascular outcomes trials (CVOTs) have included Latino participants in the US and other countries [8]. However, cultural, genetic, and environmental differences make more difficult establishing the generalizability of CVOT results to specific Latino populations, such as Colombia.
OWS has been available in Colombia since 2020 with comprehensive healthcare coverage, serving as an adjunct to diet, exercise, and other hypoglycemic medications to improve glycemic control and reduce the risk of major adverse cardiovascular events. The COLIBRI Study sought to explore the impact on glucose control and body weight with the real-world use of OWS on a diverse T2D patient population in Colombia.
Methods
Study Design
COLIBRI was a single-arm, retrospective, multi-center study. Data were collected from the electronic medical records (EMR) of adult patients with T2D who switched to or initiated treatment with OWS. The index date was defined as the date each patient initiated OWS. Patients were required to have at least one glycated hemoglobin (HbA1c) measurement at the index date (week 0) and one measurement during the OWS treatment period (week 26 ± 6 weeks post index date). In cases where the endpoint variables were unavailable at the index date, the most recent value recorded < 12 weeks before OWS initiation was used. The end of study (EOS) was defined as week 26 ± 6 weeks post-index date, and the measurements closest to week 26 were collected. At each center, one investigator collected the information in an electronic case report form. The COLIBRI Study protocol was approved by The National Institute of Drug and Food Surveillance (INVIMA) under the number NN9535-4986 (Supplementary Material).
Study Population and Sample Size
Adult patients (≥ 18 years) with a confirmed diagnosis of T2D for at least 12 months before the index date or initiation of OWS were considered. These patients were required to have at least one HbA1c measurement at the index date (week − 12 to week 0) and at least one in the OWS treatment period (week 26 ± 6 weeks post index date). A sample size of at least 150 patients was estimated to detect a difference of − 0.4% (± 1.5%) on HbA1c between treatment initiation and end of study with an alpha of 0.5% and 90% statistical power. The sample size was adjusted to 200 patients to account for a potential 25% incomplete data from medical records.
Endpoints
The primary endpoint assessed the change in HbA1c from index date to EOS. The secondary endpoint explored the change in body weight and OWS at EOS. Exploratory endpoints included the percentage of patients achieving HbA1c < 7%, HbA1c < 7%, and no weight gain and weight loss ≥ 5% at EOS. World Health Organization (WHO) categories for body mass index (BMI) classification were used.
Statistical Analyses
Data from all study sites were analyzed and reported together. Descriptive statistics according to the distribution of the variable for continuous variables were used [mean, standard deviation (SD), median, 95% CI, and p25-p75]. Categorical variables are presented as frequencies and percentages for categorical variables.
For the analysis of primary and secondary endpoints, a baseline-adjusted change was planned using an analysis of covariance (ANCOVA) model, with the change from baseline in HbA1c or body weight as dependent variables. Age, sex, duration of diabetes, and body mass index (BMI) were intended as covariates. However, despite attempts to find suitable variable transformations and explore several combinations of independent variables, none of the models met the required assumptions to conduct the ANCOVA analysis. A Wilcoxon test (for paired data) was used to evaluate the primary and secondary endpoints using a nonparametric confidence interval and an estimator for the pseudomedian for the median difference between index date and EOS evaluations. All statistical tests for the primary endpoint were performed as two-sided tests with a significance level of 0.05. No missing data were imputed. Based on the appropriate denominator, exploratory endpoints are presented in frequency tables as percentages with numerator counts. The relationship among BMI, weight reduction, and HbA1c reduction was explored using a logistic regression with age and sex as covariates using the normal weight population as reference and is presented as OR with 95% CI. All analyses were performed using Stata statistical software (version 17, Stata Corp., College Station, TX, USA).
Ethical Approval
The ethical considerations for this protocol are based on the Declaration of Helsinki and the Ethical Guidelines for Health-Related Research with Human Beings prepared by the Council for International Organizations of Medical Sciences (CIOMS) in collaboration with the World Health Organization (WHO). The protocol was approved by all the institution’s ethics committees or Institution Review Board (IRB). This study complies with the Colombian Ministry of Health’s Resolution 8430 and was considered a no risk study and as such none of the participants’ consent to participate was required. No participants’ consent to publish was required because no identifiable information is presented in the results. Details from each center contribution and IRB approval are provided in the Supplementary Material.
