Abstract
Type 2 diabetes (T2D) is a progressive disease, with many individuals eventually requiring basal insulin therapy to maintain glycaemic control. However, there exists considerable therapeutic inertia to the prompt initiation and optimal titration of basal insulin therapy due to barriers that include fear of injections, hypoglycaemia, weight gain, and burdensome regimens. Hypoglycaemia is thought to be a major barrier to optimal glycaemic control and is associated with significant morbidity and mortality. Newer second-generation basal insulin analogues provide comparable glycaemic control with lower risk of hypoglycaemia compared with first-generation basal insulin analogues. The present review article discusses clinical evidence for one such second-generation basal insulin analogue, insulin glargine 300 U/mL (Gla-300), in the context of hypothetical case studies that are representative of individuals who may attend routine clinical practice. These case studies discuss individualised treatment needs for people with T2D who are insulin-naïve or pre-treated. Clinical characteristics such as older age, frequent nocturnal hypoglycaemia, and renal impairment, which are known risk factors for hypoglycaemia, are also considered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Webinar Video: Author discussion on hypothetical type 2 diabetes cases in the context of personal clinical experience
Many people with type 2 diabetes (T2D) will eventually require a basal insulin (BI) to achieve or maintain glycaemic control. |
Second-generation BI analogues, such as insulin glargine 300 U/mL (Gla-300) and insulin degludec (IDeg), represent a suitable option for people needing intensification of their antihyperglycaemic regimens to meet individualised glycaemic targets, including high-risk groups such as those with renal impairment and older people. |
Improved communication between healthcare professionals and patients, along with appropriate educational tools and support, may increase patient confidence in the administration and titration of BI dose, ultimately improving glycaemic management. |
Digital Features
This article is published with digital features, including a webinar and infographic, to facilitate understanding of the article. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.17318996.
Introduction
Type 2 diabetes (T2D) is a progressive disease and people with T2D often require exogenous insulin to maintain optimal glycaemia [1,2,3]. Prompt and persistent treatment intensification is recommended to prevent the complications of diabetes [1, 3, 4]. However, clinical/therapeutic inertia (i.e. delays in treatment intensification or titration) is common, especially when intensifying therapy with insulin [3, 5]. In a retrospective cohort study, the median time to insulin intensification was at least 6 years even when the person’s HbA1c was 8% or higher, regardless of whether they were taking one, two, or three oral antihyperglycaemic drugs (OADs) [6]. In the USA, between 2005 and 2012, approximately 40% of people using insulin to control diabetes had an HbA1c greater than 8%, suggesting a lack of appropriate treatment intensification or improper titration [7]. In the International Diabetes Management and Practices Study (IDMPS), a cross-sectional real-world study involving more than 66,000 individuals with T2D living in developing countries, less than 50% of people with T2D taking any antihyperglycaemic medication and less than 30% of those taking insulin with or without OADs had an HbA1c below 7% over the entire study period (2005–2017), while the mean time to insulin initiation was greater than 8 years [8].
Various barriers contribute to delays in initiation, intensification, and optimal titration of insulin therapy, such as fear of injections, burdensome regimens, fear of hypoglycaemia, weight gain, and the perception that the need for insulin signals a failure of diabetes self-management (Fig. 1) [3, 5, 9]. Of these, hypoglycaemia is often thought to be the main barrier to glycaemic control.
The aim of this review is to summarise how hypoglycaemia can impact optimal glycaemic management in T2D, and to use hypothetical clinical cases to examine how insulin glargine 300 U/mL (Gla-300), which has been shown to provide the same glucose-lowering potential but lower hypoglycaemia risk versus first-generation basal insulin (BI) analogue, insulin glargine 100 U/mL (Gla-100) [10], may be beneficial in various clinical situations. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Hypoglycaemia as a Barrier to Optimal Management of T2D
Hypoglycaemia is common in people with T2D, and is often thought to be the main barrier to glycaemic control. In the global hypoglycaemia assessment tool (HAT) study, the estimated annual rate of any hypoglycaemia in T2D was 19.2 episodes per patient-year, and the rate of severe hypoglycaemia was 2.5 events per patient-year [11]. In another international survey, 28% of people with T2D reported experiencing non-severe hypoglycaemic events at least once per week [12]. In the Canadian InHypo-DM study, 54% of people with T2D experienced moderate hypoglycaemia with an average incidence of 28 events per-patient year [13]. Severe hypoglycaemia was experienced by 38% of people with T2D, with an average incidence of 2.5 events per-patient year [13]. These results were higher than other real-world studies capturing severe hypoglycaemia, which may be attributable to a large number of these studies relying on clinical documentation from medical records or patient registries, which may underestimate actual events.
High rates of hypoglycaemia are particularly concerning given that it is associated with high morbidity and mortality [14]. Mild to moderate hypoglycaemia can increase the risk of injury from falls or accidents [15]. Frequent hypoglycaemia can lead to hypoglycaemia unawareness and increased potential for recurrent hypoglycaemia [15,16,17]. In turn, recurrent hypoglycaemia, especially recurrent severe hypoglycaemia, is strongly associated with cardiovascular events, including sudden death, autonomic neuropathy, silent myocardial ischemia, and other adverse cardiovascular outcomes [17,18,19]. The risk of hypoglycaemia is increased in people who are elderly or frail, have renal impairment, or who have multiple comorbidities [20, 21]. For example, the risk of hypoglycaemia can be approximately twofold higher in those with chronic kidney disease [20] and 1.5-fold higher in individuals 75 years of age or older [21], while the risk of hypoglycaemia increases with increasing comorbidities [21]. However, data are more limited in such groups as they are often excluded from typical randomized controlled trials. Therefore, real-world studies that have minimal exclusion criteria and are more representative of everyday clinical practice can provide valuable information about hypoglycaemia.
Hypoglycaemia places a high economic burden on people with diabetes as well as society in terms of direct (self-management, increased healthcare resource utilisation, including hospital admissions) and indirect costs (absenteeism, reduced productivity) [11, 12]. There is also an adverse impact of hypoglycaemia on daily quality of life in terms of reduced feelings of psychological wellbeing and fear of experiencing further events [22]. Hypoglycaemia has an adverse impact on family members who care for people with diabetes, with 64% reporting that it caused them anxiety or worry [23]. Concerns about hypoglycaemia pose barriers to timely insulin initiation and optimal uptitration for both clinicians and people with diabetes [5, 9, 24]. People with diabetes have been shown to alter self-management behaviours, including refusing to start insulin or undertitrating or omitting insulin doses, to avoid hypoglycaemia [5, 12]. The adverse impact of hypoglycaemia on optimising insulin treatment is highlighted by the finding that 72–79% of physicians in the multinational Global Attitudes of Patients and Physicians in Insulin Therapy study reported they would treat diabetes more aggressively if they were not worried about hypoglycaemia [25].
Second-Generation Basal Insulins: Why Pharmacokinetic/Pharmacodynamic Profiles Matter
Guideline recommendations call for use of BI analogues, which carry a lower risk of hypoglycaemia compared with neutral protamine Hagedorn insulin (NPH) [1, 2, 26]. The ideal BI has a pharmacokinetic (PK) and pharmacodynamic (PD) profile that has little to no within-day variability and a long duration of action, resulting in better glycaemic control before and between meals with less potential for hypoglycaemia [27]. Of the BI analogues, Gla-300 and insulin degludec (IDeg)—both second-generation BI analogues—have more stable and prolonged PK/PD profiles than Gla-100, which are associated with a lower risk of hypoglycaemia [28, 29]. In the next section, evidence for the efficacy and safety of Gla-300 from randomized controlled trials and real-world studies will be reviewed in the context of hypothetical clinical case studies.
