Abstract
Introduction
Severe hypoglycemic events (SHE) represent a clinical and economic burden in patients with diabetes. Nasal glucagon (NG) is a novel treatment for SHEs with similar efficacy, but with a usability advantage over injectable glucagon (IG) that may translate to improved economic outcomes. The economic implications of this usability advantage on SHE-related spending in Spain were explored in this analysis.
Methods
A cost-offset and budget impact analysis (BIA) was conducted using a decision tree model, adapted for the Spanish setting. The model calculated average costs per SHE over the SHE treatment pathway following a treatment attempt with IG or NG. Analyses were performed separately in three populations with insulin-treated diabetes: children and adolescents (4–17 years) with type 1 diabetes (T1D), adults with T1D and adults with type 2 diabetes (T2D), with respective population estimates applied in BIA. Treatment probabilities were assumed to be equal for IG and NG, except for treatment success following glucagon administration. Epidemiologic and cost data were obtained from Spanish-specific sources. BIA results were presented at a 3-year time horizon.
Results
On a per SHE level, NG was associated with lower costs compared to IG (children and adolescents with T1D, EUR 820; adults with T1D, EUR 804; adults with T2D, EUR 725). Lower costs were attributed to reduced costs of professional medical assistance in patients treated with NG. After 3 years, BIA showed that relative to IG, the introduction of NG was projected to reduce SHE-related spending by EUR 1,158,969, EUR 142,162,371, and EUR 6,542,585 in children and adolescents with T1D, adults with T1D, and adults with insulin-treated T2D, respectively.
Conclusions
In Spain, the usability advantage of NG over IG translates to potential cost savings per SHE in three populations with insulin-treated diabetes, and the introduction of NG was associated with a lower budget impact versus IG in each group.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Severe hypoglycemia events (SHEs) place a significant burden on the individual, caregivers of people with diabetes and the wider healthcare system. During a SHE, people with diabetes often rely on a non-medically trained person to successfully deliver a full dose of glucagon in a stressful, emergency situation. |
Injectable glucagon (IG) and nasal glucagon (NG) are recommended treatments for SHEs. While IG and NG have similar efficacy, evidence suggests NG is easier to administer as no reconstitution step is required. |
The present analysis aimed to quantify the economic impact of the usability advantage of NG versus IG in three populations with insulin-treated diabetes in Spain who are at risk of SHE. |
What was learned from the study? |
Relative to IG or no glucagon, the introduction of NG in Spain resulted in lower SHE-related costs at a per event and population level in three populations with insulin-treated diabetes. |
Findings suggest the usability advantage of NG versus IG has the potential to reduce the economic burden of SHEs in Spain. |
Introduction
Hypoglycemia occurs in people with diabetes when blood glucose levels are low [1]. Impaired glucose counter-regulation can result in falling plasma glucose concentrations when there is an excess of insulin. Falling plasma glucose concentrations can progress to hypoglycemia because of compromised physiologic defenses [2]. Hypoglycemia can be non-severe and resolved by the individual [3]. In a severe hypoglycemic event (SHE) the individual may require assistance by a third-party to resolve the event, which can include onsite treatment by a caregiver (CG) or acquaintance, emergency medical services (EMS), transport to and treatment in an emergency department (ED), and/or inpatient hospitalization [4, 5].
In Spain, SHEs have been estimated to occur at a rate between 0.90 and 1.50 per person per year in people with type 1 diabetes (T1D), and between 0.30 and 0.63 per person per year in people with type 2 diabetes (T2D) [6]. Furthermore, total cost per SHE was estimated to be EUR 409.97 in people with T1D and EUR 713.10 in people with T2D [6]. In 2016, the estimated total cost of SHEs in Spain was EUR 293 million per year, accounting for nearly half of the total cost of insulin-related hypoglycemia (EUR 662 million) [5]. The high economic burden of SHEs can partly be attributed to hospitalization, with an average length of stay due to hypoglycemia in 2015 being 8.8 and 6.7 days in men and women, respectively [7]. Additionally, SHEs can have a large emotional impact on patients and CGs, for which an economic cost cannot be quantified. The scope of SHE emotional impact was evaluated in 377 Spanish people with T1D or insulin-treated T2D and CGs, and over a third of participants agreed/strongly agreed that experiencing/witnessing the most recent SHE made them feel unprepared, scared, and/or helpless. Furthermore, between 17.9% and 23.9% of patients with diabetes, and between 27.6% and 32.3% of CGs reported that the most recent SHE impacted their mood or emotional status [8].