Results
Data from 225 patients were included in the final analysis. Most patients were women (65%), and the mean age of the population was 57 (± 11.7) years. The median time from diagnosis was 4 years, and 95% of patients had some comorbidity, the most common being hypertension. Table 1 summarizes the patients’ main characteristics. The most common reasons for starting OWS were poor glycemic control and weight reduction. The motivation to start OWS is presented in Fig. 1.
The most common medications at index date were metformin in combination with a sodium-glucose two transporter inhibitor (43%) or monotherapy (26.6%). Figure 2 shows all the diabetes medications at the index date.
Most patients at the index date started with an OWS dose of 0.25 mg (91%), and most patients at EOS reached a dose of 0.5 mg (76%).
HbA1c Reduction
Most patients (81.3%) presented a reduction in HbA1c. The median HbA1c at index date was 7.6% (IQR 6.8, 8.9), and at EOS, it reduced significantly to a median of 6.8% (IQR 6.1, 7.3) (Fig. 3). The median of the difference was – 0.88% (95% CI − 1.06% to − 0.71%).
Body Weight
Body weight reduced from a median of 86 kg at the index date (IQR 78, 101) to 82 kg (IQR 82.2, 87) at EOS. The median of the difference was − 4.04 kg (95% CI − 4.75 kg to − 3.50 kg).
Exploratory Outcomes
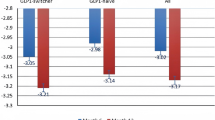
The proportion of patients with HbA1c < 7% increased from 32 to 66% at EOS (p < 0.001). The effect on HbA1c reduction was more significant in the population with overweight and obese populations. The number of patients achieving HbA1c < 7% was 68% in those who were overweight, 61% in those with obesity, and 76% in the morbidly obese group (Fig. 4). Forty-four percent of patients achieved a weight reduction of at least 5%.
The combined effect of achieving an HbA1c < 7.0% with no weight gain was seen in 57% of the patients, and this effect was more significant in the morbidly obese (Fig. 4). The impact on reductions in HbA1c was found to be related to the initial BMI; however, this relationship was not observed regarding weight reduction. Table 2 summarizes the OR for each BMI category.
Discussion
COLIBRI was the first study to assess the real-world use of semaglutide in adults with T2D in Colombia. It gathers data from several centers nationwide with patients from different regions, in different clinical settings, and with different healthcare coverage.
The glucose control outcomes in COLIBRI are similar to those of other real-world experiences. The SURE investigated OWS in a real-world setting in several European countries [9,10,11,12,13,14,15,16] and Canada [17]. The mean change in the SURE studies for HbA1c ranged from − 0.8% to − 1.5%. Compared to the glucose control outcomes reported in the SUSTAIN [18,19,20,21,22,23,24] clinical trial program, where a change in HbA1c ranged from − 1.1 to − 1.8%, results from COLIBRI are near the lower end. This has been reported in other real-world studies [13] and may be related to the differences in real-life scenarios with those of the clinical trials, like lower adherence [25] and a more diverse population.
A more important finding than average glucose reduction is the proportion of patients achieving the glucose control proposed in the guidelines [4]. In line with the results reported in the SUSTAIN program [5], more than half of the treated patients achieved an HbA1c < 7.0%. This is an important finding in a country where glucose control goals are achieved only in 50% of patients [2]. Glucose control remains an important factor when choosing a glucose-lowering medication.
Body weight outcomes in COLIBRI are like those reported in clinical trials [18,19,20,21,22,23,24], ranging from − 3.5 to 6.56 kg, and in real-life studies [9,10,11,12,13,14,15,16,17], the reported reductions range from − 4.2 to − 7.8 kg. Weight reduction remains an elusive goal for many people with T2D. Achieving and sustaining a ≥ 5% weight reduction is essential in people with T2D as it helps improve glucose control and reduces the need for other glucose-lowering medications [25]. In the SUSTAIN program, 37–62% of participants treated with OWS achieved this goal. COLIBRI proves that achieving a substantial body weight reduction in real life is possible when selecting the right medication to treat T2D.