Gla-300 Use in People with T2D: Hypothetical Case Studies
The following hypothetical case studies were developed to illustrate key concepts in initiating BI therapy with a second-generation insulin analogue or switching BI therapy to a second-generation insulin analogue in people with T2D.
Case 1: Insulin Initiation—Uncontrolled on Multiple Oral Agents
Patient profile | |
|---|---|
Name | Joseph |
Age | 52 years |
Occupation | Delivery driver |
Lifestyle/activity | Plays tennis three times a week, non-smoker |
Diabetes duration | 6 years |
BMI | 32 kg/m2 |
Laboratory results | Current HbA1c 8.2% (66 mmol/mol) with an HbA1c > 8% (> 64 mmol/mol) for the previous year, blood pressure 126/80 mmHg, estimated glomerular filtration rate (eGFR) 90 ml/min/1.73 m2, urine albumin to creatinine ratio (ACR) normal |
Medical history | Tried a glucagon-like peptide 1 receptor agonist (GLP-1 RA) but stopped treatment as he was unable to tolerate the gastrointestinal side effects. No established cardiovascular disease. Hypercholesterolaemia |
Current medication | Metformin (extended release 1000 mg twice daily), a sodium-glucose co-transporter 2 (SGLT2) inhibitor (canagliflozin 300 mg/day), sulfonylurea (glimepiride 4 mg/day), dipeptidyl peptidase 4 (DPP4) inhibitor (sitagliptin 100 mg once daily), two anti-hypertension medications (ramipril 10 mg once daily and amlodipine 10 mg once daily) and atorvastatin 40 mg once daily |
Relevant family history | Father had type 2 diabetes and died of myocardial infarction |
Joseph’s Concerns
Joseph is reluctant to start insulin because he believes insulin injections will be inconvenient and difficult, particularly because he does not keep a regular schedule because of long hours at his work. Hypoglycaemia is a concern as he has already experienced an event after initiating therapy with sulfonylurea. Many countries have requirements regarding driving licence eligibility for people with diabetes, particularly those with insulin-treated diabetes, including the need for self-measured plasma glucose (SMPG) measurements prior to driving and requiring individuals not drive if their blood glucose is below certain thresholds [30,31,32]. Furthermore, many regulations stipulate that those experiencing hypoglycaemia while driving, or who experience a severe hypoglycaemia event at any time, could have their driving privileges revoked for a period of time [30]. Joseph is concerned about potential additional self-care requirements and monitoring needed to ensure he meets driving regulations should he start insulin therapy and about the potential impact on his job. Given his familial history of cardiovascular disease, he is also worried about the potential for weight gain.
Research Evidence
It is a common perception that insulin therapy is difficult and inconvenient, but a second-generation BI analogue like Gla-300 can provide flexibility to match people’s daily activities. The efficacy and safety of Gla-300 is maintained when it is injected up to 3 h before or after the usual time of once-daily administration [33], which may allow people with T2D more freedom in timing their BI injections to deal with the situational irregularities of daily life [33].
Key to a flexible insulin therapy regimen is an individual’s ability to self-manage their diabetes. Guidelines highlight the importance of instruction and involving individuals in insulin management and highlight that self-titration of BI can be beneficial [26]. In the TAKE CONTROL study, self-titration of Gla-300 resulted in superior HbA1c reduction compared with physician-led titration (p = 0.0247), without an increased risk of hypoglycaemia [34]. Furthermore, significantly more participants reached fasting SMPG targets without hypoglycaemia with self- versus physician-led titration (p = 0.0187) [34]. The TITRATION study compared self-titration of Gla-300 using a simple titration algorithm of 1 unit/day used in the INSIGHT study [35] with that of the more complex physician-led EDITION algorithm [36,37,38] and showed that both algorithms resulted in comparable glycaemic control and rates of hypoglycaemia [39]. Furthermore, the AUTOMATIX study showed that device-supported titration with Gla-300 demonstrated a good safety profile and was non-inferior to diabetes specialist-led titration, with a trend towards shorter times to reach glycaemic target [40].
Glycaemic outcomes during the first few months following initiation of BI therapy are important for longer-term clinical outcomes [41]. Given the likely fear of hypoglycaemia following the initiation of BI therapy, to optimise glycaemic control in the early stages it is important to closely follow up the patient post-BI initiation and to choose a BI with a lower risk of hypoglycaemia. Failure to achieve glycaemic control within the first 3 months following initiating a BI has been shown to increase the risk of not achieving glycaemic targets after 2 years [41]. Furthermore, experiencing hypoglycaemia during this initial 3 months also increased the risk of long-term hypoglycaemia [41]. In a retrospective study of over 55,000 insulin-naïve individuals with T2D initiating BI therapy, those who experienced hypoglycaemia within the first 6 months after starting BI were more likely to discontinue their therapy after 12 months compared with those who did not [42]; these individuals, in turn, had greater healthcare resource utilisation and associated costs [42].
The EDITION 3 study demonstrated that, in people with T2D newly initiating BI therapy, Gla-300 provides comparable glycaemic control with a lower risk of hypoglycaemia compared with first-generation BI, particularly during the initial 8 weeks of therapy [38]. The BRIGHT study compared the efficacy and safety of Gla-300 with IDeg in insulin-naïve participants with T2D [43]. Gla-300 and IDeg 100 U/mL (IDeg-100) yielded similar HbA1c reductions of 1.6%, as well as similar variability in 24-h SMPG [43]. Hypoglycaemia was comparable between Gla-300 and IDeg-100 during the full study and maintenance periods [43]. During the titration period, however, the incidence and rates of confirmed hypoglycaemia (≤ 3.9 mmol/L [70 mg/dL]) were lower with Gla-300 versus IDeg (by 26% [p = 0.03] and 23% [p = 0.02], respectively), as were incidence and rates of confirmed hypoglycaemia at the less than 3.0 mmol/L (54 mg/dL) threshold (by 37% [p = 0.04] and 43% [p = 0.04], respectively). During the same period, the rate of nocturnal (00:00–06:00 h) confirmed hypoglycaemia (≤ 3.9 mmol/L [70 mg/dL]) was 35% lower with Gla-300 versus IDeg (p = 0.04) [43]. Given that fear of hypoglycaemia is a major barrier to optimal dose titration of insulin [9], the finding of less hypoglycaemia with Gla-300 versus first-generation BI analogues and versus IDeg-100 during the time of more intensive insulin titration could help to increase people’s confidence to initiate and properly self-titrate their BI to reach their glucose target. This is particularly important as early glycaemic control has been shown to provide improved long-term glycaemic outcomes [41].