Glucagon is used in the treatment of hypoglycemia, and in diabetes treatment guidelines injectable glucagon (IG) and nasal glucagon (NG) have been recommended for the treatment of SHEs [9]. IG requires reconstitution and injection with a needle, typically by a CG or another non-medically trained person during a stressful emergency situation [10]. NG is a ready-to-use, compact, portable, single-use drug device combination that contains 3 mg of nasally administered dry powder. Unlike with IG, no reconstitution step is required. Glucagon is absorbed passively through the nasal mucus; therefore, patients do not need to inhale, and NG can be used for the treatment of cognitively impaired and unconscious patients [11]. In clinical trials, NG was shown to have efficacy similar to IG in children aged 4 years or over, adolescents, and adults with T1D and T2D [12,13,14]. Real-world studies have demonstrated that NG was effective in treating SHE events in adults with T1D, and at treating moderate hypoglycemic events in children, adolescents, and adults with T1D [15, 16]. All adults who experienced a SHE awakened or returned to a normal status within 15 min of receiving NG, and no additional medical assistance was required [16].
Successful delivery of a full dose of glucagon is important to quickly treat the SHE to prevent further complications [10]. However, CGs of people with diabetes experience difficulties in administering IG, even after receiving education in IG administration [17]. Similarly, acquaintances of people with diabetes without IG education may also attempt to treat an individual experiencing a SHE; however, they also may find IG administration challenging [18]. A 2017 usability study of 31 people without formal medical training (16 CGs of people with diabetes and 15 acquaintances who had declared their willingness to help in an emergency) showed low rates of success with IG in simulated SHEs, especially amongst acquaintances [18]. Fifty percent of CGs and 80% of acquaintances delivered no glucagon when provided with IG. Only 13% of CGs delivered a full dose of glucagon using IG and no acquaintances delivered a full dose (the remaining participants delivered a partial dose) [18]. By comparison, the same study showed treatment of SHEs using NG had significantly higher rates of delivery of a full dose of glucagon, presenting a usability advantage over IG [18]. Findings showed 94% of CGs and 93% of acquaintances delivered a full dose of NG [18]. Similar results in a separate study, including 32 people educated to use IG or NG (trained users) and 33 without IG or NG training, confirm these findings [19]. Additionally, people often do not carry IG with them, frequently storing kits at home, and therefore may not have IG available in the event of a SHE [20].
The usability advantage of NG over IG, involving a higher probability of successful treatment by CGs or acquaintances, was expected to be associated with a reduced need to use professional medical help, e.g., EMS or ED [18], resulting in reduced SHE-related treatment costs. NG administration was therefore hypothesized to be associated with cost-offsets versus IG and to have the potential to reduce overall SHE-related spending for the healthcare payer.
The present analysis aimed to evaluate the economic impact of the usability advantage of NG over IG in the Spanish setting, for the current reimbursed population (children and adolescents aged under 18 years old, currently treated with insulin and with high risk of severe hypoglycemia) and other alternative hypothetical scenarios (adults with either T1D or insulin-treated T2D). Across the SHE treatment pathway, over a 3-year period, costs were compared between treatments to assess per event costs (cost-offset) and the budget impact of introducing NG in Spain.
Methods
Model Overview
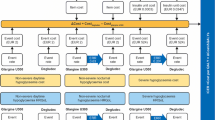
A cost-offset and budget impact analysis (BIA) was conducted using a decision tree model developed in Microsoft Excel, in line with published guidelines and previously published US analyses [21, 22]. A diagram of the decision tree model representing the SHE treatment pathway is presented in Fig. 1. The SHE treatment pathway was informed by reviews of the published literature, treatment guidelines for severe hypoglycemia, and emergency care pathways [21]. Typically, SHEs are acute in nature and long-term costs/outcomes related to SHEs are rare; therefore, this analysis took a short-term perspective, and no long-term costs were modelled. Cost-offset analysis combined decision and cost data to calculate the mean cost per SHE following a treatment attempt with IG or NG. Next, these data were applied in BIA to project population-level costs and potential cost impact over a 3-year time horizon. The BIA compared two scenarios. The current scenario, in which the only potential options for SHEs are IG or no glucagon treatment, compared to the future scenario, in which NG would be incorporated into the previous options. All analyses were carried out separately for three populations with insulin-treated diabetes: children and adolescents (4–17 years) with T1D (current reimbursed population in Spain), adults (aged 18 years or over) with T1D and, adults (aged 18 years or over) with T2D from the perspective of the Spanish National Health System.