Unlike the SURE program, where most patients at EOS were receiving an OWS dose of 1.0 mg. In COLIBRI, most patients achieved an OWS dose of 0.5 mg. This could be the result of local preferences motivated by availability or costs. Although the clinical results are very satisfying, there is room for improvement. Some patients may benefit from further titration to a 1 mg dose.
Finally, OWS was introduced in Colombia in 2020 with comprehensive healthcare coverage. The adoption of changes to guidelines takes time [26]. This could explain why weight loss and cardiovascular protection rank as the second and third reasons for starting OWS, indicating a shift from glucose-centric diabetes treatment to a more goal- and patient-centered approach.
This study has several limitations. The retrospective nature of its design prevents the analysis of important outcomes, such as adherence, adverse effects, or patient-reported outcomes, and prevents analysis of the impact of other variables such as the prescription of a nutritional plan or regimen during the observation period. Also, it may introduce bias in the selection of highly adherent or motivated patients. The absence of a control group precludes the isolation of the effects of OWS.
Conclusion
In COLIBRI, patients treated with OWS experienced a clinically significant reduction in HbA1c and body weight. These results are in line with those of previous clinical trials and real-world experiences reported worldwide. Therefore, the results from clinical trials of OWS can be generalizable to a Latino population from Colombia.
Data Availability
The datasets generated or analyzed for the current study are available from the sponsor institution upon reasonable request.
References
Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, et al. IDF Diabetes Atlas: Global, regional, and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabet Res Clin Pract. 2022;183: 109119.
Mendivil CO, Amaya-Montoya M, Hernández-Vargas JA, Ramírez-García N, Romero-Díaz C, Pérez-Londoño A, et al. Achievement of treatment goals among adults with diabetes in Colombia, 2015–2019: results from a national registry. Diabet Res Clin Pract. 2022;186: 109823.
Zoungas S, Arima H, Gerstein HC, Holman RR, Woodward M, Reaven P, et al. Effects of intensive glucose control on microvascular outcomes in patients with type 2 diabetes: a meta-analysis of individual participant data from randomised controlled trials. Lancet Diabetes Endocrinol. 2017;5(6):431–7.
Pharmacologic Approaches to Glycemic Treatment. Standards of care in diabetes-2024. Diabetes Care. 2024;47(Suppl 1):S158–78.
Aroda VR, Ahmann A, Cariou B, Chow F, Davies MJ, Jódar E, et al. Comparative efficacy, safety, and cardiovascular outcomes with once-weekly subcutaneous semaglutide in the treatment of type 2 diabetes: Insights from the SUSTAIN 1–7 trials. Diabetes Metab. 2019;45(5):409–18.
Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375(19):1834–44.
Rodríguez JE, Campbell KM. Racial and ethnic disparities in prevalence and care of patients with type 2 diabetes. Clin Diabet. 2017;35(1):66–70.
Kelley AT, Mizokami-Stout K, O’Brien MJ, Bowen ME, Sussman J. Hispanic representation in diabetes cardiovascular outcomes trials. BMJ Open Diabetes Res Care. 2019;7(1): e000656.
Rajamand Ekberg N, Bodholdt U, Catarig AM, Catrina SB, Grau K, Holmberg CN, et al. Real-world use of once-weekly semaglutide in patients with type 2 diabetes: results from the SURE Denmark/Sweden multicentre, prospective, observational study. Prim Care Diabetes. 2021;15(5):871–8.
Rudofsky G, Catarig AM, Favre L, Grau K, Häfliger S, Thomann R, Schultes B. Real-world use of once-weekly semaglutide in patients with type 2 diabetes: results from the SURE Switzerland multicentre, prospective, observational study. Diabet Res Clin Pract. 2021;178: 108931.
Holmes P, Bell HE, Bozkurt K, Catarig AM, Clark A, Machell A, Sathyapalan T. Real-world use of once-weekly semaglutide in type 2 diabetes: results from the SURE UK multicentre, prospective observational study. Diabetes Ther. 2021;12(11):2891–905.