The LIGHTNING real-world evidence study showed that Gla-300 provided 50% lower rates of severe hypoglycaemia (defined on the basis of electronic medical records, and including events defined as plasma glucose below 54 mg/dL [3.0 mmol/L]) compared with first-generation BI analogues, Gla-100 and IDet, in insulin-naïve individuals with T2D initiating BI therapy [44]. In the DELIVER naïve real-world evidence study, initiation of Gla-300 versus Gla-100 was associated with significantly improved glycaemic control, numerically fewer hypoglycaemia events and significantly more individuals reaching HbA1c target of below 7.0% without experiencing hypoglycaemia [45]. In another real-world study (DELIVER Naïve D), initiation of Gla-300 or IDeg resulted in similar improvements in glycaemic control and a similar effect on hypoglycaemia during the 6-month follow-up [46]. The real-world CONFIRM study, using the same database as the DELIVER Naïve D study, reported greater HbA1c reductions with IDeg versus Gla-300, although only 35% of the matched patients had follow-up HbA1c available. Furthermore, on-treatment incidence and rates of hypoglycaemia were comparable with IDeg and Gla-300 after 6 months as in DELIVER Naïve D [47]. It has previously been discussed that the treatment groups in CONFIRM were not completely matched at baseline for hypoglycaemia and HbA1c and that these results should be interpreted with caution [48].
Weight gain is a common concern of people starting insulin therapy, but research suggests that any such gains are a lower clinical priority than the glycaemic control insulin therapy can allow [49]. In the EDITION 3 and BRIGHT trials of people with T2D initiating BI, over 6 months of Gla-300 use weight only increased by 0.5 kg and 2.0 kg respectively, while HbA1c decreases were 1.4% and 1.6% [38, 43].
People with T2D are at an increased risk of cardiovascular disease, but the ORIGIN study showed that BI therapy with Gla-100 did not increase the risk of major adverse cardiovascular events, such as myocardial infarctions, versus standard care [50]. Furthermore, the DEVOTE study demonstrated that IDeg was noninferior to Gla-100 in terms of the proportion who experienced an adjudicated major cardiovascular event (death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke) in a population with T2D [51].
Possible Clinical Considerations for Joseph
Given that adequate glycaemic control was not achieved with a combination of three OADs, one treatment pathway to consider is the addition of a BI alongside a reduction of sulfonylurea dose, with the intent to eventually discontinue sulfonylurea treatment as the BI dose is optimised. Second-generation BIs, such as Gla-300 or IDeg, may be relevant because of their hypoglycaemia benefits compared with other BIs. The BRIGHT study, which included people with T2D initiating BI, indicated that Gla-300 and IDeg were similar in terms of glycaemic control and overall hypoglycaemia. However, Gla-300 was associated with lower rates of hypoglycaemia in the first 8 weeks of treatment versus IDeg, which may be important in this case considering Joseph’s profession as a driver [43]. In addition to intensifying treatment with insulin, Joseph may benefit from reiteration of advice on lifestyle and diet modification and introduction to technology that can help him manage his diet and busy lifestyle. Guidance, and training, on performing blood glucose measurements one or two times daily may be of benefit to Joseph so that he is comfortable managing this at home. Flash glucose monitoring (FGM) may assist Joseph with self-managing his glucose measurements. If Gla-300 were to be selected for treatment, an initial dose of 0.2 units/kg once daily, as per the label [52, 53], can be considered, and titration performed according to an evidence-based titration algorithm [26]. The EDITION studies employed weekly adjustment of insulin doses (no more frequently than every 3 days) by the physician until fasting SMPG targets were reached without hypoglycaemia [36,37,38]; however, it should be noted that patient-driven titration using daily dose adjustments of 1 U/day until fasting glucose targets are reached has been shown to provide comparable efficacy and safety to the EDITION algorithm [39]. It is important to note that different patients require individualized glucose targets based on their comorbidities and life expectancy. In the case of Joseph, he is young and has no established macrovascular disease; therefore, his target of fasting self-monitored plasma glucose could be 4.4–7.2 mmol/L (80–130 mg/dL), with an HbA1c target below 7% without hypoglycaemia to ensure tight glycaemic control given Joseph’s relatively young age and high cardiovascular risk [54]. As Joseph is a delivery driver, he will need advice on the local driving regulations for people with insulin-treated diabetes, including further education to improve self-management behaviours and guidance on hypoglycaemia avoidance and management, including the need for SMBG measurements prior to driving [30,31,32].
Case 2: Hypoglycaemia Concerns with First-Generation BI
Patient profile | |
|---|---|
Name | Anna |
Age | 61 years |
Occupation | Office administrator |
Lifestyle/activity | Lives alone, regularly walks 3 km every other day |
Diabetes duration | 10 years |
BMI | 31 kg/m2 |
Laboratory results | HbA1c 7.5% (58 mmol/mol), blood pressure 120/85 mmHg, eGFR 92 ml/min/1.73 m2, urine ACR normal |
Medical history | Suspected nocturnal hypoglycaemia was recently confirmed by FGM, which Anna had been using to monitor her 24-h glucose profile in response to her concerns about hypoglycaemia. On the basis of the FGM data she is experiencing 1–2 nocturnal episodes every 2 or 3 weeks. She had previously tried a fixed-ratio combination of a GLP-1 RA and a BI; however, she had returned to using BI only as a result of gastrointestinal side effects. Anna has no established cardiovascular disease |
Current medication | Gla-100 (28 U) at bedtime, metformin (1000 mg twice daily), an SGLT2 inhibitor (empagliflozin 10 mg once daily), and a DPP4 inhibitor (linagliptin 5 mg once daily) |
Anna’s Concerns
Anna is fatigued during the day as a result of her frequent nocturnal hypoglycaemia, which is interfering with her job and daytime activities, but she is also concerned that she is unaware of the nocturnal hypoglycaemic events. While she has not required third-party help with any of her hypoglycaemic events so far, she is worried about experiencing a severe event, particularly given that she lives alone and needs to drive for her work and social life. Anna is at her individualised temporary target of HbA1c 7.5%, which was chosen following discussions with her physician because of her frequent nocturnal hypoglycaemia. She believes that because her HbA1c levels are under control she does not need to take insulin anymore.
Research Evidence
Hypoglycaemia, and particularly distressing events of nocturnal hypoglycaemia, are a concern for many people with T2D. Switching to Gla-300, or another second-generation BI analogue, may reduce the frequency of hypoglycaemia throughout the day. In EDITION 2, which included people with T2D uncontrolled on BI plus OADs, Gla-300 provided similar glycaemic control to Gla-100 with lower incidence and rates of nocturnal (00:00–05:59 h) and anytime (24 h) confirmed (≤ 3.9 mmol/L [70 mg/dL]) or severe hypoglycaemia over the full 26-week study period and across both the initial 8-week (titration) and 9–16-week (maintenance) periods [37]. The CONCLUDE study examined switching of BI to IDeg (200 U/mL) or Gla-300, with both insulins demonstrating similar hypoglycaemia risk on the primary outcome [55], thereby affirming the overall results of the BRIGHT study of the superior safety benefits for second-generation BIs.
In the real-world study DELIVER 2, individuals with T2D switching BI therapy to Gla-300 experienced lower hypoglycaemia incidence and adjusted event rates compared with those switching to another BI (IDeg, IDet, or Gla-100). This translated to reduced healthcare utilization [56]. In DELIVER D+, switching from a first-generation (Gla-100 or IDet) to a second-generation (Gla-300 or IDeg) BI analogue provided similar improvements in glycaemic control with similar incidence and event rates of hypoglycaemia observed for Gla-300 versus IDeg [46]. Analysis of the LIGHTNING study indicates that treatment with Gla-300 in a real-life setting is associated with lower rates of severe hypoglycaemia compared with Gla-100 or IDet in those switching from another BI [44]. Modelling analysis from the LIGHTNING study also predicted no significant difference in the rate of severe hypoglycaemia between Gla-300 and IDeg in BI switchers [44].