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Model Inputs
Clinical Data
Clinical input data related to the probability of treatment decision and outcomes were sourced from published literature (Table 1). These data include Spanish-specific inputs from the Conversations and Reactions Around Severe Hypoglycemia (CRASH) Study, a global survey of people with T1D or insulin-treated T2D and CGs [8]. Assumptions based on previously published analyses were used where no data were available for an item [21]. Probabilities associated with treatment and outcomes were assumed to be equal for IG and NG, except for treatment success following glucagon administration by CGs and acquaintances. It was also assumed that either IG or NG would be used to attempt treatment and only one treatment attempt would be made, after which the CG or acquaintance would seek professional medical help.
Economic Data
Direct costs associated with treatment and resource use were sourced from national databases and published studies capturing real-life conditions in Spain (Table 2). Resource use included the cost of EMS (release on scene and transport to ED), ED treatment, inpatient care, and general practitioner (GP) visit. Consumer price inflation data for Spain were sourced from the Organization for Economic Co-operation and Development [23]; 2012 resource use costs were inflated by 4.9% to align with the latest available year at the time of analysis (2019) [23].
In the base case analyses, the same unit costs associated with medical resource use were assumed for all treatment pathway decision and outcomes. Glucagon costs were based on public prices [24].
Epidemiological Data
For the budget impact analysis, the target population in each group was estimated using the most recent data from publicly available sources (Table 3). Population data were obtained from the Spanish National Institute of Statistics. Next, rates for diagnosed diabetes were applied to population estimates. For this analysis, it was assumed all people with T1D were treated with insulin. To calculate the population with T2D treated with insulin, first a treatment rate of 80.6% [25] was applied to the total number of cases with T2D, and of these it was assumed 23.5% [26] would be treated with insulin.
To calculate the number of SHEs per year, separate SHE incidence rates were applied to each population. Background mortality was not indexed by age or gender in the model; therefore, the same rate was applied across all groups in each year of analysis. This approach represented a simplifying assumption, although it was considered appropriate for the short-term time horizon.
Market Shares
For the BIA, it was assumed the distribution of market shares varied between each group (Table 4). In all groups, a proportion of patients were assumed not to own any glucagon; this was projected to decrease overtime in both populations with T1D and remain constant in the population with insulin-treated T2D. For those who did own a glucagon kit (IG or NG), a positive uptake for NG was assumed each year of analysis with corresponding decreases for IG.
Sensitivity Analyses
One-way sensitivity analyses were performed to explore uncertainty around the parameters and assumptions of the analysis (Table 5). As with the base case analyses, sensitivity analyses were conducted separately for the three populations. Probabilities associated with the model pathway were explored using alternative values. Changes in treatment success probabilities, probabilities of taking action following a successful or unsuccessful treatment attempt, and in probabilities of professional medical assistance were included in the sensitivity analyses. Costs incurred with the use of professional medical help were varied using alternative values from the literature for EMS treatment with release on scene, EMS treatment with transport to hospital, ED treatment, and GP follow-up. Additionally, an analysis was conducted using the lower cost estimates for all emergency professional medical help and analysis using only higher cost estimates was also conducted.
For budget impact analyses, sensitivity analyses with the market share of IG increased or decreased by 10% in each year versus the base case were also conducted.
Results
Cost-offset Analysis
Overall, results of the base case analysis showed that on a per SHE level, the use of NG was associated with lower mean event costs compared with IG across each population.
While pharmacologic costs were higher where treatment was attempted with NG than where treatment was attempted with IG (EUR 68 vs. EUR 14), costs were offset by medical resource use (EMS treatment, EMS transport to ED, ED treatment, and inpatient stay). There was no difference in the cost associated with follow-up GP visits in both arms across each group.
Base case results of the cost-offset analysis in children and adolescents with T1D are presented in (Fig. 2). Mean total costs were EUR 967 per SHE treated with IG, compared with EUR 147 per SHE treated with NG, yielding a cost-offset of approximately EUR 820, or an 84.8% decrease in cost. Costs associated with EMS call-out and release on the scene were EUR 138 lower with NG than IG, and EMS transport to ED was associated with EUR 172 reduction with NG than IG. ED treatment with no inpatient stay was EUR 503 lower with NG than IG, and the costs associated with hospitalization including prior ED stay were EUR 61 lower with NG than IG (Fig. 2).