Napoli R, Berra C, Catarig AM, Di Loreto C, Donatiello E, Berentzen TL, et al. Once-weekly semaglutide use in patients with type 2 diabetes: real-world data from the SURE Italy observational study. Diabetes Obes Metab. 2023;25(6):1658–67.
Menzen M, Berentzen TL, Catarig AM, Pieperhoff S, Simon J, Jacob S. Real-World Use of Once-Weekly Semaglutide in Type 2 Diabetes: Results from SemaglUtide Real-world Evidence (SURE) Germany. Exp Clin Endocrinol Diabet Off J German Soc Endocrinol German Diabet Assoc. 2023;131(4):205–15.
Wolffenbuttel BHR, Brugts MP, Catarig AM, Clark A, Kok M, Lieverse AG, van Soest J. Once-weekly semaglutide use in type 2 diabetes: real-world data from the SURE Netherlands observational study. Adv Ther. 2023;40(3):920–33.
Bellido V, Abreu Padín C, Catarig AM, Clark A, Barreto Pittol S, Delgado E. Once-Weekly Semaglutide Use in Patients with Type 2 Diabetes: Results from the SURE Spain Multicentre, Prospective, Observational Study. J Clin Med. 2022;11(17).
Mohammedi K, Belhatem N, Berentzen TL, Catarig AM, Potier L. Once-weekly semaglutide use in patients with type 2 diabetes: results from the SURE France multicentre, prospective, observational study. Diabetes Obes Metab. 2023;25(7):1855–64.
Yale JF, Catarig AM, Grau K, Harris S, Klimek-Abercrombie A, Rabasa-Lhoret R, et al. Use of once-weekly semaglutide in patients with type 2 diabetes in routine clinical practice: results from the SURE Canada multicentre, prospective, observational study. Diabetes Obes Metab. 2021;23(10):2269–78.
Ahmann AJ, Capehorn M, Charpentier G, Dotta F, Henkel E, Lingvay I, et al. Efficacy and safety of once-weekly semaglutide versus exenatide er in subjects with type 2 diabetes (SUSTAIN 3): a 56-week, open-label, Randomized Clinical Trial. Diabetes care. 2018;41(2):258–66.
Ahrén B, Masmiquel L, Kumar H, Sargin M, Karsbøl JD, Jacobsen SH, Chow F. Efficacy and safety of once-weekly semaglutide versus once-daily sitagliptin as an add-on to metformin, thiazolidinediones, or both, in patients with type 2 diabetes (SUSTAIN 2): a 56-week, double-blind, phase 3a, randomised trial. Lancet Diabetes Endocrinol. 2017;5(5):341–54.
Aroda VR, Bain SC, Cariou B, Piletič M, Rose L, Axelsen M, et al. Efficacy and safety of once-weekly semaglutide versus once-daily insulin glargine as add-on to metformin (with or without sulfonylureas) in insulin-naive patients with type 2 diabetes (SUSTAIN 4): a randomised, open-label, parallel-group, multicentre, multinational, phase 3a trial. Lancet Diabetes Endocrinol. 2017;5(5):355–66.
Capehorn MS, Catarig AM, Furberg JK, Janez A, Price HC, Tadayon S, et al. Efficacy and safety of once-weekly semaglutide 1.0mg vs once-daily liraglutide 1.2 mg as add-on to 1–3 oral antidiabetic drugs in subjects with type 2 diabetes (SUSTAIN 10). Diabetes Metab. 2020;46(2):100–9.
Lingvay I, Catarig AM, Frias JP, Kumar H, Lausvig NL, le Roux CW, et al. Efficacy and safety of once-weekly semaglutide versus daily canagliflozin as add-on to metformin in patients with type 2 diabetes (SUSTAIN 8): a double-blind, phase 3b, randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7(11):834–44.
Pratley RE, Nauck MA, Barnett AH, Feinglos MN, Ovalle F, Harman-Boehm I, et al. Once-weekly albiglutide versus once-daily liraglutide in patients with type 2 diabetes inadequately controlled on oral drugs (HARMONY 7): a randomised, open-label, multicentre, non-inferiority phase 3 study. Lancet Diabetes Endocrinol. 2014;2(4):289–97.