When switching from Gla-100 to Gla-300, or other second-generation BI, it should be remembered that the insulins may not be bioequivalent and therefore may not be directly interchangeable, so some dose adjustment may be necessary [52]. For Gla-100 and Gla-300, the cause of the difference in insulin dosage is not known, but may be related to a potentially longer residence time of Gla-300 in subcutaneous tissues, which is consistent with the more stable and prolonged PK and PD profiles. The longer residence time might lead to an increased amount of non-specific degradation of the SC depot at that injection site [57]. In support of this theory, the dose differences between Gla-300 and Gla-100 do not have any adverse clinical consequences, as weight gain and hypoglycaemia risk were lower with Gla-300 versus Gla-100 in a pooled meta-analysis of the EDITION studies [10].
Possible Clinical Considerations for Anna
To address Anna’s concerns about the suitability of BI treatment and her perception that her BI therapy is no longer needed, she may benefit from education on the nature of T2D, being reminded that diabetes is a progressive disease and that most individuals with T2D eventually require a BI for optimal glycaemic control [3]. Contrary to what Anna might currently believe, good glycaemic control does not mean the remission of diabetes. The fact that her glycaemic control is good reflects the fact that her current BI therapy has been effective in controlling her blood glucose, whereas a different class of antihyperglycaemic drug may not have been as effective.
Given Anna’s issues with hypoglycaemia, education around hypoglycaemia self-management and recognising and preventing situations in which hypoglycaemia occurs are important so that she can manage her diabetes more closely. Switching to a second-generation BI, such as Gla-300, may help reduce the risk of hypoglycaemia, particularly overnight. As hypoglycaemia is the major reason for Anna’s physician to consider a switch from Gla-100 to a second-generation BI analogue, a reduction in the daily dose of insulin could also be an option, as could a change in administration time from evening to morning. Moreover, further adjustments of the insulin dose might be most effective when done at least once weekly until the achievement of the target of fasting glycaemia without hypoglycaemia. Given the lower risk of hypoglycaemia versus first-generation BI analogues, switching Anna’s treatment to a second-generation BI analogue such as Gla-300 may help her to maintain her HbA1c at her individualized target while reducing her fear of nocturnal hypoglycaemia. Once she gains confidence in the safety of her BI therapy, her dose could be further uptitrated to reach the general guideline recommended target of below 7%.
Case 3: Basal Insulin Treatment in Older People with T2D
Patient profile | |
|---|---|
Name | Lorenzo |
Age | 80 years |
Occupation | Retired |
Lifestyle/activity | Lives with son and daughter-in-law, swims twice a week |
Diabetes duration | 16 years |
BMI | 27 kg/m2 |
Laboratory results | HbA1c 8.9% (74 mmol/mol), blood pressure 130/85 mmHg, eGFR 64 mL/min/1.73 m2, urine ACR normal |
Medical history | Pre-proliferative retinopathy and peripheral neuropathy. Hypertension and lipids are well managed on appropriate treatments. Dementia |
Current medication | A fixed-dose combination of a DPP4 inhibitor and metformin (linagliptin 2.5 mg/metformin 500 mg, twice daily), an SGLT2 inhibitor (dapagliflozin 10 mg once daily) |
Lorenzo’s Concerns
Lorenzo’s caregivers, his son and daughter-in-law, are unsure whether Lorenzo’s glycaemic control is adequate, but are also aware that there may be risks associated with hypoglycaemia considering his age, including worsening dementia symptoms. They are concerned for his safety. Both caregivers have demanding jobs and they and Lorenzo are worried about the complexity of BI therapy, and Lorenzo in particular is reluctant to start an injectable therapy.
Research Evidence
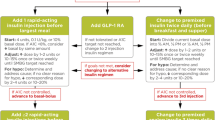
Older people with diabetes are particularly at risk of hypoglycaemia, are more likely to have hypoglycaemia unawareness, are more vulnerable to falls, and also generally have more comorbidities and polypharmacy [58,59,60,61,62]. Consequently, guidelines recommend that glycaemic targets in older individuals may be adjusted to minimise the risk of hypoglycaemia [63]. An HbA1c target of below 8.0% may be most appropriate for older people with mild to moderate cognitive impairment and multiple existing comorbidities [63], and avoiding hypoglycaemia may be important to prevent worsening cognitive decline as severe hypoglycaemia has been linked to increased risk of dementia [64]. However, chronic hyperglycaemia should also be avoided to prevent worsening of complications such as retinopathy and peripheral neuropathy [1, 63, 65]. BI therapy is recommended for older people who need additional glycaemic control; simplified titration algorithms for older individuals are recommended (Fig. 2) [63].
ADA recommendations for glycaemic targets and basal insulin regimen simplification in older people aged over 65 years [63]. *The titration approach shown assumes a person with ‘complex/intermediate’ health status and associated fasting SMPG target. ADL activities of daily living, BI basal insulin, SMPG self-monitored plasma glucose
Second-generation BIs may be particularly relevant in potentially more vulnerable older adults because of lower levels of hypoglycaemia and more flexibility in dosing versus first-generation BIs. In the SENIOR study, Gla-300 demonstrated good efficacy and safety in older people with T2D, particularly in those of advanced age (75 years of age and older). In this older age group, rates of documented symptomatic hypoglycaemia and severe hypoglycaemia were significantly lower with Gla-300 versus Gla-100 [66]. A post hoc analysis of the EDITION 1–3 studies showed that Gla-300 provided similar glycaemic control to Gla-100, with a lower incidence of nocturnal confirmed (≤ 3.9 mmol/L [70 mg/dL]) or severe hypoglycaemia irrespective of age (less than 65 years or at least 65 years) [67]. In a post hoc analysis of the BRIGHT study, Gla-300 was associated with greater reductions in HbA1c versus IDeg-100, without increased incidence or rates of hypoglycaemia in people with T2D who were 70 years of age or older [68].
Real-world evidence from the DELIVER 3 study showed that in older adults (at least 65 years of age) with T2D, switching BI therapy from the first-generation BIs (Gla-100 or IDet) to Gla-300 resulted in similar HbA1c reductions with lower incidence and rates of any hypoglycaemia at 3–6 months fixed follow-up. When assessed by the variable follow-up (i.e. up until discontinuation of therapy or 6 months follow-up, whichever occurred earlier), HbA1c reductions were significantly greater with Gla-300 versus other BIs [69]. The DELIVER HIGH RISK study showed similar HbA1c reductions alongside lower incidence and rates of any hypoglycaemia and lower rates of hypoglycaemia associated with inpatient/emergency department (ED) visits in people who switched BI to Gla-300 versus those who switched to other first-generation BI analogues [70]. Subgroup analyses in high-risk individuals from the DELIVER D+ showed that older people (at least 65 years of age) switching from first-generation BIs to either Gla-300 or IDeg had comparable levels of HbA1c reduction of approximately 0.5% (5.5 mmol/mol) and similar incidence of hypoglycaemia between the second-generation BIs [71]. Modelling subanalyses of another real-world study, the LIGHTNING study, predicted significantly lower rates of severe hypoglycaemia with Gla-300 versus first-generation BIs, Gla-100, and IDet, and similar rates versus IDeg in insulin-naïve older individuals (at least 65 years of age) [44].