Sensitivity analyses showed that relative to IG, NG was associated with cost-offset with all differing assumptions (Supplementary Material Table S1). Decision/outcome probabilities and cost of medical treatment drove cost-offset results.
The scenario where the probability of taking action after successful treatment attempt action was increased from 0% (base case) to 75% (SA6) notably reduced the incremental cost between IG and NG (base case EUR 820 vs. SA6 EUR 129). In contrast, scenarios in which costs associated with medical resource use were increased (e.g., cost of inpatient care set to EUR 2,318.21 [SA24]) made the cost savings with NG, compared to IG, even greater than in the base case (up to EUR 929).
When adults with T1D were considered in the analysis, mean total cost was EUR 943 per SHE treated with IG compared with EUR 138 per SHE treated with NG, yielding a cost-offset of approximately EUR 804, or an 85.3% decrease in cost.
Cost associated with EMS call-out and release on the scene were EUR 207 lower with NG than IG, and EMS transport to ED was associated with EUR 227 reduction with NG than IG. ED treatment with no inpatient stay was EUR 378 lower with NG than IG, and the cost associated with hospitalization including prior ED stay were EUR 46 lower with NG than IG (Fig. 3).
As in children and adolescents with T1D, findings from the sensitivity analyses revealed that compared to IG, NG was associated with potential cost-offset in all differing assumptions (Supplementary Material Table S2).
In adults with insulin-treated T2D, mean total cost per SHE was EUR 865 with IG compared to EUR 140 with NG, which yielded a cost-offset of approximately EUR 725, or an 83.8% decrease in costs.
Costs associated with EMS call-out and release on the scene were EUR 235 lower with NG than IG, and costs of EMS transport to ED were EUR 177 lower with NG than IG. Treatment in the ED was less frequently required with NG leading to a mean EUR 328 lower cost than IG. Cost associated with hospitalization was EUR 40 lower with NG than IG (Fig. 4).
As with the populations with T1D, sensitivity analyses showed that compared to IG, NG was associated with potential cost savings in all analyses (Supplementary Material Table S3).
Budget Impact Analysis
The estimated target population along with the base case results for the 3-year budget impact associated with SHEs following a treatment attempt with IG or NG for each group considered in the analysis is presented in Table 6.
At the population level, pharmacologic costs were higher in the future scenario (NG available) relative to the current scenario (no NG available) across all groups. When medical care costs were considered, the introduction of NG to the Spanish market was projected to lower the budget impact across each population.
For 28,485 children and adolescents with T1D, the projected total cost attributed to SHE related treatment was estimated at EUR 6,472,619 with the current scenario (no available NG) and EUR 5,313,650 with the new scenario (available NG). At 3 years, this translated to a cost reduction in total SHE-related spending of 17.9% (EUR 1,158,969). For 304,247 adults with T1D, the projected total cost attributed to SHE related treatment was estimated at EUR 833,672,302 with the current scenario and EUR 691,509,931 with the new scenario. At 3 years, this translated to a cost reduction in total SHE-related spending of 17.1% (EUR 142,162,371). For 518,647 adults with insulin-treated T2D, the projected total cost attributed to SHE related treatment was estimated at EUR 440,397,141 with the current scenario and EUR 433,854,556 with the new scenario. At 3 years, this translated to a cost reduction in total SHE-related spending of 1.5% (EUR 6,542,585).
For each group, the cost saving was associated with reduced medical care costs following a reduced need for professional emergency treatment as a result of better chances of successful treatment by CGs or acquaintances with NG.
In children and adolescents with T1D (Supplementary Material Table S4), sensitivity analyses showed the 3-year budget impact was higher in the future scenario (NG available) versus the current scenario (no NG available) when the probability of taking action after a successful treatment attempt was changed to 75% instead of 0% (SA6) as in the base case, the probability of successful treatment by a CG increased for IG (78%) and NG (100%) (SA2), and when the cost of ED treatment was set to EUR 206.74 (SA21). Among adults with T1D (Supplementary Material Table S5) and insulin treated T2D (Supplementary Material Table S6), NG was associated with a potential reduction in total SHE-related spending versus IG in almost all sensitivity analyses; the only exception was for adults with insulin-treated T2DM when the probability of taking action after a successful treatment attempt was changed to 75% instead of 0% (SA6).