Rodbard HW, Lingvay I, Reed J, de la Rosa R, Rose L, Sugimoto D, et al. Semaglutide added to basal insulin in type 2 diabetes (SUSTAIN 5): a randomized, controlled trial. J Clin Endocrinol Metab. 2018;103(6):2291–301.
Franz MJ, Boucher JL, Rutten-Ramos S, VanWormer JJ. Lifestyle weight-loss intervention outcomes in overweight and obese adults with type 2 diabetes: a systematic review and meta-analysis of randomized clinical trials. J Acad Nutr Diet. 2015;115(9):1447–63.
Wong MCS, Wang HHX, Kwan MWM, Chan WM, Fan CKM, Liang M, et al. The adoption of the reference framework for diabetes care among primary care physicians in primary care settings: a cross-sectional study. Medicine. 2016;95(31): e4108.
Acknowledgements
The authors would like to thank Carlos Mario Balcazar, Julian Coronel, Miguel Folleco, Alberto Hernandez, Omar Herrera, and Eder Hernandez for their help in data gathering.
Medical Writing/Editorial Assistance.
The authors received support for medical writing and editorial assistance from Festina Lente (funded by Novo Nordisk). ILS Clinical Research SC was responsible for the statistical analysis (funded by Novo Nordisk). Finally Cohortias was the Contract Research Organization responsible for monitoring the progress of the study and ensuring that was conducted according to the protocol.
Funding
This study was funded by Novo Nordisk Colombia. Novo Nordisk was involved in study design and data analysis, and representatives of the company also acted as authors of this paper. The journal’s Rapid Service Fee was funded by Novo Nordisk Colombia.
Author information
Authors and Affiliations
Contributions
Andres F. Suarez-Rodriguez and Maria A. Alzate-Vinasco are responsible for the concept and design of the study. Dagoberto Serpa-Díaz, Carlos A. Llanos-Florez, Ronald Serrano Uribe, Dora I. Molina de Salazar, German C. Giraldo-Gonzalez, and Miguel Urina-Triana participated in data collection. Andres F. Suarez-Rodriguez and Maria A. Alzate-Vinasco contributed to data analysis. All authors reviewed, edited, and approved the final draft of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The author(s) declared the following potential conflicts of interest concerning this article's research, authorship, and publication. Dagoberto Serpa-Díaz has acted as an advisor for Boehringer Ingelheim, received honoraria from Novo Nordisk, Pfizer, Boehringer Ingelheim, and Amgen, and received research funding from Novo Nordisk. Carlos A. Llanos-Florez has received honoraria from Novo Nordisk. German C Giraldo-Gonzalez has received honoraria from Novo Nordisk, Sanofi, Pfizer, Boehringer Ingelheim and Amgen. Andres F. Suarez-Rodriguez and Maria A. Alzate-Vinasco are Novo Nordisk employees. Ronald Serrano Uribe and, Dora I. Molina de Salazar has nothing to disclose.
Ethical Approval
The ethical considerations for this protocol are based on the Declaration of Helsinki and the Ethical Guidelines for Health-Related Research with Human Beings prepared by the Council for International Organizations of Medical Sciences (CIOMS) in collaboration with the World Health Organization (WHO). The protocol was approved by all the institution’s ethics committees or Institution Review Board (IRB). This study complies with the Colombian Ministry of Health’s Resolution 8430 and was consider a no risk study and as such no participants’ consent to participate was required. No participants’ consent to publish was required because no identifiable information is presented in the results. Details from each center contribution and IRB approval are provided in the supplementary material.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Serpa-Díaz, D., Llanos-Florez, C.A., Uribe, R.S. et al. Glycemic Control and Body Weight Reduction with Once-Weekly Semaglutide in Colombian Adults with Type 2 Diabetes: Findings from the COLIBRI Study. Diabetes Ther 15, 1451–1460 (2024). https://doi.org/10.1007/s13300-024-01586-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-024-01586-7