Gla-300 is delivered using a pre-filled SoloStar® injection pen to facilitate correct dosing [52], which has been shown to be easy to use [72, 73]. A post hoc analysis of the TAKE CONTROL study including older individuals (at least 65 years of age) demonstrated that self-titration of Gla-300 resulted in similar glycaemic target achievement to physician-led titration, without an increased risk of hypoglycaemia, irrespective of age [74]. There was a trend for more people to reach fasting SMPG targets without hypoglycaemia with self- versus physician-led titration, including those 65 years of age and older [74].
Possible Clinical Considerations for Lorenzo
Although Lorenzo is 80 years of age and has cognitive impairment, he needs better glycaemic control as he still does activities by himself and because raised blood glucose levels will be detrimental for his neuropathy and retinopathy. The symptoms associated with hyperglycaemia may also negatively impact his quality of life [75]. One way to help him improve his glycaemic control is the addition of a second-generation BI analogue, such as Gla-300, as it would provide a relatively simple treatment regimen with a lower risk of hypoglycaemia compared with the first-generation BI, Gla-100. Additionally, Lorenzo and his family/caregivers may benefit from appropriate education to enable them to manage his diabetes. In this case, a starting dose for BI of 0.2 units/kg could be considered [43]. Lorenzo may also benefit from pragmatic HbA1c (i.e. less than 8% [64 mmol/mol]) and home glucose monitoring (i.e. 5.0–8.3 mmol/L [90–150 mg/dL]) targets [63], to ensure a slow and steady drop in glucose and to minimize hypoglycaemia. The ease of insulin administration using a pen device and a clear, simple titration algorithm as reported in TAKE CONTROL (implemented either by a physician or Lorenzo’s caregivers) [34], or as recommended for older people in ADA guidelines [63], should give Lorenzo and his family the confidence to manage his diabetes.
Case 4: Insulin Treatment in People with Morbidities and Increased Risk of Hypoglycaemia
Patient profile | |
|---|---|
Name | Christina |
Age | 68 years |
Occupation | Retired |
Lifestyle/activity | Practises yoga twice a week |
Diabetes duration | 20 years |
BMI | 30 kg/m2 |
Laboratory results | HbA1c 8.8% (73 mmol/mol), blood pressure 125/85 mmHg, eGFR 40 mL/min/1.73 m2, urine ACR 10 mg/mmol |
Medical history | Previously tried a GLP-1 RA and could not tolerate the gastrointestinal side effects. Hypertension is well managed on the appropriate treatment. Stable stage 3A chronic kidney disease (CKD), background diabetic retinopathy |
Current medication | A fixed combination of DPP4 inhibitor combined with metformin (linagliptin 2.5 mg/metformin 500 mg, twice daily), an SGLT2 inhibitor (empagliflozin 10 mg once daily), an ARB (irbesartan 300 mg once daily), a calcium channel blocker (amlodipine 10 mg once daily) and atorvastatin 20 mg once daily |
Christina’s Concerns
Christina is worried about her glycaemic control, as she has never been able to get to HbA1c target since she was diagnosed with T2D 20 years ago. In particular, Christina is concerned about the effect that poor glycaemic control may have on the eye disease and CKD.
Research Evidence
Optimising glycaemic control is essential to prevent the onset of CKD and to reducing the risk of progression [65]. Improving glycaemic control reduces microalbuminuria [76], a strong predictor of renal and cardiovascular disease in people with diabetes [77]. However, optimising glycaemia can be challenging for people with CKD as these individuals have reduced renal gluconeogenesis and renal impairment is associated with altered drug metabolism and clearance, in addition to other factors that contribute to an increased risk of hypoglycaemia [78]. For these reasons, some treatment options such as sulfonylureas are not recommended in individuals with impaired renal function [26]. American Diabetes Association (ADA)/European Association for the Study of Diabetes (EASD) guidelines recommend GLP-1 RA therapy as first injectable treatment, with the caveat that tolerability is an important limitation of this approach [1, 26]. BI is recommended as a suitable therapy option in people with T2D and CKD treated with an SGLT2 inhibitor who need additional glycaemic control [1, 26], although it should be noted that indications for SGLT2 inhibitors in individuals with renal impairment differ between regulatory agencies [79].
Numerous studies support the efficacy and safety of Gla-300 in people with renal impairment. In a post hoc analysis of the EDITION 1–3 studies, glycaemic control with Gla-300 was comparable to that with Gla-100, while rates of hypoglycaemia were lower with Gla-300, in people with T2D and mild-to-moderate renal impairment (estimated glomerular filtration rate below 60 mL/min/1.73 m2) [80]. In a post hoc analysis of the BRIGHT study, Gla-300 was associated with greater reductions in HbA1c versus IDeg, without increased incidence or rates of hypoglycaemia, in people with T2D and impaired renal function (estimated glomerular filtration rate below 60 mL/min/1.73 m2) [81]. Participants using Gla-300 more often tended to change dose frequently than those using IDeg-100, a pattern particularly apparent in the subgroup of people with renal impairment (below 60 mg/min/1.73 m2) [81].
In a real-world setting, the DELIVER HIGH RISK study showed that switching from another BI to Gla-300 provided similar HbA1c reductions with lower incidence and rates of any hypoglycaemia and lower rates of inpatient/ED visit hypoglycaemia compared with switching to a first-generation BI in people with T2D with mild-to-moderate renal impairment [70]. DELIVER D+ showed that switching from first-generation BIs to Gla-300 or IDeg was associated with similar improvements in HbA1c and hypoglycaemia risk between the second-generation BI in adults with T2D and renal impairment [71].
Possible Clinical Considerations for Christina
Since glycaemic control is essential to avoid the progression of kidney disease and Christina is already using metformin, a DPP4 inhibitor, and an SGLT2 inhibitor, a next step to intensify treatment could be a BI. Additionally, she may benefit from education on self-management of her diabetes, managing hypoglycaemia risk and dealing with hypoglycaemic events. Christina should be given clear ‘sick day rules’ regarding the use of metformin, SGLT2, and hypertension medication on days she feels unwell. This is particularly important as an increased risk of diabetic ketoacidosis (DKA) has been reported in people prescribed both SGLT2 and insulin, with potential triggering factors including illness, reduced food or liquid intake, and a reduction in insulin doses [82]. On such days, she might benefit from continued insulin treatment but increasing the frequency of blood glucose monitoring. If required, Christina’s insulin dose could be adjusted. Because Gla-300 has been shown to provide similar glycaemic control and a lower risk of hypoglycaemia compared with the first-generation BI Gla-100, and potentially greater HbA1c reductions without an increase in hypoglycaemia compared with IDeg-100, in individuals with renal impairment, initiating Gla-300 (at a dose of 0.2 unit/kg and titrated once weekly up to the target of fasting glycaemia) may be considered [43]. Pragmatic fasting glucose (6–8 mmol/L [106–145 mg/dL]) and HbA1c (7–7.5% [53–58 mmol/mol]) targets may be most appropriate, with an appropriate follow-up schedule.