Discussion
During a SHE, people with diabetes are often dependent on others to administer a quick and successful dose of glucagon to recover. While IG and NG have similar efficacy, evidence suggests NG is easier to administer as no reconstitution step is required.
The present analysis quantified the economic impact of this usability advantage of NG over IG. To provide a comprehensive view of the context in Spain, the current reimbursed population (children and adolescents aged 4–17 years with T1D) and two additional populations who are at risk of experiencing SHEs (adults with either T1D or insulin-treated T2D) were considered in the analysis.
Findings suggest the usability advantage of NG over IG can lead to cost savings in the treatment of SHEs. The usability advantage of NG has been shown in a simulation study to result in a higher probability of successful treatment of SHEs, regardless of whether treatment was attempted by a CG or acquaintance [18]. Successful delivery of a full glucagon dose may reduce the need for professional medical emergency assistance, which is associated with high additional costs. In the cost-offset analysis, compared to IG, treatment with NG was associated with potential cost savings in each population with insulin-treated diabetes. Mean total cost per SHE treated with IG in children and adolescents with T1D, adults with T1D and, adults with insulin-treated T2D was EUR 967, EUR 943, and EUR 865, respectively. Previously published Spanish direct costs per SHE ranged from EUR 172 to EUR 717 [6, 27]. Owing to the dominant rate of successful NG administration relative to IG, potential cost savings were seen in multiple steps of the SHE treatment pathway. Patients treated with NG had lower costs associated with all professional medical assistance than IG, except GP visit costs which were assumed to be the same. Therefore, the cost-offset evaluation showed, compared to IG, that NG is likely to reduce costs directly associated with managing SHEs.
In Spain, SHEs were estimated to cost EUR 293 million per year in 2016, the majority of which is attributed to professional emergency assistance and hospitalization [5]. Therefore, through the projected reduction of the need for additional medical treatment, owing to the usability advantage of NG over IG, the per event cost of SHEs can be greatly reduced [18]. On a cost per event level, the cost-offset analysis projected decreases in professional medical assistance and hospitalization when using NG compared with IG. On a population level, the introduction of NG would decrease healthcare costs in the insulin-treated population compared to a world with IG or no glucagon. In the present analysis, compared with a scenario where the only glucagon available is IG, the introduction of NG was projected to result in cost savings in each population. Cost savings were higher in adult patients with T1D because of higher SHE rates compared with the other two populations with insulin-treated diabetes. However, while the rate of SHEs is lower in adults with insulin-treated T2D, as a progressive disease, the risk of SH will increase as the person with T2D requires more insulin [2]. Furthermore, T2D is more prevalent than T1D in the Spanish population, and therefore the total number of SHEs in people with T2D would be higher than in people with T1D [2]. The reduced overall cost following the introduction of NG was due to reduced total medical care costs as a result of increased rates of successful treatment and a subsequent reduced requirement for professional emergency treatment.
As with all modeling studies, the model and present analysis were not without limitations. Assumptions of certain SHE-related treatment decisions and outcomes were used because of a paucity of Spanish data. In the treatment pathway, it was assumed that following successful glucagon administration no further action would be taken. This approach is supported by two studies carried out in the real-world setting [15, 16]. Deeb et al. showed that of 33 hypoglycemic events in 14 children and adolescents with T1D, all cases reverted to normal status within 30 min after NG administration [15]. Similar observations were reported by Seaquist et al.: patients awakened or returned to normal status within 15 min of NG administration without additional external medical help in all 12 cases of SHE in adults with T1D, indicating no further action would be required [16]. Conversely, it was assumed that further action would be taken after an unsuccessful attempt at glucagon administration and the SHE would require emergency medical assistance. In the CRASH study, injectable glucagon (administered by a CG or a non-medical person) was reported as an action during a SHE [8]; however, it is unknown whether this treatment resolved the SHE. Furthermore, it was not possible to ascertain if any other actions taken (e.g., call EMS) were in response to treatment success or failure. Therefore, for this analysis, the actions of all CRASH study participants who reported glucagon use during a SHE acted as a reference. This was based on the assumption that these data provide a general average of the potential actions during a SHE in routine practice, irrespective of treatment success or failure.