Conclusions
Many people with T2D will eventually require a BI to achieve or maintain glycaemic control. The second-generation BI analogues, Gla-300 and IDeg, represent suitable BI options for people needing intensification of their antihyperglycaemic regimens to meet individualised glycaemic targets. In randomised controlled trials of people with T2D, both Gla-300 and IDeg have been shown to provide similar glycaemic control to the first-generation BI analogue, Gla-100, with a lower risk of hypoglycaemia [83]. Furthermore, both randomized controlled trials and real-world evidence studies comparing the two second-generation BI analogues show largely similar glycaemic control and comparable risk of hypoglycaemia for both insulins [43, 46, 56, 71]; however, a lower risk of anytime (24 h) hypoglycaemia was observed with Gla-300 versus IDeg during the titration period in the BRIGHT study [43]. In this article we have focussed on Gla-300, highlighting that its well-established efficacy/effectiveness and safety profiles have also been demonstrated in high-risk groups, including those with impaired renal function and older people, which suggests that Gla-300 is one of the suitable therapy options in these populations [67, 68, 70, 81]. Glycaemic management with Gla-300 may be aided by evidence to show that self-titration is as effective and safe as physician-led titration, which should allow individuals to become more involved with managing their own diabetes.
There are many barriers to glycaemic control in people with T2D. In addition to effective therapy choices, improved communication between healthcare professionals and patients, along with appropriate educational tools and support, may increase patient confidence in the administration and titration of BI dose, ultimately improving glycaemic management and improving treatment adherence, as well as reducing healthcare costs [9].
References
Davies MJ, D’Alessio DA, Fradkin J, et al. Management of hyperglycemia in type 2 diabetes. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2018;2018(41):2669–701.
Garber AJ, Handelsman Y, Grunberger G, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive Type 2 Diabetes Management Algorithm—2020 Executive Summary. Endocr Pract. 2020;26:107–39.
Home P, Riddle M, Cefalu WT, et al. Insulin therapy in people with type 2 diabetes: opportunities and challenges? Diabetes Care. 2014;37:1499–508.
Harris SB, Ekoe JM, Zdanowicz Y, Webster-Bogaert S. Glycemic control and morbidity in the Canadian primary care setting (results of the diabetes in Canada evaluation study). Diabetes Res Clin Pract. 2005;70:90–7.
Russell-Jones D, Pouwer F, Khunti K. Identification of barriers to insulin therapy and approaches to overcoming them. Diabetes Obes Metab. 2018;20:488–96.
Khunti K, Wolden ML, Thorsted BL, Andersen M, Davies MJ. Clinical inertia in people with type 2 diabetes: a retrospective cohort study of more than 80,000 people. Diabetes Care. 2013;36:3411–7.
Selvin E, Parrinello CM, Daya N, Bergenstal RM. Trends in insulin use and diabetes control in the U.S.: 1988–1994 and 1999–2012. Diabetes Care. 2016;39:e33-35.
Aschner P, Gagliardino JJ, Ilkova H, et al. Persistent poor glycaemic control in individuals with type 2 diabetes in developing countries: 12 years of real-world evidence of the International Diabetes Management Practices Study (IDMPS). Diabetologia. 2020;63:711–21.
Berard L, Bonnemaire M, Mical M, Edelman S. Insights into optimal basal insulin titration in type 2 diabetes: results of a quantitative survey. Diabetes Obes Metab. 2018;20:301–8.
Ritzel R, Roussel R, Bolli GB, et al. Patient-level meta-analysis of the EDITION 1, 2 and 3 studies: glycaemic control and hypoglycaemia with new insulin glargine 300 U/ml versus glargine 100 U/ml in people with type 2 diabetes. Diabetes Obes Metab. 2015;17:859–67.
Aronson R, Galstyan G, Goldfracht M, Al Sifri S, Elliott L, Khunti K. Direct and indirect health economic impact of hypoglycaemia in a global population of patients with insulin-treated diabetes. Diabetes Res Clin Pract. 2018;138:35–43.
Brod M, Christensen T, Thomsen TL, Bushnell DM. The impact of non-severe hypoglycemic events on work productivity and diabetes management. Value Health. 2011;14:665–71.
Ratzki-Leewing A, Harris SB, Mequanint S, et al. Real-world crude incidence of hypoglycemia in adults with diabetes: results of the InHypo-DM Study, Canada. BMJ Open Diabetes Res Care. 2018;6:e000503.
Amiel SA, Choudhary P, Jacob P, et al. Hypoglycaemia Awareness Restoration Programme for People with Type 1 Diabetes and Problematic Hypoglycaemia Persisting Despite Optimised Self-care (HARPdoc): protocol for a group randomised controlled trial of a novel intervention addressing cognitions. BMJ Open. 2019;9:e030356.
Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and the Endocrine Society. Diabetes Care. 2013;36:1384–95.
Cryer PE. Mechanisms of hypoglycemia-associated autonomic failure in diabetes. N Engl J Med. 2013;369:362–72.
Cryer PE. Severe hypoglycemia predicts mortality in diabetes. Diabetes Care. 2012;35:1814–6.
Zoungas S, Patel A, Chalmers J, et al. Severe hypoglycemia and risks of vascular events and death. N Engl J Med. 2010;363:1410–8.
McCoy RG, Van Houten HK, Ziegenfuss JY, Shah ND, Wermers RA, Smith SA. Increased mortality of patients with diabetes reporting severe hypoglycemia. Diabetes Care. 2012;35:1897–901.
Moen MF, Zhan M, Hsu VD, et al. Frequency of hypoglycemia and its significance in chronic kidney disease. Clin J Am Soc Nephrol. 2009;4:1121–7.
McCoy RG, Lipska KJ, Van Houten HK, Shah ND. Association of cumulative multimorbidity, glycemic control, and medication use with hypoglycemia-related emergency department visits and hospitalizations among adults with diabetes. JAMA Netw Open. 2020;3:e1919099–e1919099.
Rossi MC, Nicolucci A, Ozzello A, et al. Impact of severe and symptomatic hypoglycemia on quality of life and fear of hypoglycemia in type 1 and type 2 diabetes. Results of the Hypos-1 observational study. Nutr Metab Cardiovasc Dis. 2019;29:736–43.
Ratzki-Leewing A, Parvaresh Rizi E, Harris SB. Family members: the forgotten players in the diabetes care team (The TALK-HYPO Study). Diabetes Ther. 2019;10:2305–11.
Khunti K, Alsifri S, Aronson R, et al. Impact of hypoglycaemia on patient-reported outcomes from a global, 24-country study of 27,585 people with type 1 and insulin-treated type 2 diabetes. Diabetes Res Clin Pract. 2017;130:121–9.
Peyrot M, Barnett AH, Meneghini LF, Schumm-Draeger PM. Insulin adherence behaviours and barriers in the multinational Global Attitudes of Patients and Physicians in Insulin Therapy study. Diabet Med. 2012;29:682–9.
American Diabetes Association. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes—2021. Diabetes Care. 2021;44:S111–24.
Bailey TS, Pettus J, Roussel R, et al. Morning administration of 0.4U/kg/day insulin glargine 300U/mL provides less fluctuating 24-hour pharmacodynamics and more even pharmacokinetic profiles compared with insulin degludec 100U/mL in type 1 diabetes. Diabetes Metab. 2018;44:15–21.
Becker RH, Dahmen R, Bergmann K, Lehmann A, Jax T, Heise T. New insulin glargine 300 Units. mL-1 provides a more even activity profile and prolonged glycemic control at steady state compared with insulin glargine 100 Units. mL-1. Diabetes Care. 2015;38:637–43.
Heise T, Nørskov M, Nosek L, Kaplan K, Famulla S, Haahr HL. Insulin degludec: lower day-to-day and within-day variability in pharmacodynamic response compared with insulin glargine 300 U/mL in type 1 diabetes. Diabetes Obes Metab. 2017;19:1032–9.
American Diabetes Association. Diabetes and Driving. Diabetes Care. 2014;37:S97–103.