Additionally, while the option to call for EMS was included in treatment pathway, evidence from the CRASH study suggests that in addition to calling EMS, some CGs or acquaintances may also call a healthcare professional (HCP) during a SHE [8]. However, as a result of uncertainty around the next steps taken by the CG or acquaintance (e.g., would EMS still be required?), inclusion of this pathway in the model was challenging. However, integrating this option into the model would most likely reduce the average cost following a SHE as immediate follow-up with medical services, other than emergency services, after glucagon administration would incur minimal cost. Furthermore, the decision probabilities for post-event care (i.e., follow-up visit with a GP) were based on the proportion of CRASH study participants who had an additional HCP visit to discuss the most recent SHE [8]. However, it is unknown whether this additional visit occurred with a GP, an endocrinologist, or some other HCP; therefore, this variable was used as a proxy in the model. For the budget impact analysis, the most recently available publications (data up to 2017) providing incidence of SHEs in Spain at the time of the analysis were selected. While SHE frequency may decrease as the use of continuous glucose monitoring, flash glucose monitoring, and continuous subcutaneous insulin infusion systems widens [9], the present analysis does not consider changes in incidence rates over time.
Conclusion
The present analysis suggests that the usability advantage of NG over IG translates to potential cost savings per SHE, owing to reduced overall SHE-related spending as SHEs would be more likely to be resolved without professional medical assistance (i.e., EMS, ED treatment, and hospitalization). The introduction of NG in Spain was associated with a lower budget impact relative to IG or no glucagon in three populations with insulin-treated diabetes.
References
Cryer PE, Davis SN, Shamoon H. Hypoglycemia in diabetes. Diabetes Care. 2003;26:1902–12.
Cryer PE. The barrier of hypoglycemia in diabetes. Diabetes. 2008;57:3169–76.
American Diabetes Association. 6. Glycemic targets: standards of medical care in diabetes 2021. Diabetes Care. 2021;44:S73–84.
Barranco RJ, Gomez-Peralta F, Abreu C, et al. Incidence and care-related costs of severe hypoglycaemia requiring emergency treatment in Andalusia (Spain): the PAUEPAD project. Diabet Med. 2015;32:1520–6.
Parekh W, Hoskins N, Baker-Knight J, Ramirez-de-Arellano A, Mezquita-Raya P. The economic burden of insulin-related hypoglycemia in Spain. Diabetes Ther. 2017;8:899–913.
Núñez M, Díaz S, Dilla T, Reviriego J, Pérez A. Epidemiology, quality of life, and costs associated with hypoglycemia in patients with diabetes in Spain: a systematic literature review. Diabetes Ther. 2019;10:375–92.
Orozco-Beltrán D, Guillen-Mollá A, Cebrián-Cuenca AM, et al. Hospital admissions trends for severe hypoglycemia in diabetes patients in Spain, 2005 to 2015. Diabetes Res Clin Pract. 2021;171:108565.
Ampudia-Blasco FJ, Artime E, Díaz S, et al. Conversations and reactions around severe hypoglycaemia (CRASH): Spanish results of a global survey of people with type 1 diabetes or insulin-treated type 2 diabetes and caregivers. Endocrinol Diabetes Nutr. 2021;68:557–66.
Reyes-García R, Mezquita-Raya P, Moreno-Pérez Ó, et al. Executive summary: Position document: evaluation and management of hypoglycemia in the patient with diabetes mellitus 2020. Diabetes mellitus working group of the Spanish Society of Endocrinology and Nutrition. Endocrinol Diabetes Nutr. 2021;68:270–6.
Kedia N. Treatment of severe diabetic hypoglycemia with glucagon: an underutilized therapeutic approach. Diabetes Metab Syndr Obes. 2011;4:337–46.
Thieu VT, Mitchell BD, Varnado OJ, Frier BM. Treatment and prevention of severe hypoglycaemia in people with diabetes: current and new formulations of glucagon. Diabetes Obes Metab. 2020;22:469–79.
Sherr JL, Ruedy KJ, Foster NC, et al. Glucagon nasal powder: a promising alternative to intramuscular glucagon in youth with type 1 diabetes. Diabetes Care. 2016;39:555–62.
Rickels MR, Ruedy KJ, Foster NC, et al. Intranasal glucagon for treatment of insulin-induced hypoglycemia in adults with type 1 diabetes: a randomized crossover noninferiority study. Diabetes Care. 2016;39:264–70.
Seaquist ER, Khunti K, Zhang MX, et al. Nasal glucagon reversed insulin-induced hypoglycemia in adults with diabetes: a pooled analysis. Diabetes. 2020;69(Supplement_1):1074-P.