UK Driver and Vehicle Licensing Agency. A guide for drivers with insulin treated diabetes who wish to apply for group 2 entitlement (bus and lorry). https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/863517/ins186_a_guide_for_drivers_with_insulin_treated_diabetes.pdf. Accessed 6 Dec 2021.
UK Driver and Vehicle Licensing Agency. A guide to insulin treated diabetes and driving. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/834451/inf294-a-guide-to-insulin-treated-diabetes-and-driving.pdf. Accessed 6 Dec 2021.
Riddle MC, Bolli GB, Home PD, et al. Efficacy and safety of flexible versus fixed dosing intervals of insulin glargine 300 U/mL in people with type 2 diabetes. Diabetes Technol Ther. 2016;18:252–7.
Russell-Jones D, Dauchy A, Delgado E, et al. Take control: a randomized trial evaluating the efficacy and safety of self- versus physician-managed titration of insulin glargine 300 U/mL in patients with uncontrolled type 2 diabetes. Diabetes Obes Metab. 2019;21:1615–24.
Gerstein HC, Yale J-F, Harris SB, Issa M, Stewart JA, Dempsey E. A randomized trial of adding insulin glargine vs. avoidance of insulin in people with Type 2 diabetes on either no oral glucose-lowering agents or submaximal doses of metformin and/or sulphonylureas. The Canadian INSIGHT (Implementing New Strategies with Insulin Glargine for Hyperglycaemia Treatment) Study. Diabet Med. 2006;23:736–42.
Riddle MC, Bolli GB, Ziemen M, Muehlen-Bartmer I, Bizet F, Home PD. New insulin glargine 300 units/mL versus glargine 100 units/mL in people with type 2 diabetes using basal and mealtime insulin: glucose control and hypoglycemia in a 6-month randomized controlled trial (EDITION 1). Diabetes Care. 2014;37:2755–62.
Yki-Järvinen H, Bergenstal R, Ziemen M, et al. New insulin glargine 300 units/mL versus glargine 100 Units/mL in people with type 2 diabetes using oral agents and basal insulin: glucose control and hypoglycemia in a 6-month randomized controlled trial (EDITION 2). Diabetes Care. 2014;37:3235–43.
Bolli GB, Riddle MC, Bergenstal RM, et al. New insulin glargine 300 U/ml compared with glargine 100 U/ml in insulin-naïve people with type 2 diabetes on oral glucose-lowering drugs: a randomized controlled trial (EDITION 3). Diabetes Obes Metab. 2015;17:386–94.
Yale JF, Berard L, Groleau M, Javadi P, Stewart J, Harris SB. TITRATION: a randomized study to assess 2 treatment algorithms with new insulin glargine 300 units/mL. Can J Diabetes. 2017;41:478–84.
Davies M, Bain S, Charpentier G, et al. A randomized controlled, treat-to-target study evaluating the efficacy and safety of insulin glargine 300 U/mL (Gla-300) administered using either device-supported or routine titration in people with type 2 diabetes. J Diabetes Sci Technol. 2019;13:881–9.
Mauricio D, Meneghini L, Seufert J, et al. Glycaemic control and hypoglycaemia burden in patients with type 2 diabetes initiating basal insulin in Europe and the USA. Diabetes Obes Metab. 2017;19:1155–64.
Dalal MR, Kazemi M, Ye F, Xie L. Hypoglycemia after initiation of basal insulin in patients with type 2 diabetes in the united states: implications for treatment discontinuation and healthcare costs and utilization. Adv Ther. 2017;34:2083–92.
Rosenstock J, Cheng A, Ritzel R, et al. More similarities than differences testing insulin glargine 300 units/mL versus insulin degludec 100 units/mL in insulin-naive type 2 diabetes: the randomized head-to-head BRIGHT trial. Diabetes Care. 2018;41:2147–54.
Pettus J, Roussel R, Liz Zhou F, et al. Rates of hypoglycemia predicted in patients with type 2 diabetes on insulin glargine 300 U/ml versus first- and second-generation basal insulin analogs: the Real-World LIGHTNING Study. Diabetes Ther. 2019;10:617–33.
Bailey TS, Zhou FL, Gupta RA, et al. Glycaemic goal attainment and hypoglycaemia outcomes in type 2 diabetes patients initiating insulin glargine 300 units/mL or 100 units/mL: real-world results from the DELIVER Naive cohort study. Diabetes Obes Metab. 2019;21:1596–605.
Sullivan SD, Nicholls CJ, Gupta RA, et al. Comparable glycaemic control and hypoglycaemia in adults with type 2 diabetes after initiating insulin glargine 300 units/mL or insulin degludec: The DELIVER Naive D real-world study. Diabetes Obes Metab. 2019;21:2123–32.
Tibaldi J, Hadley-Brown M, Liebl A, et al. A comparative effectiveness study of degludec and insulin glargine 300 U/mL in insulin-naïve patients with type 2 diabetes. Diabetes Obes Metab. 2019;21:1001–9.
Freemantle N, Jourdan S. Comment on “a comparative effectiveness study of degludec and insulin glargine 300 U/mL in insulin-naïve patients with type 2 diabetes.” Diabetes Obes Metab. 2019;21:1758–9.
Hodish I. Insulin therapy, weight gain and prognosis. Diabetes Obes Metab. 2018;20:2085–92.
Gerstein HC, Bosch J, ORIGIN Trial Investigators, et al. Basal insulin and cardiovascular and other outcomes in dysglycemia. N Engl J Med. 2012;367:319–28.
Marso SP, McGuire DK, Zinman B, et al. Efficacy and safety of degludec versus glargine in type 2 diabetes. N Engl J Med. 2017;377:723–32.
Toujeo (insulin glargine injection) U-300 prescribing information. Bridgewater: sanofi-aventis U.S. LLC; 2019. https://products.sanofi.us/toujeo/toujeo.pdf. Accessed 14 Aug 2020.
Toujeo Summary of Product Characteristics. Frankfurt am Main: Sanofi-Aventis Deutschland GmbH; 2020. https://www.ema.europa.eu/en/documents/product-information/toujeo-epar-product-information_en.pdf. Accessed 14 Aug 2020.
American Diabetes Association. 6. Glycemic targets: standards of medical care in diabetes—2021. Diabetes Care. 2021;44:S73–84.
Philis-Tsimikas A, Klonoff DC, Khunti K, et al. Risk of hypoglycaemia with insulin degludec versus insulin glargine U300 in insulin-treated patients with type 2 diabetes: the randomised, head-to-head CONCLUDE trial. Diabetologia. 2020;63:698–710.
Zhou FL, Ye F, Berhanu P, et al. Real-world evidence concerning clinical and economic outcomes of switching to insulin glargine 300 units/mL vs other basal insulins in patients with type 2 diabetes using basal insulin. Diabetes Obes Metab. 2018;20:1293–7.
Lindauer K, Becker R. Insulin depot absorption modeling and pharmacokinetic simulation with insulin glargine 300 U/mL. Int J Clin Pharmacol Ther. 2019;57:1–10.
Lee PG, Halter JB. The pathophysiology of hyperglycemia in older adults: clinical considerations. Diabetes Care. 2017;40:444–52.
Du YF, Ou HY, Beverly EA, Chiu CJ. Achieving glycemic control in elderly patients with type 2 diabetes: a critical comparison of current options. Clin Interv Aging. 2014;9:1963–80.