Deeb LC, Dulude H, Guzman CB, et al. A phase 3 multicenter, open-label, prospective study designed to evaluate the effectiveness and ease of use of nasal glucagon in the treatment of moderate and severe hypoglycemia in children and adolescents with type 1 diabetes in the home or school setting. Pediatr Diabetes. 2018;19:1007–13.
Seaquist ER, Dulude H, Zhang XM, et al. Prospective study evaluating the use of nasal glucagon for the treatment of moderate to severe hypoglycaemia in adults with type 1 diabetes in a real-world setting. Diabetes Obes Metab. 2018;20:1316–20.
Harris G, Diment A, Sulway M, Wilkinson MJPDI. Glucagon administration–underevaluated and undertaught. Pract Diabetes Int. 2001;18:22–5.
Yale J-F, Dulude H, Egeth M, et al. Faster use and fewer failures with needle-free nasal glucagon versus injectable glucagon in severe hypoglycemia rescue: a simulation study. Diabetes Technol Ther. 2017;19:423–32.
Settles JA, Gerety GF, Spaepen E, Suico JG, Child CJ. Nasal glucagon delivery is more successful than injectable delivery: a simulated severe hypoglycemia rescue. Endocr Pract. 2020;26:407–15.
Haymond MW, Liu J, Bispham J, Hickey A, McAuliffe-Fogarty AH. Use of glucagon in patients with type 1 diabetes. Clin Diabetes. 2019;37:162–6.
Pöhlmann J, Mitchell BD, Bajpai S, Osumili B, Valentine WJ. Nasal glucagon versus injectable glucagon for severe hypoglycemia: a cost-offset and budget impact analysis. J Diabetes Sci Technol. 2019;13:910–8.
Sullivan SD, Mauskopf JA, Augustovski F, Jaime Caro J, Lee KM, Minchin M, et al. Budget impact analysis-principles of good practice: report of the IPSOR 2012 Budget Impact Analysis Good Practice II Task Force. Value Health. 2014;17:5–14.
Organisation for Economic Co-operation and Development (OECD). OECD.Stat/prices and purchasing power parities/consumer and producer price indices/g20—cpi all items. 2019. Available at https://stats.oecd.org/. Accessed 11 June 2020.
Bot Plus Web. Base de datos de medicamentos del consejo general de farmacéuticos (bot plus 2.0). 2021. Available at http://www.oblikue.com/bddcostes/. Accessed 21 June 2021.
Mata-Cases M, Franch-Nadal J, Real J, Mauricio D. Glycaemic control and antidiabetic treatment trends in primary care centres in patients with type 2 diabetes mellitus during 2007–2013 in Catalonia: a population-based study. BMJ Open. 2016;6:e012463.
Yurgin NR, Boye KS, Dilla T, Suriñach NL, Llach XB. Physician and patient management of type 2 diabetes and factors related to glycemic control in Spain. Patient Prefer Adherence. 2008;2:87–95.
Jakubczyk M, Lipka I, Pawęska J, et al. Cost of severe hypoglycaemia in nine European countries. J Med Econ. 2016;19:973–82.
Caballero-Corchuelo J, Guerrero-Pérez F, García-Sancho-de-la-Jordana P, Pérez-Maraver M. Analysis of the characteristics of patients with diabetes mellitus who attend a tertiary hospital emergency department for a hypoglycemic event. Endocrinol Diabetes Nutr. 2019;66:19–25.
Arroyo-Borrell E, Renart-Vicens G, Saez M, Carreras M. Hospital costs of foreign non-resident patients: a comparative analysis in Catalonia, Spain. Int J Environ Res Public Health. 2017;14:1062.
Gisbert R, Brosa M. Spanish health costs and cost-effectiveness ratios database: Esalud. Barcelona: Oblikue Consulting; 2007.
Isla Pera P, Palacin Lois M, López Matheu C, et al. Perceptions of diabetes obtained through drawing in childhood and adolescence. Patient Prefer Adherence. 2013;7:595–605.
Soriguer F, Goday A, Bosch-Comas A, et al. Prevalence of diabetes mellitus and impaired glucose regulation in Spain: The diabetes study. Diabetologia. 2012;55:88–93.
International Diabetes Federation. IDF diabetes atlas, 8th edition. Brussels, Belgium: International Diabetes Federation; 2017.
Colino E, Martín-Frías M, Yelmo R, Álvarez M, Roldán B, Barrio R. Impact of insulin pump therapy on long-term glycemic control in a pediatric Spanish cohort. Diabetes Res Clin Pract. 2016;113:69–76.
Orozco-Beltrán D, Mezquita-Raya P, Ramírez-deArellano A, Galán M. Self-reported frequency and impact of hypoglycemic events in Spain. Diabetes Ther. 2014;5:155–68.
Orozco-Beltrán D, Sánchez E, Garrido A, Quesada JA, Carratalá-Munuera MC, Gil-Guillén VF. Evolución de la mortalidad por diabetes mellitus en España: Análisis del periodo 1998–2013. Rev Esp Cardiol. 2017;70:433–43.
Acknowledgements
Funding
This work and associated article processing charges, including the journal’s Rapid Service Fee, were fully funded by Eli Lilly and Company.
Medical Writing, Editorial, and Other Assistance
The authors would like to thank Jennifer Redondo, intern at the HO-RWE department in Eli Lilly and Company, for reviewing and for providing feedback on all drafts of this article; Dr. Marsha Tracey, an employee of Eli Lilly and Company, for providing project management support, medical writing and editorial assistance in the preparation of this article; and Dana Schamberger, an employee of Syneos Health, for providing editorial assistance.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work, and have given their approval for this version to be published.
Author Contributions
Beatrice Osumili made substantial contributions to the conception of the work, the design of the work, the acquisition of data, the analysis of data, the interpretation of data and the drafting of the manuscript. Esther Artime made substantial contributions to the acquisition of data, the analysis of data, the interpretation of data and critical revision of the manuscript for important intellectual content. Beth Mitchell made substantial contributions to the interpretation of data and critical revision of the manuscript for important intellectual content. Miriam Rubio-de Santos made substantial contributions to the conception of the work, the design of the work, the interpretation of data and critical revision of the manuscript for important intellectual content. Silvia Díaz-Cerezo made substantial contributions to the conception of the work, the design of the work, the interpretation of data and critical revision of the manuscript for important intellectual content. Marga Giménez made substantial contributions to the interpretation of data and critical revision of the manuscript for important intellectual content. Erik Spaepen made substantial contributions to the analysis of data, the interpretation of data and critical revision of the manuscript for important intellectual content. Helen Sharland made substantial contributions to the interpretation of data, the drafting and critical revision of the manuscript for important intellectual content. William J Valentine made substantial contributions to the analysis of data, the interpretation of data and critical revision of the manuscript for important intellectual content.
Prior Presentation
This manuscript is based on an abstract and corresponding poster presented at the XXXII Sociedad Española de Diabetes (SED) virtual congress, 16–18 June 2021. The abstract is publicly available via the online version of SED 2021 congress abstract book: Foster C, Osumili B, Giménez M, et al. Cost of severe hypoglycemia and budgetary impact of nasal glucagon in patients with diabetes in Spain. XXXII Congreso Nacional de la Sociedad Española de Diabetes. Virtual: Endocrinología, Diabetes y Nutrición 2021, p 005.
Disclosures
Beatrice Osumili is a full-time employee of Eli Lilly and Company. Esther Artime, Beth Mitchell, Miriam Rubio-de Santos, and Silvia Díaz-Cerezo are employees of and minor shareholders in Eli Lilly and Company. Marga Giménez has received lecturing and consulting fees from Eli Lilly and Company, Medtronic Inc., Novo Nordisk A/S, Sanofi-Aventis, AstraZeneca and MSD. Erik Spaepen is a consultant to Eli Lilly and Company. Helen Sharland is a full-time employee of Ossian Health Economics and Communications, which received consulting fees from Eli Lilly and Company for model development and for performing the analysis described in this paper. William J Valentine is a full-time employee of Ossian Health Economics and Communications, which received consulting fees from Eli Lilly and Company for model development and for performing the analysis described in this paper.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available; the data used to develop and populate the model are all publicly available and referenced in the article. The model itself is proprietary and will not be made publicly available. Results of the study are presented in full in the article.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Osumili, B., Artime, E., Mitchell, B. et al. Cost of Severe Hypoglycemia and Budget Impact with Nasal Glucagon in Patients with Diabetes in Spain. Diabetes Ther 13, 775–794 (2022). https://doi.org/10.1007/s13300-022-01238-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-022-01238-8