Kachroo S, Kawabata H, Colilla S, et al. Association between hypoglycemia and fall-related events in type 2 diabetes mellitus: analysis of a US.. commercial database. J Manag Care Spec Pharm. 2015;21:243–53.
Munshi MN, Segal AR, Suhl E, et al. Frequent hypoglycemia among elderly patients with poor glycemic control. Arch Intern Med. 2011;171:362–4.
Zammitt NN, Frier BM. Hypoglycemia in type 2 diabetes: pathophysiology, frequency, and effects of different treatment modalities. Diabetes Care. 2005;28:2948–61.
American Diabetes Association. 12. Older adults: standards of medical care in diabetes—2021. Diabetes Care. 2021;44:S168–79.
Lee AK, Rawlings AM, Lee CJ, et al. Severe hypoglycaemia, mild cognitive impairment, dementia and brain volumes in older adults with type 2 diabetes: the Atherosclerosis Risk in Communities (ARIC) cohort study. Diabetologia. 2018;61:1956–65.
American Diabetes Association. 11. Microvascular complications and foot care: standards of medical care in diabetes—2021. Diabetes Care. 2021;44:S151–67.
Ritzel R, Harris SB, Baron H, et al. A randomized controlled trial comparing efficacy and safety of insulin glargine 300 units/mL versus 100 Units/mL in older people with type 2 diabetes: results from the SENIOR Study. Diabetes Care. 2018;41:1672–80.
Yale JF, Aroda VR, Charbonnel B, et al. Glycaemic control and hypoglycaemia risk with insulin glargine 300 U/mL versus glargine 100 U/mL: a patient-level meta-analysis examining older and younger adults with type 2 diabetes. Diabetes Metab. 2020;46(2):110–18.
Bolli GB, Cheng A, Charbonnel B, et al. Glycaemic control and hypoglycaemia risk with insulin glargine 300 U/mL and insulin degludec 100 U/mL in older participants in the BRIGHT trial. Diabetes Obes Metab. 2021;23(7):1588–93.
Bailey TS, Wu J, Zhou FL, et al. Switching to insulin glargine 300 units/mL in real-world older patients with type 2 diabetes (DELIVER 3). Diabetes Obes Metab. 2019;21:2384–93.
Sullivan SD, Freemantle N, Menon AA, et al. High-hypoglycaemia-risk patients with type 2 diabetes on first-generation basal insulins (BIs) in the US have a lower risk of hypoglycemia after 1 year following switch from insulin glargine 300 U/mL versus first-generation BIs (DELIVER- High Risk). Poster 133-LB presented at ADA 2019, San Francisco, CA, June 7–11 2019.
Sullivan SD, Bailey TS, Roussel R, et al. Clinical outcomes in real-world patients with type 2 diabetes switching from first- to second-generation basal insulin analogues: Comparative effectiveness of insulin glargine 300 units/mL and insulin degludec in the DELIVER D+ cohort study. Diabetes Obes Metab. 2018;20:2148–58.
Klonoff D, Nayberg I, Erbstein F, Cali A, Brulle-Wohlhueter C, Haak T. Usability of the Gla-300 injection device compared with three other commercialized disposable insulin pens: results of an interview-based survey. J Diabetes Sci Technol. 2015;9:936–8.
Pohlmeier H, Berard L, Brulle-Wohlhueter C, et al. Ease of use of the insulin glargine 300 U/mL pen injector in insulin-naive people with type 2 diabetes. J Diabetes Sci Technol. 2017;11:263–9.
Strojek K, Bigot G, Bonnemaire M et al. Self- vs. physician-led titration of insulin glargine 300 U/mL (Gla-300)—improved or comparable efficacy at week 24 without increased risk of hypoglycemia, irrespective of age (<65 or ≥65 years)—TAKE CONTROL. Diabetes Care. 2018;67: 81.
Testa MA, Simonson DC. Health economic benefits and quality of life during improved glycemic control in patients with type 2 diabetes mellitus: a randomized, controlled, double-blind trial. JAMA. 1998;280:1490–6.
DCCT/EDIC research group. Effect of intensive diabetes treatment on albuminuria in type 1 diabetes: long-term follow-up of the Diabetes Control and Complications Trial and Epidemiology of Diabetes Interventions and Complications study. Lancet Diabetes Endocrinol. 2014;2:793–800.
Mann JF, Yi QL, Gerstein HC. Albuminuria as a predictor of cardiovascular and renal outcomes in people with known atherosclerotic cardiovascular disease. Kidney Int Suppl. 2004;66:S59–62.
Alsahli M, Gerich JE. Hypoglycemia in patients with diabetes and renal disease. J Clin Med. 2015;4:948–64.
Gorriz JL, Navarro-Gonzalez JF, Ortiz A, et al. Sodium-glucose cotransporter 2 inhibition: towards an indication to treat diabetic kidney disease. Nephrol Dial Transplant. 2020;35:i13–23.
Escalada FJ, Halimi S, Senior PA, et al. Glycaemic control and hypoglycaemia benefits with insulin glargine 300 U/mL extend to people with type 2 diabetes and mild-to-moderate renal impairment. Diabetes Obes Metab. 2018;20:2860–8.
Haluzik M, Cheng A, Müller-Wieland D, et al. Differential glycaemic control with basal insulin glargine 300 U/mL versus degludec 100 U/mL according to kidney function in type 2 diabetes—a subanalysis from the BRIGHT trial. Diabetes Obes Metab. 2020;22(8):1369–77.
Rosenstock J, Ferrannini E. Euglycemic diabetic ketoacidosis: a predictable, detectable, and preventable safety concern with SGLT2 inhibitors. Diabetes Care. 2015;38:1638–42.
Roussel R, Ritzel R, Boelle-Le Corfec E, Balkau B, Rosenstock J. Clinical perspectives from the BEGIN and EDITION programmes: trial-level meta-analyses outcomes with either degludec or glargine 300U/mL vs glargine 100U/mL in T2DM. Diabetes Metab. 2018;44:402–409.
Acknowledgements
Funding
Support for this work and the journal’s Rapid Service Fee was provided by Sanofi.
Medical Writing and Editorial Assistance
Medical writing and editorial assistance was provided by Hannah Brown, PhD, and Simon Rees, PhD, of Fishawack Communications Ltd, and was funded by Sanofi.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Stewart B. Harris, Erika B. Parente and Janaka Karalliedde contributed to interpretation of published data to develop the article content, reviewed all drafts of the article and approved the final version for submission.
Disclosures
Stewart B. Harris—Consultant: Abbott, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Janssen, Merck, Sanofi; Research support: Abbott, AstraZeneca, Boehringer Ingelheim, Eli Lilly, HLS Therapeutics, Janssen, Novo Nordisk, Sanofi. Erika B. Parente—Received lecture honorariums and consulting fees from Eli Lilly, Abbott, AstraZeneca, Sanofi, Boehringer Ingelheim and is an advisory board member of Sanofi. Janaka Karalliedde—Served on scientific advisory boards and received honoraria or consulting fees from Eli Lilly, Novo Nordisk, Sanofi, Napp, AstraZeneca, Boehringer Ingelheim; and has received grants/research support from Sanofi and AstraZeneca.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Harris, S.B., Parente, E.B. & Karalliedde, J. Clinical Use of Insulin Glargine 300 U/mL in Adults with Type 2 Diabetes: Hypothetical Case Studies. Diabetes Ther 13, 913–930 (2022). https://doi.org/10.1007/s13300-022-01247-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-022-01247-7